Le Châtelier`s Principle
... • Make more reactants because more collisions with the products will occur • A stress was applied and the system compensated for this change What will removing CO2 do? • Shift to make more products ...
... • Make more reactants because more collisions with the products will occur • A stress was applied and the system compensated for this change What will removing CO2 do? • Shift to make more products ...
Bagian 2 termodinamika
... and propels itself without the use of coal or oil in the following way. It pumps in warm sea water, extracts heat from that sea water, concentrates the extracted heat in its boilers, and discharges the cooled seawater back into the ocean. The discharged water may be ice if enough heat has been taken ...
... and propels itself without the use of coal or oil in the following way. It pumps in warm sea water, extracts heat from that sea water, concentrates the extracted heat in its boilers, and discharges the cooled seawater back into the ocean. The discharged water may be ice if enough heat has been taken ...
Energy: Conservation and Interconversion Demonstration:
... W = -P × A × ∆h (note the minus sign) A × ∆h = ∆V = Vfinal - Vinitial W = -P∆V = -P(Vfinal - Vinitial) Let’s do a couple of sample problems: Apply an external pressure of 1.0 atmospheres as a gas ...
... W = -P × A × ∆h (note the minus sign) A × ∆h = ∆V = Vfinal - Vinitial W = -P∆V = -P(Vfinal - Vinitial) Let’s do a couple of sample problems: Apply an external pressure of 1.0 atmospheres as a gas ...
Fluids and Thermodynamic Review BCE AAB DCD BDB CBE CEA
... (a) it increases (b) it remains constant (c) it decreases (d) it may increase or decrease depending on the shape of the rock 29. Salt water is denser than fresh water. A ship floats in both fresh water and salt water, the amount of water displaced by the salt water is (a) more (b) less (c) the same ...
... (a) it increases (b) it remains constant (c) it decreases (d) it may increase or decrease depending on the shape of the rock 29. Salt water is denser than fresh water. A ship floats in both fresh water and salt water, the amount of water displaced by the salt water is (a) more (b) less (c) the same ...
Why The SEB?
... Thickness: typically 1 km; varying from 20 m to several km; deeper with strong solar heating, strong winds, rough surface, or upward mean vertical motion in the free troposphere. Both structure and thickness have a strong diurnal cycle. Turbulent motions (opposite to laminar flow) , temperature, moi ...
... Thickness: typically 1 km; varying from 20 m to several km; deeper with strong solar heating, strong winds, rough surface, or upward mean vertical motion in the free troposphere. Both structure and thickness have a strong diurnal cycle. Turbulent motions (opposite to laminar flow) , temperature, moi ...
Chapter Summary
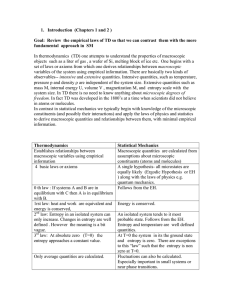
... A cycle is a sequence of processes that returns a system to its original state. The cycle as a whole satisfies the first law of thermodynamics, as does each of its processes. The change in internal energy for any cycle is always zero, because the system returns to its initial state, and the area of ...
... A cycle is a sequence of processes that returns a system to its original state. The cycle as a whole satisfies the first law of thermodynamics, as does each of its processes. The change in internal energy for any cycle is always zero, because the system returns to its initial state, and the area of ...
BCJ0205-15 Thermal phenomena (3-1-4)
... Week 8: Kinetic theory of gases: Maxwell’s velocity distribution. Week 9: Reversible and irreversible processes. Carnot cycle. Definition of entropy. Week 10: Thermal machines and the second law of thermodynamics. Week 11: Second law of thermodynamics and the statistical interpretation of entropy. W ...
... Week 8: Kinetic theory of gases: Maxwell’s velocity distribution. Week 9: Reversible and irreversible processes. Carnot cycle. Definition of entropy. Week 10: Thermal machines and the second law of thermodynamics. Week 11: Second law of thermodynamics and the statistical interpretation of entropy. W ...
15.3 The First Law of Thermodynamics
... In part a of figure, the system gains 1500J of heat and 2200J of work is done by the system on its surroundings. In part b, the system also gains 1500J of heat, but 2200J of work is done on the system. In each case, determine the change in internal energy of the system. ...
... In part a of figure, the system gains 1500J of heat and 2200J of work is done by the system on its surroundings. In part b, the system also gains 1500J of heat, but 2200J of work is done on the system. In each case, determine the change in internal energy of the system. ...
Second review [Compatibility Mode]
... Review of Chapter 6 • Hess’s Law • For a chemical reaction that can be written as the sum of two or more steps, the enthalpy change is the sum of the changes for the individual steps • You do not need to draw enthalpy diagrams ...
... Review of Chapter 6 • Hess’s Law • For a chemical reaction that can be written as the sum of two or more steps, the enthalpy change is the sum of the changes for the individual steps • You do not need to draw enthalpy diagrams ...
Chapter 8 Thermochemistry: Chemical Energy
... Enthalpy of Fusion (DHfusion): The amount of heat necessary to melt a substance without changing its temperature Enthalpy of Vaporization (DHvap): The amount of heat required to vaporize a substance without changing its temperature Enthalpy of Sublimation (DHsubl): The amount of heat required to con ...
... Enthalpy of Fusion (DHfusion): The amount of heat necessary to melt a substance without changing its temperature Enthalpy of Vaporization (DHvap): The amount of heat required to vaporize a substance without changing its temperature Enthalpy of Sublimation (DHsubl): The amount of heat required to con ...
EF - hrsbstaff.ednet.ns.ca
... *Remember: Heat and temperature are not the same thing!* 3 Types of Systems In thermochemistry, a boundary can be visable (i.e. walls of a beaker), or invisible (i.e. the separation of warm and cold air along a weather front). There are 3 possible types of systems depending on whether matter or ener ...
... *Remember: Heat and temperature are not the same thing!* 3 Types of Systems In thermochemistry, a boundary can be visable (i.e. walls of a beaker), or invisible (i.e. the separation of warm and cold air along a weather front). There are 3 possible types of systems depending on whether matter or ener ...
Planetary Sciences
... exponential dropoff with depth, with “scale depth” equal to LT conduction takes time, so heating/cooling not immediate surface is insulator at night because conductivity depends on … temperature seasonal effects can also be significant observations are made at radio wavelengths ...
... exponential dropoff with depth, with “scale depth” equal to LT conduction takes time, so heating/cooling not immediate surface is insulator at night because conductivity depends on … temperature seasonal effects can also be significant observations are made at radio wavelengths ...
Heat transfer

Heat transfer is the exchange of thermal energy between physical systems, depending on the temperature and pressure, by dissipating heat. The fundamental modes of heat transfer are conduction or diffusion, convection and radiation.Heat transfer always occurs from a region of high temperature to another region of lower temperature. Heat transfer changes the internal energy of both systems involved according to the First Law of Thermodynamics. The Second Law of Thermodynamics defines the concept of thermodynamic entropy, by measurable heat transfer.Thermal equilibrium is reached when all involved bodies and the surroundings reach the same temperature. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature.
















![Second review [Compatibility Mode]](http://s1.studyres.com/store/data/003692853_1-a578e4717b0c8365c11d7e7f576654ae-300x300.png)