Unit 3: 1 Equilibrium and the Constant, K
... environmental processes that are reversible, construct an explanation that connects the observations to the reversibility of the underlying chemical reactions or processes. [See SP 6.2; Essential knowledge 6.A.1] Learning objective 6.2 The student can, given a manipulation of a chemical reaction or ...
... environmental processes that are reversible, construct an explanation that connects the observations to the reversibility of the underlying chemical reactions or processes. [See SP 6.2; Essential knowledge 6.A.1] Learning objective 6.2 The student can, given a manipulation of a chemical reaction or ...
(III) ion and a cobalt (II) - Iowa State University Digital Repository
... of the lower figure (240 nm) were collected in 1 second and the second fifty collected in 119 seconds. The family of traces results from stopped-flow experiments performed on the same solution at various times over a 2 hr period (t = 0 hr (A), t = 2 hr (B)) ...
... of the lower figure (240 nm) were collected in 1 second and the second fifty collected in 119 seconds. The family of traces results from stopped-flow experiments performed on the same solution at various times over a 2 hr period (t = 0 hr (A), t = 2 hr (B)) ...
CARBOXYLIC ACIDS
... (1) Reactions at the acidic hydrogen on the carboxyl group. (2) Reactions at the carbonyl group (3) Reactions at the carboxylate oxygen (4) Reactions that lead to loss of the carboxyl group as CO2 ...
... (1) Reactions at the acidic hydrogen on the carboxyl group. (2) Reactions at the carbonyl group (3) Reactions at the carboxylate oxygen (4) Reactions that lead to loss of the carboxyl group as CO2 ...
CHAPTER 5
... 1-Butene has an unbranched carbon chain with a double bond between C-1 and C-2. It is a constitutional isomer of the other three. Similarly, 2-methylpropene, with a branched carbon chain, is a constitutional isomer of the other three. The pair of isomers designated cis- and trans-2-butene have the s ...
... 1-Butene has an unbranched carbon chain with a double bond between C-1 and C-2. It is a constitutional isomer of the other three. Similarly, 2-methylpropene, with a branched carbon chain, is a constitutional isomer of the other three. The pair of isomers designated cis- and trans-2-butene have the s ...
Abdullah F. Eid
... be easily removed on heating, whereby the acid strength is increased due to the dehydration of protons. This is a reversible process accompanied by changing the volume of crystal cell. Unlike the rigid network structure of zeolites, in HPA crystal the Keggin anions are quite mobile. Not only water b ...
... be easily removed on heating, whereby the acid strength is increased due to the dehydration of protons. This is a reversible process accompanied by changing the volume of crystal cell. Unlike the rigid network structure of zeolites, in HPA crystal the Keggin anions are quite mobile. Not only water b ...
1H-Imidazol-4(5H)-ones and thiazol-4(5H)
... prior trimethylsilyl enol ether formation which is necessary to avoid O-alkylation. The starting thiohydantoins, in turn, are obtained by heating a mixture of the corresponding N-substituted amino acid or its methyl ester 3 and thiourea at 195 °C [59] (Scheme 1b). Following this protocol different 1 ...
... prior trimethylsilyl enol ether formation which is necessary to avoid O-alkylation. The starting thiohydantoins, in turn, are obtained by heating a mixture of the corresponding N-substituted amino acid or its methyl ester 3 and thiourea at 195 °C [59] (Scheme 1b). Following this protocol different 1 ...
Document
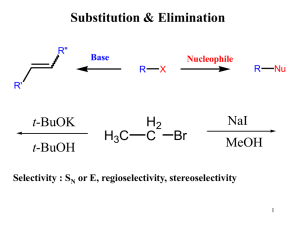
... Nucleophilic attack of ¯OCH3 occurs from the backside at either C—O bond, because both ends are similarly substituted. Since attack at either side occurs with equal probability, an equal amount of the two enantiomers (i.e., a ...
... Nucleophilic attack of ¯OCH3 occurs from the backside at either C—O bond, because both ends are similarly substituted. Since attack at either side occurs with equal probability, an equal amount of the two enantiomers (i.e., a ...
dr.ebtehal Lec3
... Oxidation is the beginning of the deterioration process. Think of how a slice of apple turns brown when exposed to air. Oxidation leads to the formation of free radicals which are unstable molecules in the body that have one unpaired electron. They can cause oxidation and damage to the cells. This ...
... Oxidation is the beginning of the deterioration process. Think of how a slice of apple turns brown when exposed to air. Oxidation leads to the formation of free radicals which are unstable molecules in the body that have one unpaired electron. They can cause oxidation and damage to the cells. This ...
Immobilization and stability studies of a lipase from
... Lipases, as a class of enzymes, are stable and extremely valuable catalysts for many practical/industrial applications (Bjorkling et al. 1991). They have been used to generate chiral entities from alcohols, carboxylic acid esters, cyanohydrins, chlorohydrins, diols, amines, diamines and amino alcoho ...
... Lipases, as a class of enzymes, are stable and extremely valuable catalysts for many practical/industrial applications (Bjorkling et al. 1991). They have been used to generate chiral entities from alcohols, carboxylic acid esters, cyanohydrins, chlorohydrins, diols, amines, diamines and amino alcoho ...
A. 3-chloro-1-ethoxybutane B. p
... a. Hydrogen bonding between like molecules (aldehyde with aldehyde) b. Hydrogen bonding with water c. Dipolar bonding between like molecules (aldehyde with aldehyde) d. Both b and c e. Both a and c ANS: D ...
... a. Hydrogen bonding between like molecules (aldehyde with aldehyde) b. Hydrogen bonding with water c. Dipolar bonding between like molecules (aldehyde with aldehyde) d. Both b and c e. Both a and c ANS: D ...
CHAPTER 4 - Myschoolpages.com
... Increasing concentration of acid (H+) decreases concentration of base (OH-). Increasing concentration of base (OH-) decreases the concentration of acid (H+) Strong and Weak Acids and Bases - Strong indicates complete dissociation (strong electrolyte) and weak refers to only partial dissociation ...
... Increasing concentration of acid (H+) decreases concentration of base (OH-). Increasing concentration of base (OH-) decreases the concentration of acid (H+) Strong and Weak Acids and Bases - Strong indicates complete dissociation (strong electrolyte) and weak refers to only partial dissociation ...
lec-3- 211( Elim+ Re..
... Oxidation is the beginning of the deterioration process. Think of how a slice of apple turns brown when exposed to air. Oxidation leads to the formation of free radicals which are unstable molecules in the body that have one unpaired electron. They can cause oxidation and damage to the cells. This ...
... Oxidation is the beginning of the deterioration process. Think of how a slice of apple turns brown when exposed to air. Oxidation leads to the formation of free radicals which are unstable molecules in the body that have one unpaired electron. They can cause oxidation and damage to the cells. This ...
Chapter 9 - ComSizo.com.br
... The reason is that: the first step can occur equally well from either face of the double bond, to give a 50:50 mixture of enantiomeric bromonium ions. Because the bromonium ions do not have symmetry planes, reaction on the left and right is not equally likely, so an unequal mix of (2S,3R) or (2R,3S) ...
... The reason is that: the first step can occur equally well from either face of the double bond, to give a 50:50 mixture of enantiomeric bromonium ions. Because the bromonium ions do not have symmetry planes, reaction on the left and right is not equally likely, so an unequal mix of (2S,3R) or (2R,3S) ...
Complete Solution Manual
... molecules, leading to weaker LD forces and lower boiling points. All the function groups in Table 22.4 have a very electronegative atom bonded to the carbon chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Mo ...
... molecules, leading to weaker LD forces and lower boiling points. All the function groups in Table 22.4 have a very electronegative atom bonded to the carbon chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Mo ...
x - SharpSchool
... a pair of substances that differ only by a proton is called a conjugate acid-base pair…the acid is on one side of the reaction and the base is on the other in general, the reaction can be shown as follows: ...
... a pair of substances that differ only by a proton is called a conjugate acid-base pair…the acid is on one side of the reaction and the base is on the other in general, the reaction can be shown as follows: ...
CHAPTER 22 ORGANIC AND BIOLOGICAL MOLECULES 1
... molecules, leading to weaker LD forces and lower boiling points. All the function groups in Table 22.4 have a very electronegative atom bonded to the carbon chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Mo ...
... molecules, leading to weaker LD forces and lower boiling points. All the function groups in Table 22.4 have a very electronegative atom bonded to the carbon chain in the compound. This creates bond dipoles in the molecule leading to a polar molecule which exhibits additional dipole-dipole forces. Mo ...
Transition Metal Reagents and Catalysts
... important method of commercial production of synthetic oil. In 1938 RoÈlen, one of Fischer's co-workers, tried the reaction of alkene with synthesis gas using a Co catalyst and found the formation of aldehydes. This reaction is now called the oxo reaction or hydroformylation, because hydrogen and a ...
... important method of commercial production of synthetic oil. In 1938 RoÈlen, one of Fischer's co-workers, tried the reaction of alkene with synthesis gas using a Co catalyst and found the formation of aldehydes. This reaction is now called the oxo reaction or hydroformylation, because hydrogen and a ...
File
... 5. Explain the term “homologous series.” 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsa ...
... 5. Explain the term “homologous series.” 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsa ...
Chapter 1 - University of Amsterdam
... combination with convenient ligand-field approximations the thus obtained d-electron configuration is a quite useful concept, applicable for spectroscopic interpretations or predictions of the magnetic behavior of these complexes. However, for complexes bearing redox active ligands, formal oxidation ...
... combination with convenient ligand-field approximations the thus obtained d-electron configuration is a quite useful concept, applicable for spectroscopic interpretations or predictions of the magnetic behavior of these complexes. However, for complexes bearing redox active ligands, formal oxidation ...
Alkenes
... Orbital Overlap In Ethene In ethylene (ethene), each carbon atom use an sp2 orbital to form a single C-C bond. Because of the two sp2 orbitals overlap by endto- end the resulting bond is called σ bond. The pi (π) bond between the two carbon atoms is formed by side- by-side overlap of the two unhyb ...
... Orbital Overlap In Ethene In ethylene (ethene), each carbon atom use an sp2 orbital to form a single C-C bond. Because of the two sp2 orbitals overlap by endto- end the resulting bond is called σ bond. The pi (π) bond between the two carbon atoms is formed by side- by-side overlap of the two unhyb ...
Synthetic Polymers
... always bonded to the $-carbon of the next, as shown in the following partial structure for polystyrene. The polymer is said to be formed by head-to-tail bonding of the monomer units. ...
... always bonded to the $-carbon of the next, as shown in the following partial structure for polystyrene. The polymer is said to be formed by head-to-tail bonding of the monomer units. ...
20.2 Oxidation Numbers
... If your car is exposed to salt on the roads in the winter, why is it important to wash the salt off your car? Many parts of your car’s body are made of steel, an iron alloy. The salt can cause the metal to corrode, or rust, faster than it would otherwise because when mixed with water, the salt creat ...
... If your car is exposed to salt on the roads in the winter, why is it important to wash the salt off your car? Many parts of your car’s body are made of steel, an iron alloy. The salt can cause the metal to corrode, or rust, faster than it would otherwise because when mixed with water, the salt creat ...
M.Sc. Chemistry - Periyar University
... postulate, Microscopic reversibility. Potential energy diagrams, transition states and intermediates, methods of determining mechanisms – identification of products and determination of the presence of an intermediate, isotopic labeling, isotope effects. UNIT – III Stereochemistry (15 Hours) Homotop ...
... postulate, Microscopic reversibility. Potential energy diagrams, transition states and intermediates, methods of determining mechanisms – identification of products and determination of the presence of an intermediate, isotopic labeling, isotope effects. UNIT – III Stereochemistry (15 Hours) Homotop ...
National German Competition
... q) Write down the equation of the reaction of compound 1 with lithium dimethylcuprate and water. Give the complete names of the alcohols. Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. ...
... q) Write down the equation of the reaction of compound 1 with lithium dimethylcuprate and water. Give the complete names of the alcohols. Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.