Unit 2
... O-H and N-H Stretching • Both of these occur around 3300 cm-1, but they look different. Alcohol O-H, broad with rounded tip. Secondary amine (R2NH), broad with one ...
... O-H and N-H Stretching • Both of these occur around 3300 cm-1, but they look different. Alcohol O-H, broad with rounded tip. Secondary amine (R2NH), broad with one ...
ExamView - 2002 AP Chemistry Exam.tst
... Web or Mass distribution prohibited. (2) AP® is a registered trademark of the College Entrance Examination Board. The College Entrance Examination Board was not involved in the production of and does not endorse this product. Permission is granted for individual classroom teachers to reproduce the a ...
... Web or Mass distribution prohibited. (2) AP® is a registered trademark of the College Entrance Examination Board. The College Entrance Examination Board was not involved in the production of and does not endorse this product. Permission is granted for individual classroom teachers to reproduce the a ...
01. Introduction of bioorganic chemistry. Classification, structure
... Classification of Alcohols Primary – carbon atom to which the –OH group is attached is directly bonded to one other carbon atom Secondary – carbon atom to which the –OH group is attached is directly bonded to two other carbon atom Tertiary – carbon atom to which the –OH group is attached is dire ...
... Classification of Alcohols Primary – carbon atom to which the –OH group is attached is directly bonded to one other carbon atom Secondary – carbon atom to which the –OH group is attached is directly bonded to two other carbon atom Tertiary – carbon atom to which the –OH group is attached is dire ...
Forward
... n contrast to alcohols with their rich chemical reactivity, ethers (compounds containing a C±O±C unit) undergo relatively few chemical reactions. As you saw when we discussed Grignard reagents in Chapter 14 and lithium aluminum hydride reductions in Chapter 15, this lack of reactivity of ethers make ...
... n contrast to alcohols with their rich chemical reactivity, ethers (compounds containing a C±O±C unit) undergo relatively few chemical reactions. As you saw when we discussed Grignard reagents in Chapter 14 and lithium aluminum hydride reductions in Chapter 15, this lack of reactivity of ethers make ...
Exam 980415 - NTOU-Chem
... 2) At 25.0°C the enthalpy of vaporization of water ( Hvap) is 44.0 kJ/mol and the vapor pressure is 23.8 mm Hg. Calculate the vapor pressure of water at 50.0°C, in mm Hg. A) 6.02 B) 147 C) 58.2 D) 564 E) 94.1 Answer: E 3) Which of the following describes what happens to the solubility of a slightly ...
... 2) At 25.0°C the enthalpy of vaporization of water ( Hvap) is 44.0 kJ/mol and the vapor pressure is 23.8 mm Hg. Calculate the vapor pressure of water at 50.0°C, in mm Hg. A) 6.02 B) 147 C) 58.2 D) 564 E) 94.1 Answer: E 3) Which of the following describes what happens to the solubility of a slightly ...
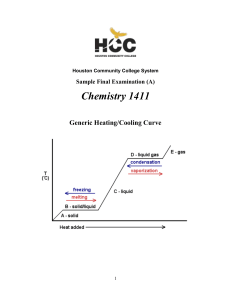
1411FINALSAMPLEs and Key
... CHEM 1411 SAMPLE FINAL EXAM (A) PART I - Multiple Choice (2 points each) _____ 1. The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses this distance in meters? A. 1.34 x 10–13 m ...
... CHEM 1411 SAMPLE FINAL EXAM (A) PART I - Multiple Choice (2 points each) _____ 1. The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses this distance in meters? A. 1.34 x 10–13 m ...
CHAPTER 8 Structures and nomenclature of organic
... in chemical reactions. To identify each unique organic compound from the millions of compounds, chemists use a formal set of nomenclatural rules agreed upon by an international committee. Many of the organic substances that we come across in our everyday lives are known to us by their common names, ...
... in chemical reactions. To identify each unique organic compound from the millions of compounds, chemists use a formal set of nomenclatural rules agreed upon by an international committee. Many of the organic substances that we come across in our everyday lives are known to us by their common names, ...
Chapter 14: Chemical Kinetics
... For a reaction to occur as a result of a specific collision, the collision must have enough energy to overcome the energy barrier (activation energy). As you saw in Gases (Unit 10) and Intermolecular Forces and the Liquid State (Unit 11), the molecules in a given sample have a Boltzmann distribution ...
... For a reaction to occur as a result of a specific collision, the collision must have enough energy to overcome the energy barrier (activation energy). As you saw in Gases (Unit 10) and Intermolecular Forces and the Liquid State (Unit 11), the molecules in a given sample have a Boltzmann distribution ...
REASONING QUESTIONS IN ORGANIC CHEMISTRY
... (ii) There are two –NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones. (iii) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed. Ans: ...
... (ii) There are two –NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones. (iii) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed. Ans: ...
Document
... Suppose we want DH for the reaction 2C(graphite) + O2(g) 2CO(g) It is difficult to measure directly. However, two other reactions are known: C(graphite) + O2(g) CO2(g); DH = -393.5 kJ 2CO2(g) 2CO(g) + O2(g); DH = – 566.0 kJ In order for these to add to give the reaction we want, we must multi ...
... Suppose we want DH for the reaction 2C(graphite) + O2(g) 2CO(g) It is difficult to measure directly. However, two other reactions are known: C(graphite) + O2(g) CO2(g); DH = -393.5 kJ 2CO2(g) 2CO(g) + O2(g); DH = – 566.0 kJ In order for these to add to give the reaction we want, we must multi ...
Chemistry - Andhra University
... D,L and R,S configuration for asymmetric and disymmetric molecules. Cahn-Ingold-Prelog rules. Racemic mixture- racemisation and resolution techniques. Diastereomers: definition- geometrical isomerism with reference to alkenes- cis, trans and E,Z- configuration. 4. General Principles of Inorganic qua ...
... D,L and R,S configuration for asymmetric and disymmetric molecules. Cahn-Ingold-Prelog rules. Racemic mixture- racemisation and resolution techniques. Diastereomers: definition- geometrical isomerism with reference to alkenes- cis, trans and E,Z- configuration. 4. General Principles of Inorganic qua ...
Amines, Amides, & Amino Acids
... • It’s like taking a carboxylic acid, RCOOH, and replacing the -OH with NH2. ...
... • It’s like taking a carboxylic acid, RCOOH, and replacing the -OH with NH2. ...
hydrolysis
... o They are polyhydroxy aldehydes and ketones . o Carbohydrates are compounds of C, H, and O. ...
... o They are polyhydroxy aldehydes and ketones . o Carbohydrates are compounds of C, H, and O. ...
Chapter 10:Alcohols, Phenols and Ethers
... Phenol groups can be incorporated into an aromatic ring by sulfonation of the aromatic rings followed by melting the product with sodium hydroxide to convert the sulfonic acid group to a phenol. The reaction conditions are harsh and only alkyl-substituted phenols can be prepared by this method. SO3H ...
... Phenol groups can be incorporated into an aromatic ring by sulfonation of the aromatic rings followed by melting the product with sodium hydroxide to convert the sulfonic acid group to a phenol. The reaction conditions are harsh and only alkyl-substituted phenols can be prepared by this method. SO3H ...
12 - Wiley
... 12 Reaction with Bases • Carboxylic acids react with sodium bicarbonate and sodium carbonate to form water-soluble salts and carbonic acid • carbonic acid, in turn, breaks down to carbon dioxide and water H2 O CH3 CO2 H + Na HCO3 ...
... 12 Reaction with Bases • Carboxylic acids react with sodium bicarbonate and sodium carbonate to form water-soluble salts and carbonic acid • carbonic acid, in turn, breaks down to carbon dioxide and water H2 O CH3 CO2 H + Na HCO3 ...
Elimination Reactions
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
Elimination Reactions
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
Chapter 21 - Jordi Alemany Sabater
... i) neutral salts (metal + non metal) containing sufixe-ur after the non metal: LiF……………..Florur Lithium CuBr…………..Bromide cupric CuBr2……….....Bromide cuprous K2S……………Sulphur. potassium ii) with respect to the nomenclature consisting of salts non metal+ is easy to understand: IBr3.......bromide iodin ...
... i) neutral salts (metal + non metal) containing sufixe-ur after the non metal: LiF……………..Florur Lithium CuBr…………..Bromide cupric CuBr2……….....Bromide cuprous K2S……………Sulphur. potassium ii) with respect to the nomenclature consisting of salts non metal+ is easy to understand: IBr3.......bromide iodin ...
Reactants Products
... reaction is second order in NO2. Between the second and third experiments, the concentration of NO2 stays constant, the concentration of CO doubles, and the rate remains constant (the small change in the least significant figure is simply experimental error), suggesting that the reaction is zero ord ...
... reaction is second order in NO2. Between the second and third experiments, the concentration of NO2 stays constant, the concentration of CO doubles, and the rate remains constant (the small change in the least significant figure is simply experimental error), suggesting that the reaction is zero ord ...
STUDY MATERIAL 2016-17 CHEMISTRY CLASS XII
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
Kinetics Workbook - School District 67
... This workbook will allow you to demonstrate your understanding of all aspects of the kinetics unit. The minimum expectation is that you do all of these questions by the due dates given by your teacher. Do the questions. Use your notes from class to assist you. Then after you have finished go to the ...
... This workbook will allow you to demonstrate your understanding of all aspects of the kinetics unit. The minimum expectation is that you do all of these questions by the due dates given by your teacher. Do the questions. Use your notes from class to assist you. Then after you have finished go to the ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.