Slide 1
... Further studies allowed to establish that Rh analogues such as HRh(CO)(PPh3)3 are 103-104 times more active in olefin hydroformylation so that it can be performed at ambient pressure of H2 and CO and room temperature. This catalyst favors also greater linear to branched aldehydes ratio (up to 20 :1) ...
... Further studies allowed to establish that Rh analogues such as HRh(CO)(PPh3)3 are 103-104 times more active in olefin hydroformylation so that it can be performed at ambient pressure of H2 and CO and room temperature. This catalyst favors also greater linear to branched aldehydes ratio (up to 20 :1) ...
Slide 1 - Catalysis Eprints database
... In aqueous, homogeneous two-phase catalysis, the active catalyst for the reaction is (and remains) dissolved in water. The reactants and the products, which are ideally organic and relatively non-polar, can be separated off after the reaction is complete by simply separating the second phase from th ...
... In aqueous, homogeneous two-phase catalysis, the active catalyst for the reaction is (and remains) dissolved in water. The reactants and the products, which are ideally organic and relatively non-polar, can be separated off after the reaction is complete by simply separating the second phase from th ...
lect7
... The metal atom is "holding" the reactive species together. Step 3. Insertion of alkene into Co-H bond Hydride H atom adds to C atom of alkene H(RCH=CH2)Co(CO)3 + CO RCH2CH2 - Co(CO)4 (18 e) Step 4. Insertion of CO into Co-alkyl bond RCH2CH2Co(CO)4 + CO RCH2CH2 - C(O) - Co(CO)4 (18 e) ...
... The metal atom is "holding" the reactive species together. Step 3. Insertion of alkene into Co-H bond Hydride H atom adds to C atom of alkene H(RCH=CH2)Co(CO)3 + CO RCH2CH2 - Co(CO)4 (18 e) Step 4. Insertion of CO into Co-alkyl bond RCH2CH2Co(CO)4 + CO RCH2CH2 - C(O) - Co(CO)4 (18 e) ...
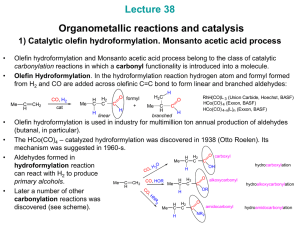
Organometallic Reactions and Catalysis
... more stable. – Coordinatively saturated complexes containing alkyl ligands are also more stable. ...
... more stable. – Coordinatively saturated complexes containing alkyl ligands are also more stable. ...
Hydroformylation Hydroformylation, also known as oxo synthesis or
... observed regioselectivity: Asymmetric hydroformylation Hydroformylation of internal alkenes creates new stereocenters. Using chiral phosphine ligands, the hydroformylation can be tailored to favor one enantiomer.[8] Other substrates ...
... observed regioselectivity: Asymmetric hydroformylation Hydroformylation of internal alkenes creates new stereocenters. Using chiral phosphine ligands, the hydroformylation can be tailored to favor one enantiomer.[8] Other substrates ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.




