organic chemistry
... Arabic numeric prefix) and the kind of substituent (by its name). The position of a substituent on the chain is indicated by the lowest number possible. The number precedes the name of the substituent. When there are two or more substituents of a given kind, use prefixes to indicate the number of ...
... Arabic numeric prefix) and the kind of substituent (by its name). The position of a substituent on the chain is indicated by the lowest number possible. The number precedes the name of the substituent. When there are two or more substituents of a given kind, use prefixes to indicate the number of ...
Document
... • In the body, ingested ethanol is oxidized in the liver first to CH3CHO (acetaldehyde), and then to CH3COO¯ (the acetate anion). • This oxidation is catalyzed by alcohol dehydrogenase. • If more ethanol is ingested than can be metabolized, the concentration of acetaldehyde increases. Acetaldehyde, ...
... • In the body, ingested ethanol is oxidized in the liver first to CH3CHO (acetaldehyde), and then to CH3COO¯ (the acetate anion). • This oxidation is catalyzed by alcohol dehydrogenase. • If more ethanol is ingested than can be metabolized, the concentration of acetaldehyde increases. Acetaldehyde, ...
Supplementary material - Royal Society of Chemistry
... The structure of 16 determined by X-ray crystallography shows that the epoxide forms exclusively on the opposite face of the ring to that of the N-diisopropylamino moiety. Epoxide opening in an antiperiplanar fashion would require attack by the bulky diethyl acetamidomalonate anion (19) on the same ...
... The structure of 16 determined by X-ray crystallography shows that the epoxide forms exclusively on the opposite face of the ring to that of the N-diisopropylamino moiety. Epoxide opening in an antiperiplanar fashion would require attack by the bulky diethyl acetamidomalonate anion (19) on the same ...
Introduction to Inorganic Chemistry
... possible, look at samples yourself. (If you see a sample of uranium, this will be of the “depleted” element, with its most radioactive isotopes removed.) Questions (1) Why are the metals listed in the last section so much sought after? (2) Are there any elements that you would reckon to be important ...
... possible, look at samples yourself. (If you see a sample of uranium, this will be of the “depleted” element, with its most radioactive isotopes removed.) Questions (1) Why are the metals listed in the last section so much sought after? (2) Are there any elements that you would reckon to be important ...
Synthesis and Characterization of Tetradentate Complexes Type
... within the ligand. This, in turn suggested that the chemistry of metal-bonded oximes should be rich. The inspection of data accumulated in the literature confirmed these assumptions (2).Transition metal complexes of vic- dioximes are of particular interest as biological model compounds. Numerous che ...
... within the ligand. This, in turn suggested that the chemistry of metal-bonded oximes should be rich. The inspection of data accumulated in the literature confirmed these assumptions (2).Transition metal complexes of vic- dioximes are of particular interest as biological model compounds. Numerous che ...
Chapter 2 Phenols
... 5- Reactions of Phenols A hydroxyl group is very powerful activating substituent, and electrophilic aromatic substitution in phenol occurs far faster, and under milder condition, than in benzene. ...
... 5- Reactions of Phenols A hydroxyl group is very powerful activating substituent, and electrophilic aromatic substitution in phenol occurs far faster, and under milder condition, than in benzene. ...
Chapter 16, Amines
... C6H5NH2, the simplest aromatic amine. ¾Name simple derivatives of aniline by using locator numbers starting with the amino group, or locate substituents by using the prefixes ortho (o), meta (m), and para (p). ¾Several derivatives of aniline have common names that are still widely used; among them i ...
... C6H5NH2, the simplest aromatic amine. ¾Name simple derivatives of aniline by using locator numbers starting with the amino group, or locate substituents by using the prefixes ortho (o), meta (m), and para (p). ¾Several derivatives of aniline have common names that are still widely used; among them i ...
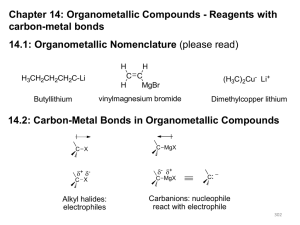
Organometallic Compounds - Reagents
... 14.13: Carbenes and Carbenoids Carbene: highly reactive intermediate, 6-electron species. The carbon is sp2 hybridized; it possesses a vacant hybridized p-orbital and an sp2 orbital with a non-bonding pair of electrons ...
... 14.13: Carbenes and Carbenoids Carbene: highly reactive intermediate, 6-electron species. The carbon is sp2 hybridized; it possesses a vacant hybridized p-orbital and an sp2 orbital with a non-bonding pair of electrons ...
CI 12.4 - Sackville School
... The carbocation has a C with a positive charge; that C had a half share of the two electrons in the bond which was broken. It now has no share of that pair and is now a C atom with one less electron so it has a single positive charge. ...
... The carbocation has a C with a positive charge; that C had a half share of the two electrons in the bond which was broken. It now has no share of that pair and is now a C atom with one less electron so it has a single positive charge. ...
Introduction to Inorganic Chemistry
... 2 The relative importance of different elements From an academic point of view, all elements might be said to be equally important. The chemistry of each has to be understood in detail if the chemistry of matter as a whole is to be understood in detail. From the point of view of the world at large, ...
... 2 The relative importance of different elements From an academic point of view, all elements might be said to be equally important. The chemistry of each has to be understood in detail if the chemistry of matter as a whole is to be understood in detail. From the point of view of the world at large, ...
NITRO COMPOUNDS
... It may be noted that nitration of arenes takes place at relatively low temperature where as nitration of alkanes requires a very high temperature . This may be due to the fact that the nitration of arenes involves the initial formation of electrophilic reagent while the nitration of alkanes is suppo ...
... It may be noted that nitration of arenes takes place at relatively low temperature where as nitration of alkanes requires a very high temperature . This may be due to the fact that the nitration of arenes involves the initial formation of electrophilic reagent while the nitration of alkanes is suppo ...
advanced chem
... 6. Before the radical name, place the # of the carbon it is attached to. 7. If 2 or more “like” groups are in the chain, use prefixes (di-,tri-, tetra-) before the radical name with the #’s of the carbons they are attached to ...
... 6. Before the radical name, place the # of the carbon it is attached to. 7. If 2 or more “like” groups are in the chain, use prefixes (di-,tri-, tetra-) before the radical name with the #’s of the carbons they are attached to ...
PDF document
... above all in carbon-carbon bond formation, mainly through metal catalysed coupling reactions. In addition, many iodinated aromatic derivatives are used in medicine as drugs or diagnostic aids, contrasting agents, and radioactively labelled markers. The chemistry dealing with selective iodination of ...
... above all in carbon-carbon bond formation, mainly through metal catalysed coupling reactions. In addition, many iodinated aromatic derivatives are used in medicine as drugs or diagnostic aids, contrasting agents, and radioactively labelled markers. The chemistry dealing with selective iodination of ...
Synthesis, identification and thermal decomposition of double
... atmosphere formed by dense volatile products liberated in the latter step. # 2000 Elsevier Science B.V. All rights reserved. Keywords: Double sul®tes; Chevreul's salt; Thermal behavior; TGA; DSC ...
... atmosphere formed by dense volatile products liberated in the latter step. # 2000 Elsevier Science B.V. All rights reserved. Keywords: Double sul®tes; Chevreul's salt; Thermal behavior; TGA; DSC ...
2(C8H4O4)2(H2O)4 to Anhydrous Ni2(OH)2(C8H4O4)
... The difference between these two bands agrees with the bridging character of the −COO− groups, with two O atoms coordinated to NiII. A comparison of the spectra allows us to verify that bands corresponding to stretching vibrations of the water molecules at 3424 and 3039 cm−1 are visible for 1 and 2 b ...
... The difference between these two bands agrees with the bridging character of the −COO− groups, with two O atoms coordinated to NiII. A comparison of the spectra allows us to verify that bands corresponding to stretching vibrations of the water molecules at 3424 and 3039 cm−1 are visible for 1 and 2 b ...
4 - WebAssign
... IUPAC (International Union of Pure and Applied Chemistry) rules for naming compounds in which the cation can have more than one charge: Name the metal, give its charge in Roman numerals, in parentheses, then name the anion. Give charge only when necessary. Copyright © Houghton Mifflin Company. All r ...
... IUPAC (International Union of Pure and Applied Chemistry) rules for naming compounds in which the cation can have more than one charge: Name the metal, give its charge in Roman numerals, in parentheses, then name the anion. Give charge only when necessary. Copyright © Houghton Mifflin Company. All r ...
Organometallic Compounds: Alkyllithium Reagent
... not possible to prepare a Grignard reagent from an organic group that contains an acidic hydrogen (any hydrogen more acidic than the hydrogen atoms of an alkane or alkene). ...
... not possible to prepare a Grignard reagent from an organic group that contains an acidic hydrogen (any hydrogen more acidic than the hydrogen atoms of an alkane or alkene). ...
12. Structure Determination: Mass Spectrometry and
... McMurry Organic Chemistry 6th edition Chapter 12 (c) 2003 ...
... McMurry Organic Chemistry 6th edition Chapter 12 (c) 2003 ...
12. Structure Determination: Mass Spectrometry and Infrared
... McMurry Organic Chemistry 6th edition Chapter 12 (c) 2003 ...
... McMurry Organic Chemistry 6th edition Chapter 12 (c) 2003 ...
Solving Spectroscopy Problems: Putting it All Together Once you`ve
... 1) Keep track of the number atoms in your molecule. Make sure the sum of the atoms in your 1H‐NMR implications + functional groups from IR spectrum matches the molecular formula obtained from the mass spectrum. 2) Keep track of the DBE count, especially when doing IR analysis. 3) Check that your ...
... 1) Keep track of the number atoms in your molecule. Make sure the sum of the atoms in your 1H‐NMR implications + functional groups from IR spectrum matches the molecular formula obtained from the mass spectrum. 2) Keep track of the DBE count, especially when doing IR analysis. 3) Check that your ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.