Document
... structures of life, 4th edn, Pearson, Boston, MA. Alberts, B, Johnson, A, Lewis, J, Raff, M, Roberts, K & Walter P 2008, Molecular biology of the cell, 5th edn, Garland Science, New York. Berg, JM, Tymoczko, JL & Stryer, L 2012, Biochemistry, 7th edn, W.H. ...
... structures of life, 4th edn, Pearson, Boston, MA. Alberts, B, Johnson, A, Lewis, J, Raff, M, Roberts, K & Walter P 2008, Molecular biology of the cell, 5th edn, Garland Science, New York. Berg, JM, Tymoczko, JL & Stryer, L 2012, Biochemistry, 7th edn, W.H. ...
Organic Chemistry
... J = 7.5 Hz, 3H), 7.74 (d, J = 7.8 Hz, 3H), 7.35 (dt, J = 22.8, 7.2 Hz, 6H). 2.3.2 3,8,13-Tribromo-10,15-dihydro-5Hdiindolo [3,2-a:3’,2’-c]carbazole (5): A solution of N-bromosuccinimide (NBS) (0.28 g, 1.55 mmol) in dimethylformamide (2 mL) was added dropwise to a mixture of 4 (0.17 g, 0.5 mmol) in a ...
... J = 7.5 Hz, 3H), 7.74 (d, J = 7.8 Hz, 3H), 7.35 (dt, J = 22.8, 7.2 Hz, 6H). 2.3.2 3,8,13-Tribromo-10,15-dihydro-5Hdiindolo [3,2-a:3’,2’-c]carbazole (5): A solution of N-bromosuccinimide (NBS) (0.28 g, 1.55 mmol) in dimethylformamide (2 mL) was added dropwise to a mixture of 4 (0.17 g, 0.5 mmol) in a ...
tests for FLAVONOIDS
... Primary and secondary alcohols and aldehydes react with chromic acid or Jone’s reagent and give a blue-green precipitate in this test. Aldehydes are oxidized to carboxylic acids, secondary alcohols are oxidized to ketones and primary alcohols are oxidized first to aldehydes and then to carboxylic ac ...
... Primary and secondary alcohols and aldehydes react with chromic acid or Jone’s reagent and give a blue-green precipitate in this test. Aldehydes are oxidized to carboxylic acids, secondary alcohols are oxidized to ketones and primary alcohols are oxidized first to aldehydes and then to carboxylic ac ...
Chapter 11 Intermolecular Forces
... • There are three parts to a compound name: Base: This tells how many carbons are in the longest continuous chain. Suffix: This tells what type of compound it is. Prefix: This tells what groups are attached to the chain. ...
... • There are three parts to a compound name: Base: This tells how many carbons are in the longest continuous chain. Suffix: This tells what type of compound it is. Prefix: This tells what groups are attached to the chain. ...
Organic Chem Class #2
... labeled with prefixes in table P. All end in –ane. 29. AKLENES: hydrocarbons with only one double C=C bond. Only contain carbon and hydrogen atoms. Chains of 1 to 10 labeled with prefixes in table P. All end in –ene. In college molecules can have multiple double bonding, but we have a single double ...
... labeled with prefixes in table P. All end in –ane. 29. AKLENES: hydrocarbons with only one double C=C bond. Only contain carbon and hydrogen atoms. Chains of 1 to 10 labeled with prefixes in table P. All end in –ene. In college molecules can have multiple double bonding, but we have a single double ...
Alkenes—The Products of Elimination
... General Features of Elimination • Removal of the elements HX is called dehydrohalogenation. • Dehydrohalogenation is an example of elimination. • The curved arrow formalism shown below illustrates how four bonds are broken or formed in the process. ...
... General Features of Elimination • Removal of the elements HX is called dehydrohalogenation. • Dehydrohalogenation is an example of elimination. • The curved arrow formalism shown below illustrates how four bonds are broken or formed in the process. ...
Skill Practice 1
... 2. For which substance, A or B, does the freezing point decrease as the pressure is increased? 3. One of the substances behaves more like most other substances. Which substance and what property allows you to tell? 4. Assuming that the temperature scales for both phase diagrams are the same, which c ...
... 2. For which substance, A or B, does the freezing point decrease as the pressure is increased? 3. One of the substances behaves more like most other substances. Which substance and what property allows you to tell? 4. Assuming that the temperature scales for both phase diagrams are the same, which c ...
Alcohols, haloalkanes and analysis - MCQ topic quiz
... This formative assessment resource has been produced as part of our free Chemistry teaching and learning support package. All the Chemistry teaching and learning resources, including delivery guides, topic exploration packs, lesson elements and more are available on the qualification webpages. If y ...
... This formative assessment resource has been produced as part of our free Chemistry teaching and learning support package. All the Chemistry teaching and learning resources, including delivery guides, topic exploration packs, lesson elements and more are available on the qualification webpages. If y ...
File
... (b) Write a balanced symbol equation for the reaction. (Include state symbols and assume the product salt to be soluble in water, i.e. it is aqueous). ...
... (b) Write a balanced symbol equation for the reaction. (Include state symbols and assume the product salt to be soluble in water, i.e. it is aqueous). ...
CHAPTER-7 EQUILIBRIUM Equilibrium state- When
... The acid-base pair thatdiffers only by one proton is called a conjugateacidbase pair. IfBrönsted acid is a strong acid then itsconjugate base is a weak base and viceversa. Ionic product of water.Kw = [H+][OH–] pH = -log [H+] ; here[H+] is molar concentration of hydrogen ion. pH + pOH =14 p ...
... The acid-base pair thatdiffers only by one proton is called a conjugateacidbase pair. IfBrönsted acid is a strong acid then itsconjugate base is a weak base and viceversa. Ionic product of water.Kw = [H+][OH–] pH = -log [H+] ; here[H+] is molar concentration of hydrogen ion. pH + pOH =14 p ...
prs-A3
... This of course will allow for the room that the dissolved salt will take up as part of the total volume ...
... This of course will allow for the room that the dissolved salt will take up as part of the total volume ...
Document
... D) The agents: The agents are the opposite of their names. In short, the "agent" is that species whose presence enables the activity for which it is named. ... HUH???? e.g.) The presence of the oxidizing agent allows oxidation to proceed, hence the oxidizing agent is the reduced species. 1) Oxidizi ...
... D) The agents: The agents are the opposite of their names. In short, the "agent" is that species whose presence enables the activity for which it is named. ... HUH???? e.g.) The presence of the oxidizing agent allows oxidation to proceed, hence the oxidizing agent is the reduced species. 1) Oxidizi ...
Document
... D) The agents: The agents are the opposite of their names. In short, the "agent" is that species whose presence enables the activity for which it is named. ... HUH???? e.g.) The presence of the oxidizing agent allows oxidation to proceed, hence the oxidizing agent is the reduced species. 1) Oxidizi ...
... D) The agents: The agents are the opposite of their names. In short, the "agent" is that species whose presence enables the activity for which it is named. ... HUH???? e.g.) The presence of the oxidizing agent allows oxidation to proceed, hence the oxidizing agent is the reduced species. 1) Oxidizi ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... First of all, I would like to acknowledge Professor Gregory Dudley, without whom I could not have achieved what I have today. From him, I learned not only new ideas and information, but also how to properly conduct research. The way he teaches is not by giving a simple answer, but by guiding us to t ...
... First of all, I would like to acknowledge Professor Gregory Dudley, without whom I could not have achieved what I have today. From him, I learned not only new ideas and information, but also how to properly conduct research. The way he teaches is not by giving a simple answer, but by guiding us to t ...
Polymerization - Cornell University
... David A. Katz, 2005: Polyvinyl Alcohol Slime (Questions 1-4 Analysis adapted from his work) University of Minnesota, Joseph Franek: Nylon Rope Trick ...
... David A. Katz, 2005: Polyvinyl Alcohol Slime (Questions 1-4 Analysis adapted from his work) University of Minnesota, Joseph Franek: Nylon Rope Trick ...
LESSON 23: Exploding Bags
... chemical reaction are called reactants. The new substance(s) that are produced as a result of the reaction are called products. In this experiment, students work with common household vinegar and baking soda, which forms a basic solution when dissolved in water. Adding baking soda to vinegar starts ...
... chemical reaction are called reactants. The new substance(s) that are produced as a result of the reaction are called products. In this experiment, students work with common household vinegar and baking soda, which forms a basic solution when dissolved in water. Adding baking soda to vinegar starts ...
Organic Chemistry - Functional Groups
... C-terminus is a carboxylic acid Principles of Chemistry II ...
... C-terminus is a carboxylic acid Principles of Chemistry II ...
File
... _______16. How many electrons are there on an Fe 3+ ion ? A) 23 B) 24 C) 25 D) 26 E) 29 _______17. How many moles are there in 3.40 grams of ammonia, NH 3 ? A) 57.8 B) 2.00 C) 5.00 D) 0.461 E) 0.200 _______18. 0.150 mole of propanone has a mass of 8.7 grams. What is the molar mass of propanone? (in ...
... _______16. How many electrons are there on an Fe 3+ ion ? A) 23 B) 24 C) 25 D) 26 E) 29 _______17. How many moles are there in 3.40 grams of ammonia, NH 3 ? A) 57.8 B) 2.00 C) 5.00 D) 0.461 E) 0.200 _______18. 0.150 mole of propanone has a mass of 8.7 grams. What is the molar mass of propanone? (in ...
4.1 Writing and Balancing Chemical Equations
... Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.) Answer: 2NH 4 NO 3 ⟶ 2N 2 + O 2 + 4H 2 O ...
... Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen, molecular oxygen, and water. (Hint: Balance oxygen last, since it is present in more than one molecule on the right side of the equation.) Answer: 2NH 4 NO 3 ⟶ 2N 2 + O 2 + 4H 2 O ...
Ch. 11 Notes with Answers
... max, in this case, for acyclic pieces) 4. Remember which direction is the “true” laboratory direction. 5. Be careful that you aren’t adding or substracting carbons by mistake H3CO ...
... max, in this case, for acyclic pieces) 4. Remember which direction is the “true” laboratory direction. 5. Be careful that you aren’t adding or substracting carbons by mistake H3CO ...
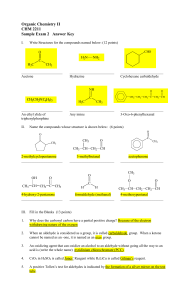
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... IV. Write a multi step reaction mechanism for the reaction of phenylmagnesium bromide ( MgBr) with acetone to form an alcohol. Indicate intermediates that may or may not be isolated. Use curved arrows to indicate electron attack and show charges and intermediates. (9 points) ...
... IV. Write a multi step reaction mechanism for the reaction of phenylmagnesium bromide ( MgBr) with acetone to form an alcohol. Indicate intermediates that may or may not be isolated. Use curved arrows to indicate electron attack and show charges and intermediates. (9 points) ...
Rapid Ether and Alcohol CO Bond Hydrogenolysis Catalyzed by
... first part of the catalytic cycle (5 → 8), the key reaction is the Lewis-acid-catalyzed ring-opening of ether 6 via transition state TS1 upon ether binding and activation of the etheric oxygen. This pathway is similar to the Ln(OTf)3-catalyzed tandem hydrogenolysis of ethers to alcohols.14 The comput ...
... first part of the catalytic cycle (5 → 8), the key reaction is the Lewis-acid-catalyzed ring-opening of ether 6 via transition state TS1 upon ether binding and activation of the etheric oxygen. This pathway is similar to the Ln(OTf)3-catalyzed tandem hydrogenolysis of ethers to alcohols.14 The comput ...
Functional Group Handout
... E. Amides: contain a carbonyl group. The carbon atom of the carbonyl group is bonded to another carbon atom and a nitrogen atom. Amides can be primary secondary or tertiary. The nitrogen atom of the primary amide is bonded to the carbonyl carbon and two hydrogens. The nitrogen atom of a secondary am ...
... E. Amides: contain a carbonyl group. The carbon atom of the carbonyl group is bonded to another carbon atom and a nitrogen atom. Amides can be primary secondary or tertiary. The nitrogen atom of the primary amide is bonded to the carbonyl carbon and two hydrogens. The nitrogen atom of a secondary am ...
Use the following answers for questions 10
... When the concentration of substance B in the reaction above is doubled, all other factors being held constant, it is found that the rate of the reaction remains unchanged. The most probable explanation for this observation is that (A) the order of the reaction with respect to substance B is 1 (B) s ...
... When the concentration of substance B in the reaction above is doubled, all other factors being held constant, it is found that the rate of the reaction remains unchanged. The most probable explanation for this observation is that (A) the order of the reaction with respect to substance B is 1 (B) s ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.