12.1 Thermodynamic Systems, States, and Processes 12.3
... MC There is no heat flow into or out of the system in an (a) isothermal process, (b) adiabatic process, (c) isobaric process, (d) isometric process. MC According to the first law of thermodynamics, if work is done on a system, then (a) the internal energy of the system must change, (b) heat must be ...
... MC There is no heat flow into or out of the system in an (a) isothermal process, (b) adiabatic process, (c) isobaric process, (d) isometric process. MC According to the first law of thermodynamics, if work is done on a system, then (a) the internal energy of the system must change, (b) heat must be ...
ph202_overhead_ch15
... • A measure of the disorder (or randomness) of a system • For a reversible the change in entropy is measured as the ratio of heat gained to temperature DS = (Q/T)R = Sfinal - Sinitial – When heat energy is gained by a system, entropy is gained by the system (and lost by the surrounding environment) ...
... • A measure of the disorder (or randomness) of a system • For a reversible the change in entropy is measured as the ratio of heat gained to temperature DS = (Q/T)R = Sfinal - Sinitial – When heat energy is gained by a system, entropy is gained by the system (and lost by the surrounding environment) ...
Diapositiva 1
... precise definitions of the basic concepts. The Continuum Model Matter may be described at a molecular (or microscopic) level using the techniques of statistical mechanics and kinetic theory. For engineering purposes, however, we want ...
... precise definitions of the basic concepts. The Continuum Model Matter may be described at a molecular (or microscopic) level using the techniques of statistical mechanics and kinetic theory. For engineering purposes, however, we want ...
HormonesCascade
... Water Global Circulation during one Year because of evaporation and condensation equilibrium between liquid (oceans, rivers, lakes, clouds and sees) water and atmospheric gas water that has one called the vapors of water: n evaporatio ...
... Water Global Circulation during one Year because of evaporation and condensation equilibrium between liquid (oceans, rivers, lakes, clouds and sees) water and atmospheric gas water that has one called the vapors of water: n evaporatio ...
History of Thermodynamics
... development in their idiosyncratic histories. Some of the milestones of its development are given here: • first century AD: Hero of Alexandria documents many early thermal engines. • 1593: Galileo develops water thermometer. • 1650: Otto von Guericke designs and builds the first vacuum pump. • 1662: ...
... development in their idiosyncratic histories. Some of the milestones of its development are given here: • first century AD: Hero of Alexandria documents many early thermal engines. • 1593: Galileo develops water thermometer. • 1650: Otto von Guericke designs and builds the first vacuum pump. • 1662: ...
Golden Valley HS • AP Chemistry
... considered the surroundings. An open system can transfer energy and matter to and from the surroundings. A closed system is where energy can be transferred to the surroundings, but matter cannot. State functions depend only on the difference between the final and initial state of the system. The pat ...
... considered the surroundings. An open system can transfer energy and matter to and from the surroundings. A closed system is where energy can be transferred to the surroundings, but matter cannot. State functions depend only on the difference between the final and initial state of the system. The pat ...
Thermochemistry www.AssignmentPoint.com Thermochemistry is
... reactions and/or physical transformations. A reaction may release or absorb energy, and a phase change may do the same, such as in melting and boiling. Thermochemistry focuses on these energy changes, particularly on the system's energy exchange with its surroundings. Thermochemistry is useful in pr ...
... reactions and/or physical transformations. A reaction may release or absorb energy, and a phase change may do the same, such as in melting and boiling. Thermochemistry focuses on these energy changes, particularly on the system's energy exchange with its surroundings. Thermochemistry is useful in pr ...
Document
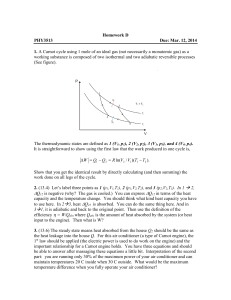
... efficiency η = W/Qabs where Qabs is the amount of heat absorbed by the system (or heat input to the engine). Then what is W? 3. (13.6) The steady state means heat absorbed from the house Q2 should be the same as the heat leakage into the house Q. For this air conditioner (a type of Carnot engine), t ...
... efficiency η = W/Qabs where Qabs is the amount of heat absorbed by the system (or heat input to the engine). Then what is W? 3. (13.6) The steady state means heat absorbed from the house Q2 should be the same as the heat leakage into the house Q. For this air conditioner (a type of Carnot engine), t ...
Applied Physics - Revision World
... - ΔQ = ΔW. If the gas expands to do work ΔW, & amount of heat ΔQ must be supplied - compression or expansion produces the same graph ...
... - ΔQ = ΔW. If the gas expands to do work ΔW, & amount of heat ΔQ must be supplied - compression or expansion produces the same graph ...
Dr.Eman Zakaria Hegazy Quantum Mechanics and Statistical
... therefore explaining thermodynamics as a natural result of statistics and mechanics (classical and quantum) at the microscopic level. - It provides a molecular-level interpretation of thermodynamic quantities such as work, heat, free energy, and entropy, allowing the thermodynamic properties of bulk ...
... therefore explaining thermodynamics as a natural result of statistics and mechanics (classical and quantum) at the microscopic level. - It provides a molecular-level interpretation of thermodynamic quantities such as work, heat, free energy, and entropy, allowing the thermodynamic properties of bulk ...