Corporate Profile
... Want to relate variables of state to each other. Experiments have found that gases follow approximately the same equation of state over a wide range of conditions. Although the atmosphere is a mixture of gases, it behaves as though it is a single “ideal” gas: • made up of a large number of molecules ...
... Want to relate variables of state to each other. Experiments have found that gases follow approximately the same equation of state over a wide range of conditions. Although the atmosphere is a mixture of gases, it behaves as though it is a single “ideal” gas: • made up of a large number of molecules ...
Chapter 6 Thermal Energy
... transferred from the stove to the surrounding air by conduction, convection, and radiation. One disadvantage of this system is that heat transfer from the room in which the stove is located to other rooms in the building can be slow. ...
... transferred from the stove to the surrounding air by conduction, convection, and radiation. One disadvantage of this system is that heat transfer from the room in which the stove is located to other rooms in the building can be slow. ...
Slide 1
... Now we can say that the second law does not forbid certain processes; all microstates are equally likely. However, some of them have an extraordinarily low probability of occurring – a lake freezing on a hot summer day, broken crockery re-assembling itself; all the air in a room moving into a single ...
... Now we can say that the second law does not forbid certain processes; all microstates are equally likely. However, some of them have an extraordinarily low probability of occurring – a lake freezing on a hot summer day, broken crockery re-assembling itself; all the air in a room moving into a single ...
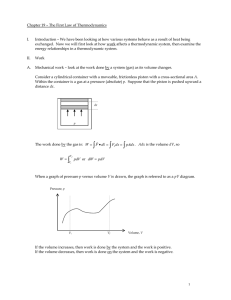
Chapter 1: The first law of thermodynamics
... and volume we say that the quantity is a function of state. Therefore, for an ideal gas in equilibrium, the system’s temperature is a function of state ( θ = F ( P,V ) ). A quantity, dG, is said to be an exact differential if it only depends on the difference in the function of state between two clo ...
... and volume we say that the quantity is a function of state. Therefore, for an ideal gas in equilibrium, the system’s temperature is a function of state ( θ = F ( P,V ) ). A quantity, dG, is said to be an exact differential if it only depends on the difference in the function of state between two clo ...
Chapter 14 The Ideal Gas Law and Kinetic Theory
... (pressure 1.00x105 Pa) from the blood in the capillaries. These sacs are called alveoli. The average radius of the alveoli is 0.125 mm, and the air inside contains 14% oxygen. Assuming that the air behaves as an ideal gas at 310K, find the number of oxygen molecules in one of these sacs. ...
... (pressure 1.00x105 Pa) from the blood in the capillaries. These sacs are called alveoli. The average radius of the alveoli is 0.125 mm, and the air inside contains 14% oxygen. Assuming that the air behaves as an ideal gas at 310K, find the number of oxygen molecules in one of these sacs. ...
Lecture 1
... Chapter 1. The properties of gases Gas - a form of matter that fills any container it occupies Physical state - the state of physical properties of a sample such as volume, mole, pressure, and temperature Mole - the number of atoms or molecules equal to Avogradro’s number NA = 6.022137 × 1023, which ...
... Chapter 1. The properties of gases Gas - a form of matter that fills any container it occupies Physical state - the state of physical properties of a sample such as volume, mole, pressure, and temperature Mole - the number of atoms or molecules equal to Avogradro’s number NA = 6.022137 × 1023, which ...
lecture notes-growth kinetics-3-heat evolution
... YX /S The higher degree of oxidation of the substrate has lower amounts of heat released: 1/YH ...
... YX /S The higher degree of oxidation of the substrate has lower amounts of heat released: 1/YH ...
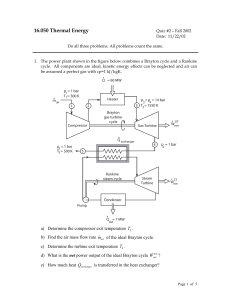
16.050 Thermal Energy
... b) Is the final internal energy Ufinal of the system (both compartments together) lower, equal or higher than the initial internal energy Uinitial of the system? Why? (A sentence or two is expected, perhaps bolstered by some equations.) c) What is the final temperature of the system Tfinal expressed ...
... b) Is the final internal energy Ufinal of the system (both compartments together) lower, equal or higher than the initial internal energy Uinitial of the system? Why? (A sentence or two is expected, perhaps bolstered by some equations.) c) What is the final temperature of the system Tfinal expressed ...
Thermo I
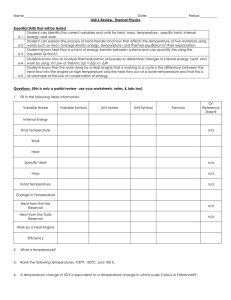
... The amount of heat Q needed for a certain temperature change ΔT is proportional to the temperature change and to the number of moles n of the substance ...
... The amount of heat Q needed for a certain temperature change ΔT is proportional to the temperature change and to the number of moles n of the substance ...
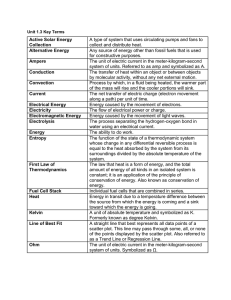
Energy & Power
... easily – the reverse is a different challenge • From the second law of thermodynamics, we know that : – it is not possible to change completely into work, with no other change taking place ...
... easily – the reverse is a different challenge • From the second law of thermodynamics, we know that : – it is not possible to change completely into work, with no other change taking place ...