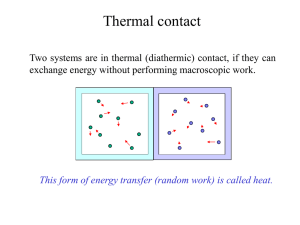
HeatTransfer
... • The kinetic molecular theory explains that all matter is made up of tiny particles. These atoms and molecules are constantly in motion. Kinetic energy is energy due to motion. • The particles of a substance move differently for different states. In solids, particles vibrate slightly but do n ...
... • The kinetic molecular theory explains that all matter is made up of tiny particles. These atoms and molecules are constantly in motion. Kinetic energy is energy due to motion. • The particles of a substance move differently for different states. In solids, particles vibrate slightly but do n ...
South Pasadena • AP Chemistry
... b) the enthalpy change of the reverse reaction is positive. c) the enthalpy of the products is greater than the enthalpy of the reactants. d) the energy released during bond formation is greater than the energy absorbed during bonding breaking for the reaction. 12. Given the two equations: 2 S(s) + ...
... b) the enthalpy change of the reverse reaction is positive. c) the enthalpy of the products is greater than the enthalpy of the reactants. d) the energy released during bond formation is greater than the energy absorbed during bonding breaking for the reaction. 12. Given the two equations: 2 S(s) + ...
States of matter - Tennessee State University
... of a system is W (statistical sum), the entropy S of the system in this state is S kB ln W where kB is a physical constant (Boltzmann’s constant). ...
... of a system is W (statistical sum), the entropy S of the system in this state is S kB ln W where kB is a physical constant (Boltzmann’s constant). ...
Daniel Bernoulli
... – Energy = mass x light speed squared – Light speed in vacuum is 3 x 108 m/s !! – Simple elegant equation relating matter and energy which led to the atomic bomb. ...
... – Energy = mass x light speed squared – Light speed in vacuum is 3 x 108 m/s !! – Simple elegant equation relating matter and energy which led to the atomic bomb. ...
Lecture 1
... You can slide a book across a table. It stops due to energy lost from friction. But, have you ever seen a book start sliding taking energy from the thermal energy of the table? You could start out with all of the air in this room in one half and vacuum in the other half, separated by a partition. Th ...
... You can slide a book across a table. It stops due to energy lost from friction. But, have you ever seen a book start sliding taking energy from the thermal energy of the table? You could start out with all of the air in this room in one half and vacuum in the other half, separated by a partition. Th ...
Introduction to Heat Transfer
... derivations. Key terms, such as enthalpy and free energies, as well as experimental variables, such as the coefficient of thermal expansion and compressibility, will be defined. Maxwell relations will then be used to incorporate the experimental variables into expressions for thermodynamic parameter ...
... derivations. Key terms, such as enthalpy and free energies, as well as experimental variables, such as the coefficient of thermal expansion and compressibility, will be defined. Maxwell relations will then be used to incorporate the experimental variables into expressions for thermodynamic parameter ...
Midterm Examination
... temperature can the gas be warmed before the tank ruptures? You can ignore the thermal expansion of the tank. (b) Based on your answer to part (a), is it reasonable to ignore the thermal expansion of the tank? Explain? 2/ (20 pts) Estimate the mean free path, the average time per collision, and the ...
... temperature can the gas be warmed before the tank ruptures? You can ignore the thermal expansion of the tank. (b) Based on your answer to part (a), is it reasonable to ignore the thermal expansion of the tank? Explain? 2/ (20 pts) Estimate the mean free path, the average time per collision, and the ...
Thermal Physics
... First Law of Thermodynamics DU = Q - W DU : change in internal energy of system (J) Q: heat added to the system (J). This heat exchange is driven by temperature difference. W: work done on the system (J). Work will be related to the change in the system’s volume. This law is sometimes par ...
... First Law of Thermodynamics DU = Q - W DU : change in internal energy of system (J) Q: heat added to the system (J). This heat exchange is driven by temperature difference. W: work done on the system (J). Work will be related to the change in the system’s volume. This law is sometimes par ...
v = Y
... reservoir when heat is discarded into it (TC). ◦ Any finite temperature drop would result in an irreversible processes. ◦ Every process that involves heat transfer must be isothermal. ◦ Any process in which the the working substance is between TH and TC, there must be no heat transfer into the hot o ...
... reservoir when heat is discarded into it (TC). ◦ Any finite temperature drop would result in an irreversible processes. ◦ Every process that involves heat transfer must be isothermal. ◦ Any process in which the the working substance is between TH and TC, there must be no heat transfer into the hot o ...
HEAT- Chapter 9
... Temperature and Thermal Equilibrium The temperature of an object is proportional to the average kinetic energy of particles in a substance The energy associated with atomic motion is internal energy(U). ...
... Temperature and Thermal Equilibrium The temperature of an object is proportional to the average kinetic energy of particles in a substance The energy associated with atomic motion is internal energy(U). ...
Lesson 6.5 – Hess`s Law
... Example #1: Calculate the heat of reaction for the combustion of nitrogen monoxide gas (NO) to form nitrogen dioxide gas (NO2), as given in the following thermochemical equation: ...
... Example #1: Calculate the heat of reaction for the combustion of nitrogen monoxide gas (NO) to form nitrogen dioxide gas (NO2), as given in the following thermochemical equation: ...
15 Thermodynamics
... Thermodynamics is the branch of physics that is built upon the fundamental laws that heat and work obey. ...
... Thermodynamics is the branch of physics that is built upon the fundamental laws that heat and work obey. ...
1. (a) Consider that an entropy S is as function of temperature T and
... from each other as well. Let the heat capacity of each gas be C1 and C2 . Box 1 starts with temperature Ti,1 , whereas box 2 starts with Ti,2 . (The subscript “i” means “initial,” and “f ” will mean “final.”) Assume that both gases are ideal so that the internal energy is given by Ei (T ) = Ci T . N ...
... from each other as well. Let the heat capacity of each gas be C1 and C2 . Box 1 starts with temperature Ti,1 , whereas box 2 starts with Ti,2 . (The subscript “i” means “initial,” and “f ” will mean “final.”) Assume that both gases are ideal so that the internal energy is given by Ei (T ) = Ci T . N ...
Work and Energy
... 3. the electrical contact is opened. The sudden opening of the contact produces a spark in the combustion chamber which ignites the fuel/air mixture. Rapid combustion of fuel releases heat, produces exhaust gases in the combustion chamber. (Power Stroke) ...
... 3. the electrical contact is opened. The sudden opening of the contact produces a spark in the combustion chamber which ignites the fuel/air mixture. Rapid combustion of fuel releases heat, produces exhaust gases in the combustion chamber. (Power Stroke) ...