Carboxylic acids and derivatives
... • Usually solids with no odor. • Carboxylate salts of Na+, K+, Li+, and NH4+ are soluble in water. • Soap is the soluble sodium salt of a long chain fatty acid. • Salts can be formed by the reaction of an acid with NaHCO3, releasing CO2. ...
... • Usually solids with no odor. • Carboxylate salts of Na+, K+, Li+, and NH4+ are soluble in water. • Soap is the soluble sodium salt of a long chain fatty acid. • Salts can be formed by the reaction of an acid with NaHCO3, releasing CO2. ...
Amino Acids
... proteins, utilizes separation based on a particular pH and the isoelectric points of amino acids. By varying the pH of a system, the charges on the amino acids will vary from positive to neutral to negative and separation can be achieved. The order in which amino acids are found in a protein molecul ...
... proteins, utilizes separation based on a particular pH and the isoelectric points of amino acids. By varying the pH of a system, the charges on the amino acids will vary from positive to neutral to negative and separation can be achieved. The order in which amino acids are found in a protein molecul ...
CHANNELING OF SUBSTRATES AND INTERMEDIATES IN
... by linker peptides (31–33). Direct physical evidence for the tunneling of indole in tryptophan synthase was first provided by the elegant X-ray structural analyses of the enzyme from Salmonella typhimurium (34, 35). As can be seen in Figure 1, the quaternary structure of the (αβ)2 complex is nearly ...
... by linker peptides (31–33). Direct physical evidence for the tunneling of indole in tryptophan synthase was first provided by the elegant X-ray structural analyses of the enzyme from Salmonella typhimurium (34, 35). As can be seen in Figure 1, the quaternary structure of the (αβ)2 complex is nearly ...
New insights into the mechanism of sorbitol transformation
... Sorbitol can be selectively transformed into liquid alkanes over a bifunctional catalytic system Pt/ZrO2 + TiO2–WOx. In this paper, we investigated the reaction mechanism by carefully analyzing the numerous products issued from sorbitol and by studying the reactivity of some identified intermediates ...
... Sorbitol can be selectively transformed into liquid alkanes over a bifunctional catalytic system Pt/ZrO2 + TiO2–WOx. In this paper, we investigated the reaction mechanism by carefully analyzing the numerous products issued from sorbitol and by studying the reactivity of some identified intermediates ...
Unit 12: Electrochemistry
... Watch Crash Course Chemistry Redox Reactions YouTube video https://www.youtube.com/watch?v=lQ6FBA1HM3s&list=PL8dPuuaLjXtPHzzYuWy6fYEaX9mQQ8oGr&index=10 ...
... Watch Crash Course Chemistry Redox Reactions YouTube video https://www.youtube.com/watch?v=lQ6FBA1HM3s&list=PL8dPuuaLjXtPHzzYuWy6fYEaX9mQQ8oGr&index=10 ...
Nucleophilic Acyl Substitution
... depends on the relative basicities of the two groups: The weaker the basicity of a group, the better its leaving ability. Recall from Section 10.3 that weak bases are good leaving groups because weak bases do not share their electrons as well as strong bases do. The pKa values of the conjugate acids ...
... depends on the relative basicities of the two groups: The weaker the basicity of a group, the better its leaving ability. Recall from Section 10.3 that weak bases are good leaving groups because weak bases do not share their electrons as well as strong bases do. The pKa values of the conjugate acids ...
1411FINALSAMPLE+KEY - Houston Community College
... CHEM 1411 SAMPLE FINAL EXAM (A) PART I - Multiple Choice (2 points each) _____ 1. The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses this distance in meters? A. 1.34 x 10–13 m ...
... CHEM 1411 SAMPLE FINAL EXAM (A) PART I - Multiple Choice (2 points each) _____ 1. The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses this distance in meters? A. 1.34 x 10–13 m ...
CHEM-1411 Final Practice Exam
... CHEM 1411 SAMPLE FINAL EXAM (A) PART I - Multiple Choice (2 points each) _____ 1. The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses this distance in meters? ...
... CHEM 1411 SAMPLE FINAL EXAM (A) PART I - Multiple Choice (2 points each) _____ 1. The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses this distance in meters? ...
Organic for Forensic Science
... IUPAC Names for Ketones • Replace -e with -one. Indicate the position of the carbonyl with a number. • Number the chain so that carbonyl carbon has the lowest number. • For cyclic ketones the carbonyl carbon is assigned the number 1. ...
... IUPAC Names for Ketones • Replace -e with -one. Indicate the position of the carbonyl with a number. • Number the chain so that carbonyl carbon has the lowest number. • For cyclic ketones the carbonyl carbon is assigned the number 1. ...
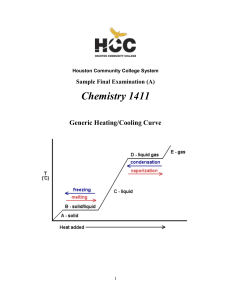
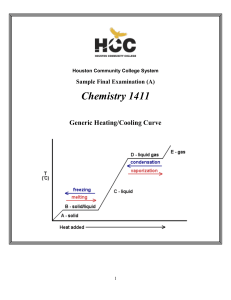
The Chemistry of Aqueous Systems
... P1 V1 P2 V2 (4.42 atm) (1 L) V1 X 4.42 L of air 1atm Solution: First, set up Boyle's Law equation: P1 equals 1 atm; V1 equals "X", P2 equals 65 psi and V2 equals 1 L. Remember, though, that you may not mix units. To bypass this fiasco, convert the 65 psi into atm (4.42 atm). Rearrange the eq ...
... P1 V1 P2 V2 (4.42 atm) (1 L) V1 X 4.42 L of air 1atm Solution: First, set up Boyle's Law equation: P1 equals 1 atm; V1 equals "X", P2 equals 65 psi and V2 equals 1 L. Remember, though, that you may not mix units. To bypass this fiasco, convert the 65 psi into atm (4.42 atm). Rearrange the eq ...
Catalytic Nucleophilic Fluorination of Secondary and Tertiary
... room temperature, 24 h; [f] [Cp*Ru(cod)Cl] (5 mol %), N,N-bis(propargyl)toluenesulfonamide, DCE, room temperature, 15 min. cod = 1,5cycloctadiene, Cp* = 1,2,3,4,5-pentamethylcyclopentadienyl, DCE = 1,2dichloroethane. ...
... room temperature, 24 h; [f] [Cp*Ru(cod)Cl] (5 mol %), N,N-bis(propargyl)toluenesulfonamide, DCE, room temperature, 15 min. cod = 1,5cycloctadiene, Cp* = 1,2,3,4,5-pentamethylcyclopentadienyl, DCE = 1,2dichloroethane. ...
Activity 1: Chapter 1: Carbon Compounds and Chemical Bonds
... 13. Mono-substituted cycloalkanes do not include the “1” locant. Thus, 1-methylcyclohexane is inaccurate. Draw methylcyclohexane; explain why adding a locant does not add any new information. D. Nomenclature of Alkenes: Functional Group = the particular group of atoms in a molecule that primarily de ...
... 13. Mono-substituted cycloalkanes do not include the “1” locant. Thus, 1-methylcyclohexane is inaccurate. Draw methylcyclohexane; explain why adding a locant does not add any new information. D. Nomenclature of Alkenes: Functional Group = the particular group of atoms in a molecule that primarily de ...
Collins CSEC® Chemistry Workbook answers A1 States of matter
... iii) The particles move around rapidly and have weak forces of attraction between them, so they spread out to fill any available space in the container. (2) ...
... iii) The particles move around rapidly and have weak forces of attraction between them, so they spread out to fill any available space in the container. (2) ...
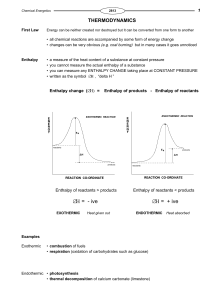
Chapter 8
... conservation of electrical charge (same total charge on the reactant and product side). This means that chemical reactions must be consistent with Dalton’s theory. ...
... conservation of electrical charge (same total charge on the reactant and product side). This means that chemical reactions must be consistent with Dalton’s theory. ...
- Angelo State University
... Chemical Reactions and Chemical Equations • A chemical reaction occurs when atoms of different elements combine and create a new chemical compound, with properties which may be completely unlike those of its constituent elements. • A chemical reaction is written in a standard format called a chemica ...
... Chemical Reactions and Chemical Equations • A chemical reaction occurs when atoms of different elements combine and create a new chemical compound, with properties which may be completely unlike those of its constituent elements. • A chemical reaction is written in a standard format called a chemica ...
1 Iron Complexes in Organic Chemistry
... tetracarbonylferrate (Collmans reagent) and provides unsymmetrical ketones (Scheme 1.2) [19, 20]. Collmans reagent is extremely sensitive towards air and moisture, but offers a great synthetic potential as carbonyl transfer reagent. It can be prepared by an in situ procedure starting from Fe(CO)5 ...
... tetracarbonylferrate (Collmans reagent) and provides unsymmetrical ketones (Scheme 1.2) [19, 20]. Collmans reagent is extremely sensitive towards air and moisture, but offers a great synthetic potential as carbonyl transfer reagent. It can be prepared by an in situ procedure starting from Fe(CO)5 ...
chapter 16
... Thus, as the reaction begins, an input of energy is necessary to produce the activated complex; as the reaction proceeds, and the system shifts from the activated complex to products, energy is released. In a chemical reaction, the minimum energy necessary for reaching the activated complex and proc ...
... Thus, as the reaction begins, an input of energy is necessary to produce the activated complex; as the reaction proceeds, and the system shifts from the activated complex to products, energy is released. In a chemical reaction, the minimum energy necessary for reaching the activated complex and proc ...
Branched-Chain Alkanes
... – However not all compounds classified has aromatic have pleasant odors, or any odor at all. ...
... – However not all compounds classified has aromatic have pleasant odors, or any odor at all. ...
The First Chiral Organometallic Triangle for Asymmetric Catalysis
... Enantiomerically pure atropisomeric 6,6′-dichloro-2,2′-diethoxy1,1′-binaphthyl-4,4′-bis(acetylene) L1 was synthesized in two steps in 67% overall yield starting from 6,6′-dichloro-4,4′-dibromo-2,2′diethoxy-1,1′-binaphthalene which was previously reported by us (Scheme 1).10a Dimethoxy analogue L2 an ...
... Enantiomerically pure atropisomeric 6,6′-dichloro-2,2′-diethoxy1,1′-binaphthyl-4,4′-bis(acetylene) L1 was synthesized in two steps in 67% overall yield starting from 6,6′-dichloro-4,4′-dibromo-2,2′diethoxy-1,1′-binaphthalene which was previously reported by us (Scheme 1).10a Dimethoxy analogue L2 an ...
California Standards Practice - Student Edition
... a. Students know large molecules (polymers), such as proteins, nucleic acids, and starch, are formed by repetitive combinations of simple subunits. b. Students know the bonding characteristics of carbon that result in the formation of a large variety of structures ranging from simple hydrocarbons to ...
... a. Students know large molecules (polymers), such as proteins, nucleic acids, and starch, are formed by repetitive combinations of simple subunits. b. Students know the bonding characteristics of carbon that result in the formation of a large variety of structures ranging from simple hydrocarbons to ...
Workshop #1 Part 1. Organic Chemistry Nomenclature
... 13. Mono-substituted cycloalkanes do not include the “1” locant. Thus, 1-methylcyclohexane is inaccurate. Draw methylcyclohexane; explain why adding a locant does not add any new information. D. Nomenclature of Alkenes: Functional Group = the particular group of atoms in a molecule that primarily de ...
... 13. Mono-substituted cycloalkanes do not include the “1” locant. Thus, 1-methylcyclohexane is inaccurate. Draw methylcyclohexane; explain why adding a locant does not add any new information. D. Nomenclature of Alkenes: Functional Group = the particular group of atoms in a molecule that primarily de ...
CHAPTER 15 ACIDS AND BASES
... where c represents the equilibrium hydrogen ion concentration found in the first stage. If c >> Ka2, we can assume (c ± y) ≈ c, and consequently y = Ka2. Is this conclusion also true for the second stage ionization of a triprotic acid like H3PO4? ...
... where c represents the equilibrium hydrogen ion concentration found in the first stage. If c >> Ka2, we can assume (c ± y) ≈ c, and consequently y = Ka2. Is this conclusion also true for the second stage ionization of a triprotic acid like H3PO4? ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.