Unit 8 Heat Study Guide A change of state is a ___ Process by
... c. Physical change that converts a substance from one physical form to another 2. Particles in a __________ move slower than particles in a __________. 3. Particles in a ___ vibrate in place. a. Solid b. Liquid c. Gas 4. Your bare feet cool on a cement floor due to ____ a. Convection b. Radiation c. ...
... c. Physical change that converts a substance from one physical form to another 2. Particles in a __________ move slower than particles in a __________. 3. Particles in a ___ vibrate in place. a. Solid b. Liquid c. Gas 4. Your bare feet cool on a cement floor due to ____ a. Convection b. Radiation c. ...
Topic 2 The first law of thermodynamics
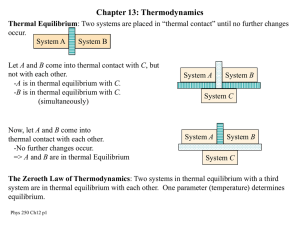
... If A and C are each in thermal equilibrium with B, A is also in equilibrium with C. Temperature as a quality of heat, by Galileo and Newton The temperatures are equal for all systems in thermal equilibrium. Temperature scale Thermometers ...
... If A and C are each in thermal equilibrium with B, A is also in equilibrium with C. Temperature as a quality of heat, by Galileo and Newton The temperatures are equal for all systems in thermal equilibrium. Temperature scale Thermometers ...
U3MEA02 Basic Engineering Thermodynamics
... of inanimate material agency, to derive mechanical effect from any portion of matter by cooling it below the temperature of the coldest of the surrounding objects. • Clausius statement: Heat can never pass from a colder to a warmer body without some other change, connected therewith, occurring at th ...
... of inanimate material agency, to derive mechanical effect from any portion of matter by cooling it below the temperature of the coldest of the surrounding objects. • Clausius statement: Heat can never pass from a colder to a warmer body without some other change, connected therewith, occurring at th ...
1 CHAPTER 1 INTRODUCTORY REMARKS 1.1 Introduction
... mathematics, but just a brief summary of the main properties of partial derivatives that you are likely to need. Thus I shan’t get down to serious thermodynamics until Chapter ...
... mathematics, but just a brief summary of the main properties of partial derivatives that you are likely to need. Thus I shan’t get down to serious thermodynamics until Chapter ...
Chapter 14
... Internal Energy, U, is the energy associated with the microscopic components of the system • Includes kinetic and potential energy associated with the random translational, rotational and vibrational motion of the atoms or molecules • Also includes any potential energy bonding the particles together ...
... Internal Energy, U, is the energy associated with the microscopic components of the system • Includes kinetic and potential energy associated with the random translational, rotational and vibrational motion of the atoms or molecules • Also includes any potential energy bonding the particles together ...
Chapter 1 Thermodynamics
... computer may work; for N ≥ 1googol = 10100 statistical physics may be the only tool. There are two standard ways to study the large N limit: • phenomenological (e.g. thermodynamics) and • fundamental (e.g. statistical mechanics). ...
... computer may work; for N ≥ 1googol = 10100 statistical physics may be the only tool. There are two standard ways to study the large N limit: • phenomenological (e.g. thermodynamics) and • fundamental (e.g. statistical mechanics). ...
Chem 521 Chemical Thermodynamics–—Syllabus, Fall 2015
... Statistical Mechanics Partition Functions in Statistical Thermodynamics ...
... Statistical Mechanics Partition Functions in Statistical Thermodynamics ...
Chapter 15 Notes - Valdosta State University
... In this case we can find the work done by multiplying the average pressure by the change in volume. Another way to find the area would be to count the blocks in the shaded area and multiply that result by the work represented by one block. If we estimate 8.9 squares, we multiply 8.9 by 2 X 105 pasc ...
... In this case we can find the work done by multiplying the average pressure by the change in volume. Another way to find the area would be to count the blocks in the shaded area and multiply that result by the work represented by one block. If we estimate 8.9 squares, we multiply 8.9 by 2 X 105 pasc ...
T - Massey University
... 2. Adiabatic expansion of the gas. In the second step the gas continues to expand (without heat flowing to or from the system) and thereby cools from the temperature of the the hot reservoir down to the temperature of the cold reservoir. ...
... 2. Adiabatic expansion of the gas. In the second step the gas continues to expand (without heat flowing to or from the system) and thereby cools from the temperature of the the hot reservoir down to the temperature of the cold reservoir. ...
entropy - Helios
... DS>0 This is also the second law of thermodynamics Entropy always increases ...
... DS>0 This is also the second law of thermodynamics Entropy always increases ...
Mass Balance for Open System
... Define, discuss, apply and analyze internal energy, first law, energy balance-closed system, thermodynamic state and state function, equilibrium, the Phase Rule, reversible process, constant-V and constant-P processes, enthalpy and heat capacity. 3. Chapter 3: Volumetric properties of pure fluids 4. ...
... Define, discuss, apply and analyze internal energy, first law, energy balance-closed system, thermodynamic state and state function, equilibrium, the Phase Rule, reversible process, constant-V and constant-P processes, enthalpy and heat capacity. 3. Chapter 3: Volumetric properties of pure fluids 4. ...
CHAPTER I
... external force mediating this process. Work is basically defined as any other transfer of energy into or out of the system. The most important form of work in thermodynamics is compressive work, which is due to a change in volume against or due to an external force (or pressure) on a gas. ...
... external force mediating this process. Work is basically defined as any other transfer of energy into or out of the system. The most important form of work in thermodynamics is compressive work, which is due to a change in volume against or due to an external force (or pressure) on a gas. ...