M.Sc - I Syllabus w.e.f. June 2015
... determination from Freezing point depression and vapour-pressure measurements. Third law entropies- Calculations of standard entropies from Cp data, Exceptions to ...
... determination from Freezing point depression and vapour-pressure measurements. Third law entropies- Calculations of standard entropies from Cp data, Exceptions to ...
Binding and Hydrolysis of Meperidine by Human Liver
... 0.33-mm film thickness. The total ion chromatogram was monitored. The aqueous phase from the reaction mixture was evaporated to dryness. The residue was refluxed with 15 ml of methanol containing 10% sulfuric acid at 70°C for 5 h. The procedure modified meperidinic acid to its methyl ester. The solu ...
... 0.33-mm film thickness. The total ion chromatogram was monitored. The aqueous phase from the reaction mixture was evaporated to dryness. The residue was refluxed with 15 ml of methanol containing 10% sulfuric acid at 70°C for 5 h. The procedure modified meperidinic acid to its methyl ester. The solu ...
Which is Aromatic?
... 1930s: X-ray crystal structures showed benzene is planar and six carboncarbon bonds have the same length (C-C 1.39 A), which is shorter than C-C single bond at 1.54 A. But longer than a C=C. Thus, benzene does not have alternating bonds! ...
... 1930s: X-ray crystal structures showed benzene is planar and six carboncarbon bonds have the same length (C-C 1.39 A), which is shorter than C-C single bond at 1.54 A. But longer than a C=C. Thus, benzene does not have alternating bonds! ...
Fragmentation pathway for glutamine identification: Loss of 73 da
... proton affinities of these sites can be compared to those of CH3CONH2 (863.6 KJ/mol), CH3COOCH3 (821.6 KJ/mol), and (CH3)2NCH⫽N-CH3 (1002.5KJ/mol) respectively°[23].°The°amine°and°imine°nitrogens°have much higher proton affinities than the oxygens. The imine nitrogen could be the initial proton hold ...
... proton affinities of these sites can be compared to those of CH3CONH2 (863.6 KJ/mol), CH3COOCH3 (821.6 KJ/mol), and (CH3)2NCH⫽N-CH3 (1002.5KJ/mol) respectively°[23].°The°amine°and°imine°nitrogens°have much higher proton affinities than the oxygens. The imine nitrogen could be the initial proton hold ...
Ch-6-Alcohols and phenols - Home
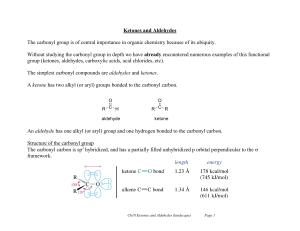
... STRUCTURE OF ALCOHOLS • Alcohols are the family of compounds that contain one or more hydroxyl (-OH) groups. The OH group is bound to a carbon atom. •They can be considered both derivatives of hydrocarbons (by replacing a hydrogen atom with a hydroxyl group -OH) and water (H2O) derivatives (the res ...
... STRUCTURE OF ALCOHOLS • Alcohols are the family of compounds that contain one or more hydroxyl (-OH) groups. The OH group is bound to a carbon atom. •They can be considered both derivatives of hydrocarbons (by replacing a hydrogen atom with a hydroxyl group -OH) and water (H2O) derivatives (the res ...
Hydrocarbons and Fuels - Deans Community High School
... • Carboxylic acids are used in household cleaning products including soap • In manufacture of important organic compounds), acetic anhydride (used in aspirins), cellulose acetate (used in synthetic fibres), various dyes, perfumes & medicines. • As a solvent it dissolves phosphorus, sulphur & iodine. ...
... • Carboxylic acids are used in household cleaning products including soap • In manufacture of important organic compounds), acetic anhydride (used in aspirins), cellulose acetate (used in synthetic fibres), various dyes, perfumes & medicines. • As a solvent it dissolves phosphorus, sulphur & iodine. ...
Preparation of d, l-Phenylalanine by Amidocarbonylation of Benzyl
... alcohol was not described before; the expected product is phenylacetic acid. It seems that in both cases the acetamide catalyzes the hydroformylation reaction in some way. The effect seems general for amides. It was found that in the presence of a stoichiometric amount of DMF rather than acetamide, ...
... alcohol was not described before; the expected product is phenylacetic acid. It seems that in both cases the acetamide catalyzes the hydroformylation reaction in some way. The effect seems general for amides. It was found that in the presence of a stoichiometric amount of DMF rather than acetamide, ...
physicochemical properties of organic medicinal agents
... In addition to hydrocarbon content, steric factors and electronic factors also play a part in the relative solubilities of ethers. Note that the cyclic ether tetrahydrofuran (THF) is significantly more water soluble than diethyl ether, even though both compounds have approximately the same hydrocarb ...
... In addition to hydrocarbon content, steric factors and electronic factors also play a part in the relative solubilities of ethers. Note that the cyclic ether tetrahydrofuran (THF) is significantly more water soluble than diethyl ether, even though both compounds have approximately the same hydrocarb ...
Esterification and Esters
... Applications of kinetic principles to industrial reactions are often useful. Initial kinetic studies of the esterification reaction are usually conducted on a small scale in a well stirred batch reactor (24). In many cases, results from batch studies can be used in the evaluation of the esterificati ...
... Applications of kinetic principles to industrial reactions are often useful. Initial kinetic studies of the esterification reaction are usually conducted on a small scale in a well stirred batch reactor (24). In many cases, results from batch studies can be used in the evaluation of the esterificati ...
boehm_rl
... atoms were removed by treatment with sodium iodide and sodium thiosulfate and because of spatial relations within the starch ring, the remaining chlorine atoms were assumed to have substituted carbon-bound hydrogen atoms on carbon atoms one and four. The work of Barham, et al. did not indicate that ...
... atoms were removed by treatment with sodium iodide and sodium thiosulfate and because of spatial relations within the starch ring, the remaining chlorine atoms were assumed to have substituted carbon-bound hydrogen atoms on carbon atoms one and four. The work of Barham, et al. did not indicate that ...
Stoichiometry: Predicting Amounts in Reactions
... 2. Measure out 2.0 g of copper (II) chloride. Record the actual amount used (ex. 2.01 or 2.08). Be sure to use the same balance for all measurements. 3. Record the mass of the aluminum wire. 4. Record the mass of the labeled cup. 5. Clamp a clean test tube to a ring stand. 6. Add 20.0 mL of distille ...
... 2. Measure out 2.0 g of copper (II) chloride. Record the actual amount used (ex. 2.01 or 2.08). Be sure to use the same balance for all measurements. 3. Record the mass of the aluminum wire. 4. Record the mass of the labeled cup. 5. Clamp a clean test tube to a ring stand. 6. Add 20.0 mL of distille ...
C 1 hapter
... most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through ...
... most useful reactions in organic synthesis. It provides a way of transforming alkenes into many different types of C*-X and C*-R bonds through the optically enriched organoboron adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through ...
Microsoft Word
... based ligands are most widely used with copper catalyst systems. Phosphorous-based ligands in conjunction with rhenium, ruthenium, iron, rhodium, nickel and palladium have also been studied. The type of ligand structure is important, as they could fine-tune selectivities and force the complex to par ...
... based ligands are most widely used with copper catalyst systems. Phosphorous-based ligands in conjunction with rhenium, ruthenium, iron, rhodium, nickel and palladium have also been studied. The type of ligand structure is important, as they could fine-tune selectivities and force the complex to par ...
Spring 2005
... 14. (3 pts) What is the oxidation number (charge) on C in H2C2O4? Explain your reasoning. 15. (8 pts) How many atoms of nitrogen are there in 15.3 mg of Ba(NO3)2? 16. (8 pts) What is the mass percent of oxygen in KMnO4? 17. (8 pts) The balanced reaction of muriatic acid with lime is 2 HCl + CaO ! Ca ...
... 14. (3 pts) What is the oxidation number (charge) on C in H2C2O4? Explain your reasoning. 15. (8 pts) How many atoms of nitrogen are there in 15.3 mg of Ba(NO3)2? 16. (8 pts) What is the mass percent of oxygen in KMnO4? 17. (8 pts) The balanced reaction of muriatic acid with lime is 2 HCl + CaO ! Ca ...
New Applications for Sulfur-Based Leaving Groups in Synthesis
... I’d firstly like to thank my supervisor, Dr. Jon Wilden for his support throughout the past four years. It has been great fun to be able to work on a varied range of chemistry within his group and his stress-free approach to the subject has allowed me to form many of my own ideas and not be afraid t ...
... I’d firstly like to thank my supervisor, Dr. Jon Wilden for his support throughout the past four years. It has been great fun to be able to work on a varied range of chemistry within his group and his stress-free approach to the subject has allowed me to form many of my own ideas and not be afraid t ...
Thermodynamics and Equilibrium
... Standard Entropies and the Third Law of Thermodynamics • The third law of thermodynamics states that a substance that is perfectly crystalline at 0 K has an entropy of zero. – When temperature is raised, however, the substance becomes more disordered as it absorbs heat. – The entropy of a substance ...
... Standard Entropies and the Third Law of Thermodynamics • The third law of thermodynamics states that a substance that is perfectly crystalline at 0 K has an entropy of zero. – When temperature is raised, however, the substance becomes more disordered as it absorbs heat. – The entropy of a substance ...
OCR answers to the examination questions File
... © Pearson Education Ltd 2008 This document may have been altered from the original ...
... © Pearson Education Ltd 2008 This document may have been altered from the original ...
Chemical Reactions and Solution Stoichiometry
... materials, the products. A combination reaction is one in which typically two or more reactants, usually elements or compounds, combine to form one product, usually a compound. In one type of combination reaction, two elements combine to form a compound. For example, the elements hydrogen and oxygen ...
... materials, the products. A combination reaction is one in which typically two or more reactants, usually elements or compounds, combine to form one product, usually a compound. In one type of combination reaction, two elements combine to form a compound. For example, the elements hydrogen and oxygen ...
kinetic and thermodynamic studies of the oxidation of perfumery
... *E-mail: [email protected] ABSTRACT Oxidation is one of the most important industrial reactions as it yields useful products. Literature survey indicates the use of a variety of organic oxidants for the oxidation of alcohols to the corresponding carbonyl compounds but inorganic oxidants have rare ...
... *E-mail: [email protected] ABSTRACT Oxidation is one of the most important industrial reactions as it yields useful products. Literature survey indicates the use of a variety of organic oxidants for the oxidation of alcohols to the corresponding carbonyl compounds but inorganic oxidants have rare ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.