1 Mole
... CuSO4(aq) Atomic symbols describe the type of atoms in the compound (copper, sulfur, oxygen) subscript numbers describe the number of atoms in the compound (1 copper, 1 sulfur, 4 oxygen) subscript letters describe the physical state of the compound (aqueous) ...
... CuSO4(aq) Atomic symbols describe the type of atoms in the compound (copper, sulfur, oxygen) subscript numbers describe the number of atoms in the compound (1 copper, 1 sulfur, 4 oxygen) subscript letters describe the physical state of the compound (aqueous) ...
Amines: The Basic Group
... classes. Class depends on the number of carbons directly bonded to the nitrogen atom: a primary amine has one bond, a secondary two, etc. ...
... classes. Class depends on the number of carbons directly bonded to the nitrogen atom: a primary amine has one bond, a secondary two, etc. ...
Strumenti tutor LIM
... A chemical transformation takes place when....................(atoms in the reactants are rearranged to form new substabces)(old bonds are broken and new bonds are formed)( at least one new substance is formed) We can realize that a chemical reaction is taking place when...........( there is a chang ...
... A chemical transformation takes place when....................(atoms in the reactants are rearranged to form new substabces)(old bonds are broken and new bonds are formed)( at least one new substance is formed) We can realize that a chemical reaction is taking place when...........( there is a chang ...
UNIT 5 - H-W Science Website
... energy (in which case it will be released) or sometimes not enough energy (in which case it will be absorbed from the surroundings). The change in energy is due mainly to bond rearrangement during a reaction. Whether there is a net absorption or release of energy depends on the number of bonds broke ...
... energy (in which case it will be released) or sometimes not enough energy (in which case it will be absorbed from the surroundings). The change in energy is due mainly to bond rearrangement during a reaction. Whether there is a net absorption or release of energy depends on the number of bonds broke ...
Grant MacEwan College - Faculty Web Pages
... Note: Students are responsible for verifying the date of the final exam (see here). Course Description:(3 credits) This course studies the molecular structure and reactivity of organic compounds based on their functional groups and is intended for students who have obtained at least three credits in ...
... Note: Students are responsible for verifying the date of the final exam (see here). Course Description:(3 credits) This course studies the molecular structure and reactivity of organic compounds based on their functional groups and is intended for students who have obtained at least three credits in ...
1 - Academics
... 11. A covalent bond is best described as: a) The complete transfer of a pair of e- between two atoms; b) The complete transfer of a single e- between two atoms; c) The sharing of a single e- between two atoms; d) When an electron falls into the nucleus of another atom. e) The sharing of a pair of e- ...
... 11. A covalent bond is best described as: a) The complete transfer of a pair of e- between two atoms; b) The complete transfer of a single e- between two atoms; c) The sharing of a single e- between two atoms; d) When an electron falls into the nucleus of another atom. e) The sharing of a pair of e- ...
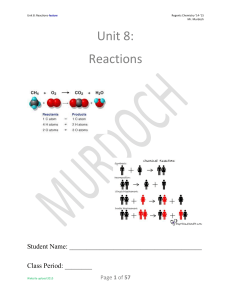
Unit 8: Reactions
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
One-Pot Synthesis of Organic-Sulfur
... at 60 °C gave a corresponding polymer in a 45% yield (run 1). Whereas the product produced via the previous polycondensation using the purified thiol was insoluble in any common solvents [11], the products in this one-pot method were soluble in THF, DMSO, and DMF. This improved solubility may be asc ...
... at 60 °C gave a corresponding polymer in a 45% yield (run 1). Whereas the product produced via the previous polycondensation using the purified thiol was insoluble in any common solvents [11], the products in this one-pot method were soluble in THF, DMSO, and DMF. This improved solubility may be asc ...
aq - Moodle@FCT
... A 0.5662-g sample of an ionic compound containing chloride ions and an unknown metal is dissolved in water and treated with an excess of AgNO3. If 1.0882 g of AgCl precipitate forms, what is the percent by mass of Cl in the original compound? ...
... A 0.5662-g sample of an ionic compound containing chloride ions and an unknown metal is dissolved in water and treated with an excess of AgNO3. If 1.0882 g of AgCl precipitate forms, what is the percent by mass of Cl in the original compound? ...
Chapter_4_Reactions_in_Aqueous_Solution
... A 0.5662-g sample of an ionic compound containing chloride ions and an unknown metal is dissolved in water and treated with an excess of AgNO3. If 1.0882 g of AgCl precipitate forms, what is the percent by mass of Cl in the original compound? ...
... A 0.5662-g sample of an ionic compound containing chloride ions and an unknown metal is dissolved in water and treated with an excess of AgNO3. If 1.0882 g of AgCl precipitate forms, what is the percent by mass of Cl in the original compound? ...
Exercises Topic 8 - OCW
... reduced with LiAlH4 to give a primary amine I (C2H7N). Deduce the structural formulas of compounds A to I. Solution: The reactions that give B, E and G products are typical nucleophilic substitutions. A must be an acid. There is only one acid with two carbon atoms: acetic acid. A acetic acid ...
... reduced with LiAlH4 to give a primary amine I (C2H7N). Deduce the structural formulas of compounds A to I. Solution: The reactions that give B, E and G products are typical nucleophilic substitutions. A must be an acid. There is only one acid with two carbon atoms: acetic acid. A acetic acid ...
3.2 Organic Synthesis (Reaction Pathways)
... The positive charge is shared between the 2 carbon atoms and the bromine atom. The Bromide ion will then attack from the other side. A TRANS arrangement. ...
... The positive charge is shared between the 2 carbon atoms and the bromine atom. The Bromide ion will then attack from the other side. A TRANS arrangement. ...
- Wiley Online Library
... reactions[1, 2] and cross-coupling reactions[3] and are useful precursors to heterocycles.[4] While a number of methods have been reported for the electrophilic halogenation of aldehydes or ketones containing only one enolizable position, the same reaction for unsymmetrical aliphatic ketones is chal ...
... reactions[1, 2] and cross-coupling reactions[3] and are useful precursors to heterocycles.[4] While a number of methods have been reported for the electrophilic halogenation of aldehydes or ketones containing only one enolizable position, the same reaction for unsymmetrical aliphatic ketones is chal ...
Titania-catalysed oxidative dehydrogenation of ethyl lactate
... Due to the importance of pyruvic acid and its derivatives, these reactions were studied by several groups, using both gas and liquid phases. In gas-phase reactions, various solid catalysts were used, including TeO2 and MoO3.5–9 These processes give high yields of pyruvate, but they are energy-intens ...
... Due to the importance of pyruvic acid and its derivatives, these reactions were studied by several groups, using both gas and liquid phases. In gas-phase reactions, various solid catalysts were used, including TeO2 and MoO3.5–9 These processes give high yields of pyruvate, but they are energy-intens ...
Topic 10
... The compound, 2-bromobutane, CH3CHBrCH2CH3, can react with sodium hydroxide to form compounds F, G and H. Compound F, C4H10O, exists as a pair of optical isomers. Compounds G and H, C4H8, are structural isomers, and compound H exists as a pair of geometrical isomers. (i) ...
... The compound, 2-bromobutane, CH3CHBrCH2CH3, can react with sodium hydroxide to form compounds F, G and H. Compound F, C4H10O, exists as a pair of optical isomers. Compounds G and H, C4H8, are structural isomers, and compound H exists as a pair of geometrical isomers. (i) ...
6.1.1 revision guide aromatic compounds
... If two or more substituents are present on the benzene ring, their positions must be indicated by the use of numbers. This should be done to give the lowest possible numbers to the substituents. When two or more different substituents are present, they are listed in alphabetical order and di, tri pr ...
... If two or more substituents are present on the benzene ring, their positions must be indicated by the use of numbers. This should be done to give the lowest possible numbers to the substituents. When two or more different substituents are present, they are listed in alphabetical order and di, tri pr ...
QUESTION BANK CHEMISTRY-XII THE SOLID STATE CHAPTER
... 17. What aspect of a reaction is influenced by presence of catalyst which increases the rate or possibility of the reaction? 18. State the role of activated complex in the reaction and state its relation with activation energy. 19. The rate constant for a first order reaction ...
... 17. What aspect of a reaction is influenced by presence of catalyst which increases the rate or possibility of the reaction? 18. State the role of activated complex in the reaction and state its relation with activation energy. 19. The rate constant for a first order reaction ...
22 Acyl Substn
... c) After nucleophile adds, though, loss of a leaving group can occur to re-form the carbonyl! This is how carboxylic acid derivatives differ from ketones and aldehydes. Overall mechanism is called addition–elimination. d) Under acidic conditions, protonations and deprotonations occur in between the ...
... c) After nucleophile adds, though, loss of a leaving group can occur to re-form the carbonyl! This is how carboxylic acid derivatives differ from ketones and aldehydes. Overall mechanism is called addition–elimination. d) Under acidic conditions, protonations and deprotonations occur in between the ...
Chapter 6
... The lone pair allows an amine inversion to occur, stereocenters on nitrogen thus become inverted ...
... The lone pair allows an amine inversion to occur, stereocenters on nitrogen thus become inverted ...
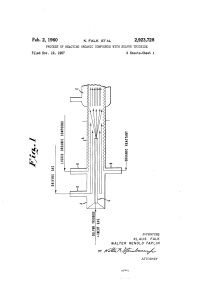
SULFUR TRIOXIDE +INERT GAS
... ing liquid sulfur trioxide to the vaporizer at a rate of solid at room temperature. In that event, the organic 0.66 lb. per hour and the air at the rate of 18.18 compound is ?rst melted by a preheater arrangement and ' (S.T.P.) cu. ft. per hour. The reaction mass passing into is fed into the reactor ...
... ing liquid sulfur trioxide to the vaporizer at a rate of solid at room temperature. In that event, the organic 0.66 lb. per hour and the air at the rate of 18.18 compound is ?rst melted by a preheater arrangement and ' (S.T.P.) cu. ft. per hour. The reaction mass passing into is fed into the reactor ...
document
... Reactions of Alkenes • The C=C double bond is very reactive since it is a centre of electron density. One of the bonds is weaker than the other and this breaks open on reaction leaving the basic carbon chain intact. • Thus alkenes undergo addition reactions and are attacked by electrophiles i.e? • ...
... Reactions of Alkenes • The C=C double bond is very reactive since it is a centre of electron density. One of the bonds is weaker than the other and this breaks open on reaction leaving the basic carbon chain intact. • Thus alkenes undergo addition reactions and are attacked by electrophiles i.e? • ...
Sample Final Questions Key/FS12
... actually have particles coming off of them – the advent of radioactivity. h. Pierre and Marie Curie (Nuclear) were Nobel laureates who discovered elements Polonium and Radium which were more radioactive than uranium. i. Ernst Rutherford (Nuclear) developed an experiment where they shot α-particles a ...
... actually have particles coming off of them – the advent of radioactivity. h. Pierre and Marie Curie (Nuclear) were Nobel laureates who discovered elements Polonium and Radium which were more radioactive than uranium. i. Ernst Rutherford (Nuclear) developed an experiment where they shot α-particles a ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.