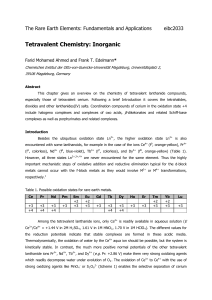
Chemistry - Andhra University
... Identification of alcohols by oxidation with KMnO4, ceric ammonium nitrate, lucas reagent and phenols by reaction with FeCl3. Polyhydroxy compounds: Pinacol-Pinacolone rearrangement. 3. Carbonyl compounds 10 h Nomenclature of aliphatic and aromatic carbonyl compounds, structure of the carbonyl group ...
... Identification of alcohols by oxidation with KMnO4, ceric ammonium nitrate, lucas reagent and phenols by reaction with FeCl3. Polyhydroxy compounds: Pinacol-Pinacolone rearrangement. 3. Carbonyl compounds 10 h Nomenclature of aliphatic and aromatic carbonyl compounds, structure of the carbonyl group ...
ALDEHYDES AND KETONES I. NUCLEOPHILIC ADDITION TO …
... •The first step is carbonyl addition of an amine, and the second step is a dehydration (elimination) to yield the C=N double bond. •HA is the catalyst ...
... •The first step is carbonyl addition of an amine, and the second step is a dehydration (elimination) to yield the C=N double bond. •HA is the catalyst ...
Reactions of Alkenes
... alkanes is catalytic hydrogenation of alkenes. This leads to a new question. "Starting with anything, how can I prepare cyclohexene in a single step by a reaction I am sure will work?" ...
... alkanes is catalytic hydrogenation of alkenes. This leads to a new question. "Starting with anything, how can I prepare cyclohexene in a single step by a reaction I am sure will work?" ...
Class Notes
... 1. LiAlH4 is strong enough to react with esters, NaBH4 isn’t 2. Selective reduction: if both an ester and an aldehyde/ketone are present: • LiAlH4 reduces both • NaBH4 selectively reduces the aldehyde/ketone but leaves the ester untouched 3. LiAlH4 is strong enough to react with and be destroyed by ...
... 1. LiAlH4 is strong enough to react with esters, NaBH4 isn’t 2. Selective reduction: if both an ester and an aldehyde/ketone are present: • LiAlH4 reduces both • NaBH4 selectively reduces the aldehyde/ketone but leaves the ester untouched 3. LiAlH4 is strong enough to react with and be destroyed by ...
Chem Soc Rev
... seriously, the reactivity of the target products (e.g., olefins and oxygenates) is typically much higher than that of the methane molecule, and thus, these products may easily undergo consecutive conversion such as deep oxidation to CO and CO2 under the reaction conditions. The activation and select ...
... seriously, the reactivity of the target products (e.g., olefins and oxygenates) is typically much higher than that of the methane molecule, and thus, these products may easily undergo consecutive conversion such as deep oxidation to CO and CO2 under the reaction conditions. The activation and select ...
Anhydrides, Esters and Amides
... • We can also hydrolyze an ester using a hot aqueous base, such as aqueous NaOH. • This reaction is often called saponification, a reference to its use in the manufacture of soaps. • The carboxylic acid formed in the hydrolysis reacts with hydroxide ion to form a carboxylic acid anion. • Each mole o ...
... • We can also hydrolyze an ester using a hot aqueous base, such as aqueous NaOH. • This reaction is often called saponification, a reference to its use in the manufacture of soaps. • The carboxylic acid formed in the hydrolysis reacts with hydroxide ion to form a carboxylic acid anion. • Each mole o ...
f-Element Disiloxanediolates: Novel Si[minus]O
... make these compounds suitable candidates for carrying out derivative chemistry. The most striking structural feature of the species, however, is their similarity with certain lanthanide metallocenes such as [(C5Me5)2LnN(SiMe3)2] (cf. 2) or [(C5Me5)2LnCl(thf)] (cf. 3).[29] Such organolanthanide compl ...
... make these compounds suitable candidates for carrying out derivative chemistry. The most striking structural feature of the species, however, is their similarity with certain lanthanide metallocenes such as [(C5Me5)2LnN(SiMe3)2] (cf. 2) or [(C5Me5)2LnCl(thf)] (cf. 3).[29] Such organolanthanide compl ...
No Slide Title
... systems containing unpaired electrons[1]. The traditional application areas for EPR include studies of transition metal complexes, stable organic radicals, transient reaction intermediates, as well as solid state and surface defects. In many cases, the extreme sensitivity of EPR allows experimental ...
... systems containing unpaired electrons[1]. The traditional application areas for EPR include studies of transition metal complexes, stable organic radicals, transient reaction intermediates, as well as solid state and surface defects. In many cases, the extreme sensitivity of EPR allows experimental ...
Synthesis, characterization and visible light photocatalytic activity of
... Malachite green (MG) (bis[p-dimethylaminophenyl]phenyl- methylium oxalate) was obtained from Merck Chemicals, Mumbai and used without any further purification. Zinc nitrate hexahydrate(Zn(NO)3 .6H2 O), cyclohexane, n-butanol, N,N,N-cetyl trimethyl ammonium bromide (CTAB), acetone, ferric nitrate nan ...
... Malachite green (MG) (bis[p-dimethylaminophenyl]phenyl- methylium oxalate) was obtained from Merck Chemicals, Mumbai and used without any further purification. Zinc nitrate hexahydrate(Zn(NO)3 .6H2 O), cyclohexane, n-butanol, N,N,N-cetyl trimethyl ammonium bromide (CTAB), acetone, ferric nitrate nan ...
BSA + TMCS + TMSI - Sigma
... and SPB-5. Normal hydrocarbons (carbon-hydrogen analytes with single bonds) are separated by these phases. More polar phases, SPB-1701 and SP-2250, separate carbon-hydrogen analytes that also contain Br, Cl, F, N, O, P, or S atoms or groups. A highly polar cyanopropylphenylsiloxane phase, SP-2330, i ...
... and SPB-5. Normal hydrocarbons (carbon-hydrogen analytes with single bonds) are separated by these phases. More polar phases, SPB-1701 and SP-2250, separate carbon-hydrogen analytes that also contain Br, Cl, F, N, O, P, or S atoms or groups. A highly polar cyanopropylphenylsiloxane phase, SP-2330, i ...
Inorganic Chemistry of LanthanideIV:
... Most of the practical uses of cerium dioxide in organic synthesis and catalysis are based on its oxidizing properties. For example, its use in the walls of so-called self-cleaning ovens make use of the fact that it assists oxidation of sticky hydrocabon deposits during the high-temperature cleaning ...
... Most of the practical uses of cerium dioxide in organic synthesis and catalysis are based on its oxidizing properties. For example, its use in the walls of so-called self-cleaning ovens make use of the fact that it assists oxidation of sticky hydrocabon deposits during the high-temperature cleaning ...
Picosecond X-ray Absorption Spectroscopy of a
... K-edge of [Fe(tren(py)3)](PF6)2 dissolved in acetonitrile solution. Static XAS measurements on the low-spin parent compound and its high-spin analogue, [Fe(tren(6-Me-py)3)](PF6)2, reveal distinct spectroscopic signatures for the two spin states in the X-ray absorption near-edge structure (XANES) and ...
... K-edge of [Fe(tren(py)3)](PF6)2 dissolved in acetonitrile solution. Static XAS measurements on the low-spin parent compound and its high-spin analogue, [Fe(tren(6-Me-py)3)](PF6)2, reveal distinct spectroscopic signatures for the two spin states in the X-ray absorption near-edge structure (XANES) and ...
research 1..13
... decrease can directly be registered in the infrared spectrum and gives the TEP its importance as an indirect descriptor for the metal−ligand (ML) bond strength. Even in the 1960s, several authors pointed out the relationship between the ML bond strength and the value of the CO stretching frequency,3 ...
... decrease can directly be registered in the infrared spectrum and gives the TEP its importance as an indirect descriptor for the metal−ligand (ML) bond strength. Even in the 1960s, several authors pointed out the relationship between the ML bond strength and the value of the CO stretching frequency,3 ...
24 BRON Chemistry of Coordination Compounds PPTS
... Structural Isomers • Some isomers differ in what ligands are bonded to the metal and what is outside the coordination sphere; these are coordination-sphere isomers. • Three isomers of CrCl3(H2O)6 are – The violet [Cr(H2O)6]Cl3, – The green [Cr(H2O)5Cl]Cl2 ∙ H2O, and – The (also) green [Cr(H2O)4Cl2] ...
... Structural Isomers • Some isomers differ in what ligands are bonded to the metal and what is outside the coordination sphere; these are coordination-sphere isomers. • Three isomers of CrCl3(H2O)6 are – The violet [Cr(H2O)6]Cl3, – The green [Cr(H2O)5Cl]Cl2 ∙ H2O, and – The (also) green [Cr(H2O)4Cl2] ...
Density functional theory study of the oxoperoxo
... a structure similar to the only known solid state structure18 for this system. To try to assess which of the isomers G-2syn or G-2anti is more stable, we have calculated the energies (with the zero point corrections) of their equilibrium geometries. The results indicate G-2syn as being 0.1 kcal mol ...
... a structure similar to the only known solid state structure18 for this system. To try to assess which of the isomers G-2syn or G-2anti is more stable, we have calculated the energies (with the zero point corrections) of their equilibrium geometries. The results indicate G-2syn as being 0.1 kcal mol ...
Ester - Net Texts
... Ester names are derived from the parent alcohol and the parent acid, where the latter may be an organic or an inorganic acid. Esters derived from the simplest carboxylic acids are commonly named according to the more traditional, so-called "trivial names" e.g. as formate, acetate, propionate, and bu ...
... Ester names are derived from the parent alcohol and the parent acid, where the latter may be an organic or an inorganic acid. Esters derived from the simplest carboxylic acids are commonly named according to the more traditional, so-called "trivial names" e.g. as formate, acetate, propionate, and bu ...
Chapter 25 Organic and Biological Chemistry
... C—C and C—H -bonds. • Therefore, great nonpolar solvents. • General rule of organic chemistry; • reactivity comes from the functional groups, ie. the part of the molecule that is not a straight alkane. • different functional groups give rise to different kinds of activity. Organic and Biological Ch ...
... C—C and C—H -bonds. • Therefore, great nonpolar solvents. • General rule of organic chemistry; • reactivity comes from the functional groups, ie. the part of the molecule that is not a straight alkane. • different functional groups give rise to different kinds of activity. Organic and Biological Ch ...
BIOC201W1_Carbohydrate Chemistry_2014
... General chemical formula of carbohydrate is [C.H2O]n Carbohydrates are defined chemically as aldehyde (-CHO) or ketone (=C=O) derivatives of polyhyric (more than one – OH groups) alcohols such as – Glycraldehyde, Dihydroxy acetone, Glucose, Fructose etc. ...
... General chemical formula of carbohydrate is [C.H2O]n Carbohydrates are defined chemically as aldehyde (-CHO) or ketone (=C=O) derivatives of polyhyric (more than one – OH groups) alcohols such as – Glycraldehyde, Dihydroxy acetone, Glucose, Fructose etc. ...
amine cured-epoxy matrices
... Epoxy resins can be cured with a variety of compounds called curing agents which are also known as curatives, hardeners, or converters. Of the many classes/types of curing agents, amines are most widely utilized as curing agents in epoxy matrices for high performance composites. This produces a hete ...
... Epoxy resins can be cured with a variety of compounds called curing agents which are also known as curatives, hardeners, or converters. Of the many classes/types of curing agents, amines are most widely utilized as curing agents in epoxy matrices for high performance composites. This produces a hete ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.




![f-Element Disiloxanediolates: Novel Si[minus]O](http://s1.studyres.com/store/data/014304880_1-eb67bfb0c3fc05ae875f0048d30df664-300x300.png)