Radioactivity - Williamstown Independent Schools
... • Mass defect is the difference between the mass of all the particles in an atom and the actual mass of the atom. • This “missing mass” comes about when the nucleus forms and a small amount of mass is converted to energy. ...
... • Mass defect is the difference between the mass of all the particles in an atom and the actual mass of the atom. • This “missing mass” comes about when the nucleus forms and a small amount of mass is converted to energy. ...
Endocrinology Regulation of Posterior pituitary hormones and
... feet and called myxedema – hypothyroidism in an adult. ...
... feet and called myxedema – hypothyroidism in an adult. ...
Name Period Nuclear Study Packet Set 1 1. What subatomic
... 1. What is the half-life of a 100.0 g sample of nitrogen-16 that decays to 12.5 g of nitrogen-16 in 21.6 s? 2. All isotopes of technetium are radioactive, but they have widely varying half-lives. If an 800.0 g sample of technetium-99 decays to 50.0 g of technetium-99 in 639 000 y, what is its ha ...
... 1. What is the half-life of a 100.0 g sample of nitrogen-16 that decays to 12.5 g of nitrogen-16 in 21.6 s? 2. All isotopes of technetium are radioactive, but they have widely varying half-lives. If an 800.0 g sample of technetium-99 decays to 50.0 g of technetium-99 in 639 000 y, what is its ha ...
Alpha Beta Fission Fusion
... The decay reaction and T½ of a substance are specific to the isotope of the element undergoing radioactive decay. For example, Bi210 can undergo decay to Tl206 with a T½ of five days. Bi215, by comparison, undergoes decay to Po215 with a T½ of 7.6 minutes, and Bi208 undergoes yet another mode of ...
... The decay reaction and T½ of a substance are specific to the isotope of the element undergoing radioactive decay. For example, Bi210 can undergo decay to Tl206 with a T½ of five days. Bi215, by comparison, undergoes decay to Po215 with a T½ of 7.6 minutes, and Bi208 undergoes yet another mode of ...
CH2ch19_1
... a) b-particle production: loss of an electron i. Fairly common for nuclides where Neutrons:Protons > 1.0 ii. Nucleus doesn’t contain electrons; loss of energy that becomes electron iii. Net effect: changes a neutron to a proton (Z increases by +1) ...
... a) b-particle production: loss of an electron i. Fairly common for nuclides where Neutrons:Protons > 1.0 ii. Nucleus doesn’t contain electrons; loss of energy that becomes electron iii. Net effect: changes a neutron to a proton (Z increases by +1) ...
Invertebrate hormones - Orange Coast College
... Perchlorate and thyroid function Perchlorate is used in rocket fuels, pyrotechnics, and other explosives It is also present in drinking water, and other foods Can inhibit iodide uptake and reduce thyroid function Can lead to metabolic disorders, but more commonly feelings of sluggishness and coldnes ...
... Perchlorate and thyroid function Perchlorate is used in rocket fuels, pyrotechnics, and other explosives It is also present in drinking water, and other foods Can inhibit iodide uptake and reduce thyroid function Can lead to metabolic disorders, but more commonly feelings of sluggishness and coldnes ...
AP Exam Questions: Nuclear
... (c) An alpha particle, or He nuclei, has a 2+ charge and would be attracted to the (-) side of the electric field. A beta particle, , or electron, has a single negative charge and is attracted to the positive side of the electric field, but since it is much lighter and faster than an alpha it wou ...
... (c) An alpha particle, or He nuclei, has a 2+ charge and would be attracted to the (-) side of the electric field. A beta particle, , or electron, has a single negative charge and is attracted to the positive side of the electric field, but since it is much lighter and faster than an alpha it wou ...
nuclear radiation
... combine at extremely high temperature, forming heavier, more stable nuclei and releasing energy. ...
... combine at extremely high temperature, forming heavier, more stable nuclei and releasing energy. ...
Hypothyroidism in Children
... The doctor or nurse will check thyroid function in several ways during clinic visits. Palpation – the doctor will feel the thyroid for any changes in size or if one side is bigger than the other. Reflexes – The doctor will tap the knee, ankle, and elbow with a rubber hammer to see if it twitches ...
... The doctor or nurse will check thyroid function in several ways during clinic visits. Palpation – the doctor will feel the thyroid for any changes in size or if one side is bigger than the other. Reflexes – The doctor will tap the knee, ankle, and elbow with a rubber hammer to see if it twitches ...
Barrett`s Esophagus Thyroid Disease
... If caught early, colorectal cancer is 90% curable. The American Cancer Society recommends that beginning at age 50, both men and women, should be tested and retested once every five to ten years.. ...
... If caught early, colorectal cancer is 90% curable. The American Cancer Society recommends that beginning at age 50, both men and women, should be tested and retested once every five to ten years.. ...
cps ch 10 notes
... • 1896 – French physicist Antoine Henri Becquerel • Experiments with uranium salts • Thought that the salts – which glow after exposure to sunlight – would produce X-rays while they glowed ...
... • 1896 – French physicist Antoine Henri Becquerel • Experiments with uranium salts • Thought that the salts – which glow after exposure to sunlight – would produce X-rays while they glowed ...
Histology Ch 21 755-762 [4-20
... destruction -low thyroid hormone causes excessive TSH release to cause hypertrophy of thyroid -adult hypothyroidism causes myxedema (sluggishness) Hyperthyroidism (toxic goiter or Grave’s disease) – excessive thyroid hormones are released, and people with Grave’s disease show autoantibodies, which b ...
... destruction -low thyroid hormone causes excessive TSH release to cause hypertrophy of thyroid -adult hypothyroidism causes myxedema (sluggishness) Hyperthyroidism (toxic goiter or Grave’s disease) – excessive thyroid hormones are released, and people with Grave’s disease show autoantibodies, which b ...
Independent Study: Nuclear Chemistry
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
Independent Study: Nuclear Chemistry
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
ThyroidGland
... • the hypothalamus secretes Thyroid releasing hormone (TRH), which stimulates the release of Thyroic stimulating hormone (TSH), • TSH travels in the bloodstream to the thyroid gland and initiates the release of thyroxine. • thyroxine increases metabolism by stimulating increased oxidation of glucose ...
... • the hypothalamus secretes Thyroid releasing hormone (TRH), which stimulates the release of Thyroic stimulating hormone (TSH), • TSH travels in the bloodstream to the thyroid gland and initiates the release of thyroxine. • thyroxine increases metabolism by stimulating increased oxidation of glucose ...
Nuclear Chemistry - Ector County ISD.
... Traditional chemical reactions occur as a result of the interaction between valence electrons around an atom's nucleus . In 1896, Henri Becquerel expanded the field of chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklo ...
... Traditional chemical reactions occur as a result of the interaction between valence electrons around an atom's nucleus . In 1896, Henri Becquerel expanded the field of chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklo ...
Half-life and Radioactive Decay guided notes
... Beta radiation is made up of a stream of beta particles. Beta particles are fast-moving released from a nucleus when a neutron ...
... Beta radiation is made up of a stream of beta particles. Beta particles are fast-moving released from a nucleus when a neutron ...
Beyond Element 83 are very unstable (radioactive)
... unstable (radioactive) • No amount neutrons can hold nucleus together once it has 83+ protons • All Elements 83 and above on PT are radioactive • Other elements may have radioactive isotopes applet ...
... unstable (radioactive) • No amount neutrons can hold nucleus together once it has 83+ protons • All Elements 83 and above on PT are radioactive • Other elements may have radioactive isotopes applet ...
What do I know about……
... nucleus understand that a nucleus of U-235 can be split (the process of fission) by collision with a neutron, and that this process releases energy in the form of kinetic energy of the fission products recall that the fission of U-235 produces two daughter nuclei and a small number of neutrons under ...
... nucleus understand that a nucleus of U-235 can be split (the process of fission) by collision with a neutron, and that this process releases energy in the form of kinetic energy of the fission products recall that the fission of U-235 produces two daughter nuclei and a small number of neutrons under ...
Endocrine Part 2 Powerpoint
... pituitary’s release of TSH • The thyroid releases thyroxine which stimulates cells to increase metabolism • Thyroxine’s key ingredient is iodine, which is required in small doses for life – Hypothyroidism (less thyroxine) causes exhaustion and weight gain – Lack of iodine can cause hypothyroidism an ...
... pituitary’s release of TSH • The thyroid releases thyroxine which stimulates cells to increase metabolism • Thyroxine’s key ingredient is iodine, which is required in small doses for life – Hypothyroidism (less thyroxine) causes exhaustion and weight gain – Lack of iodine can cause hypothyroidism an ...
disoders of thyroid gland
... TREATMENT. Sodium-L-thyroxine given orally is the treatment of choice. Because 80% of circulating T3 is formed by monodeiodination of T4, serum levels of T4 and T3 in treated infants return to normal. This is also true in the brain, where 80% of required T3 is produced locally from T4. In neonates, ...
... TREATMENT. Sodium-L-thyroxine given orally is the treatment of choice. Because 80% of circulating T3 is formed by monodeiodination of T4, serum levels of T4 and T3 in treated infants return to normal. This is also true in the brain, where 80% of required T3 is produced locally from T4. In neonates, ...
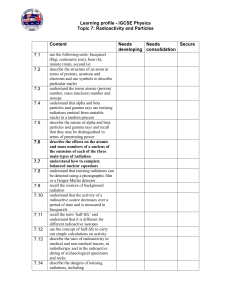
Radioactivity
... 1.I CAN Utilize appropriate scientific vocabulary to explain scientific concepts. 2.I CAN Distinguish between fission and fusion and where each process may be used. 3.I CAN Construct balanced nuclear equations (alpha, beta, gamma, neutron, etc.). 4.I CAN Complete a decay series for a given element. ...
... 1.I CAN Utilize appropriate scientific vocabulary to explain scientific concepts. 2.I CAN Distinguish between fission and fusion and where each process may be used. 3.I CAN Construct balanced nuclear equations (alpha, beta, gamma, neutron, etc.). 4.I CAN Complete a decay series for a given element. ...
Iodine-131
Iodine-131 (131I), also loosely and nonspecifically called radioiodine, is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuclear energy, medical diagnostic and treatment procedures, and natural gas production. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Fukushima nuclear crisis. This is because I-131 is a major uranium, plutonium fission product, comprising nearly 3% of the total products of fission (by weight). See fission product yield for a comparison with other radioactive fission products. I-131 is also a major fission product of uranium-233, produced from thorium.Due to its mode of beta decay, iodine-131 is notable for causing mutation and death in cells that it penetrates, and other cells up to several millimeters away. For this reason, high doses of the isotope are sometimes less dangerous than low doses, since they tend to kill thyroid tissues that would otherwise become cancerous as a result of the radiation. For example, children treated with moderate dose of I-131 for thyroid adenomas had a detectable increase in thyroid cancer, but children treated with a much higher dose did not. Likewise, most studies of very-high-dose I-131 for treatment of Graves disease have failed to find any increase in thyroid cancer, even though there is linear increase in thyroid cancer risk with I-131 absorption at moderate doses. Thus, iodine-131 is increasingly less employed in small doses in medical use (especially in children), but increasingly is used only in large and maximal treatment doses, as a way of killing targeted tissues. This is known as ""therapeutic use.""Iodine-131 can be ""seen"" by nuclear medicine imaging techniques (i.e., gamma cameras) whenever it is given for therapeutic use, since about 10% of its energy and radiation dose is via gamma radiation. However, since the other 90% of radiation (beta radiation) causes tissue damage without contributing to any ability to see or ""image"" the isotope, other less-damaging radioisotopes of iodine such as iodine-123 (see isotopes of iodine) are preferred in situations when only nuclear imaging is required. The isotope I-131 is still occasionally used for purely diagnostic (i.e., imaging) work, due to its low expense compared to other iodine radioisotopes. Very small medical imaging doses of I-131 have not shown any increase in thyroid cancer. The low-cost availability of I-131, in turn, is due to the relative ease of creating I-131 by neutron bombardment of natural tellurium in a nuclear reactor, then separating I-131 out by various simple methods (i.e., heating to drive off the volatile iodine). By contrast, other iodine radioisotopes are usually created by far more expensive techniques, starting with reactor radiation of expensive capsules of pressurized xenon gas.Iodine-131 is also one of the most commonly used gamma-emitting radioactive industrial tracer. Radioactive tracer isotopes are injected with hydraulic fracturing fluid to determine the injection profile and location of fractures created by hydraulic fracturing.Much smaller incidental doses of iodine-131 than those used in medical therapeutic procedures, are thought to be the major cause of increased thyroid cancers after accidental nuclear contamination. These cancers happen from residual tissue radiation damage caused by the I-131, and usually appear years after exposure, long after the I-131 has decayed.