Nuclear - chemmybear.com
... and write the balanced nuclear reaction for that less. decay process. (c) The neutron/proton ratio in Sr-90 and Cs-137 is (c) Gamma rays are observed during the radioactive too large and they emit beta particles (converting decay of carbon-11. Why is it unnecessary to inneutrons into protons) to low ...
... and write the balanced nuclear reaction for that less. decay process. (c) The neutron/proton ratio in Sr-90 and Cs-137 is (c) Gamma rays are observed during the radioactive too large and they emit beta particles (converting decay of carbon-11. Why is it unnecessary to inneutrons into protons) to low ...
True cat lovers take great pride and pleasure in growing old with
... them may be appropriate in certain situations. The first option is administration of an oral anti-thyroid medication that can control the effects of the over-active thyroid gland. Some cats do very well on this drug, while others may develop side effects such as vomiting, anorexia, fever and anemia. ...
... them may be appropriate in certain situations. The first option is administration of an oral anti-thyroid medication that can control the effects of the over-active thyroid gland. Some cats do very well on this drug, while others may develop side effects such as vomiting, anorexia, fever and anemia. ...
Thyroid Metabolic Hormones
... thyroid hormone receptor usually forms a heterodimer with retinoid X receptor (RXR) at specific thyroid hormone response elements on the DNA not all the proteins are increased by similar percentages-some only slightly, and others at least as much as sixfold thyroid hormones also appear to have nonge ...
... thyroid hormone receptor usually forms a heterodimer with retinoid X receptor (RXR) at specific thyroid hormone response elements on the DNA not all the proteins are increased by similar percentages-some only slightly, and others at least as much as sixfold thyroid hormones also appear to have nonge ...
endocrine2organs2013 30KB
... An excess of thyroxine (aa derivative with iodine) turns off TSH in a negative loop. Why? When thyroxine levels are low, thyroid cells are actually made to compensate. This can be due to Iodine deficiency, Iodine deficient goiter is a thyroid enlargement designed to enrich for Iodine when its level ...
... An excess of thyroxine (aa derivative with iodine) turns off TSH in a negative loop. Why? When thyroxine levels are low, thyroid cells are actually made to compensate. This can be due to Iodine deficiency, Iodine deficient goiter is a thyroid enlargement designed to enrich for Iodine when its level ...
Hormonal Regulation of Growth, Development, and Metabolism (9.2)
... secrets immature thyroid hormones into the spaces between the cells of the thyroid gland. For example Thyroxine (T4), once functional, will be released into the bloodstream, where it increases the rate at which the body metabolizes fats, proteins, and carbohydrates for energy. T4 stimulates the cell ...
... secrets immature thyroid hormones into the spaces between the cells of the thyroid gland. For example Thyroxine (T4), once functional, will be released into the bloodstream, where it increases the rate at which the body metabolizes fats, proteins, and carbohydrates for energy. T4 stimulates the cell ...
Frankie the dog `sniffs out thyroid cancer`
... study and it has high potential in areas of the world that may not have access to biopsy techniques. "There are many patients who are reluctant to undergo fine needle aspiration so I think that if you could design a technique where you have no invasive procedure that can have tremendous widespread a ...
... study and it has high potential in areas of the world that may not have access to biopsy techniques. "There are many patients who are reluctant to undergo fine needle aspiration so I think that if you could design a technique where you have no invasive procedure that can have tremendous widespread a ...
Isotopes and Radioactive Decay
... 4. Boron (B) has two naturally occurring isotopes: boron-10 (abundance = 19.8%; mass = 10.013 amu) and boron-11 (abundance = 80.2%; mass = 11.009 amu). Calculate the atomic mass of boron. ...
... 4. Boron (B) has two naturally occurring isotopes: boron-10 (abundance = 19.8%; mass = 10.013 amu) and boron-11 (abundance = 80.2%; mass = 11.009 amu). Calculate the atomic mass of boron. ...
Ch. 21.1 Nuclear Radiation
... 2. Radium-226 is a radioactive isotope that decays by releasing an alpha particle. Write a nuclear equation for the radioactive decay of radium-226. 3. Potassium-40 decays by releasing a beta particle. Write a nuclear equation that represents the radioactive decay of potassium-40. ...
... 2. Radium-226 is a radioactive isotope that decays by releasing an alpha particle. Write a nuclear equation for the radioactive decay of radium-226. 3. Potassium-40 decays by releasing a beta particle. Write a nuclear equation that represents the radioactive decay of potassium-40. ...
Chapter 9 Study Guide File
... 7. Diabetes insipidus is caused by a hyposecretion of __________________________. ...
... 7. Diabetes insipidus is caused by a hyposecretion of __________________________. ...
solutions - Physicsland
... 22. The Earth’s natural energy that heats the water in the hot spring is the energy of radioactive decay, which keeps the Earth’s interior molten. Radioactivity heats the water, but doesn’t make the water itself radioactive. The warmth of hot springs is one of the “nicer effects” of radioactive deca ...
... 22. The Earth’s natural energy that heats the water in the hot spring is the energy of radioactive decay, which keeps the Earth’s interior molten. Radioactivity heats the water, but doesn’t make the water itself radioactive. The warmth of hot springs is one of the “nicer effects” of radioactive deca ...
Homeostasis depends on mechanisms of regulation!
... Negative Feedback Negative Feedback occurs when a change in conditions in the environment triggers responses in your body that counteract that change. ...
... Negative Feedback Negative Feedback occurs when a change in conditions in the environment triggers responses in your body that counteract that change. ...
Chapter 10
... • Based on the fact that high-energy gamma rays cause damage to biological molecules. • Tumor cells are more susceptible than normal cells. • Example: cobalt-60 • Gamma radiation can cure cancer but can also cause cancer. ...
... • Based on the fact that high-energy gamma rays cause damage to biological molecules. • Tumor cells are more susceptible than normal cells. • Example: cobalt-60 • Gamma radiation can cure cancer but can also cause cancer. ...
NUCLEAR CHEMISTRY
... A. Nuclear Fission 1. A very heavy nucleus splits into more stable nuclei of intermediate mass 2. The mass of the products is less than the mass of the reactants. Missing mass is converted to energy a. Small amounts of missing mass are converted to HUGE amounts of energy (E = mc2) ...
... A. Nuclear Fission 1. A very heavy nucleus splits into more stable nuclei of intermediate mass 2. The mass of the products is less than the mass of the reactants. Missing mass is converted to energy a. Small amounts of missing mass are converted to HUGE amounts of energy (E = mc2) ...
The Endocrine System
... Hormones act by binding to specific chemical receptors on cell membranes or within cells. Cells that have receptors for a particular hormone are called target cells. If a cell does not have receptors for a particular hormone, the hormone has no effect on it. ...
... Hormones act by binding to specific chemical receptors on cell membranes or within cells. Cells that have receptors for a particular hormone are called target cells. If a cell does not have receptors for a particular hormone, the hormone has no effect on it. ...
Endocrine System
... Function: Secrete hormones into the circulatory system that will cause a change in the body in a different location Regulated by feedback mechanisms Stimulus-Response Hormones are only released until the appropriate changes are made (homeostasis is restored) ...
... Function: Secrete hormones into the circulatory system that will cause a change in the body in a different location Regulated by feedback mechanisms Stimulus-Response Hormones are only released until the appropriate changes are made (homeostasis is restored) ...
Endocrine Nuclear Medicine
... Subacute thyroidits (also known as de Quervains) • NM: Very good test as Iodine and pertechnetate are not taken up in acute phase (first 4 weeks after onset of symptoms) • Patient initially toxic • Reduced uptake persists 4-8 weeks • Tends to be normal by 12 weeks • Scan these within 10 days of ...
... Subacute thyroidits (also known as de Quervains) • NM: Very good test as Iodine and pertechnetate are not taken up in acute phase (first 4 weeks after onset of symptoms) • Patient initially toxic • Reduced uptake persists 4-8 weeks • Tends to be normal by 12 weeks • Scan these within 10 days of ...
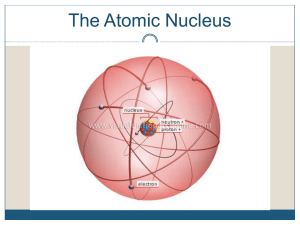
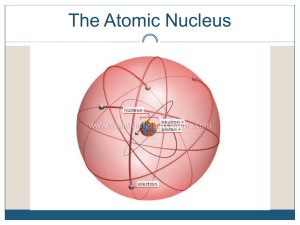
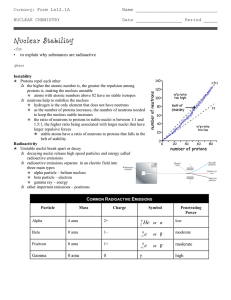
Nuclear Stability
... p hydrogen is the only element that does not have neutrons p as the number of protons increases, the number of neutrons needed to keep the nucleus stable increases p the ratio of neutrons to protons in stable nuclei is between 1:1 and 1.5:1, the higher ratio being associated with larger nuclei that ...
... p hydrogen is the only element that does not have neutrons p as the number of protons increases, the number of neutrons needed to keep the nucleus stable increases p the ratio of neutrons to protons in stable nuclei is between 1:1 and 1.5:1, the higher ratio being associated with larger nuclei that ...
Chapter 19 Nuclear Chemistry
... A first order reaction is 35% complete at the end of 55 minutes. What is the value of k? ...
... A first order reaction is 35% complete at the end of 55 minutes. What is the value of k? ...
Iodination of Tyrosine and Formation of the Thyroid Hormones
... • Interspersed in the interstitial spaces between the follicles is another secretory cell type, the C cells, which secrete the peptide hormone calcitonin. ...
... • Interspersed in the interstitial spaces between the follicles is another secretory cell type, the C cells, which secrete the peptide hormone calcitonin. ...
Radiation Questions March 4th
... Once a radioactive substance is dissolved in rainwater, it can enter the food chain. Following the Chernobyl explosion, some milk supplies were found to be radioactive. If one litre of milk contaminated with iodine-131 gives a count rate of 400 counts/second, how long will it take for the count rate ...
... Once a radioactive substance is dissolved in rainwater, it can enter the food chain. Following the Chernobyl explosion, some milk supplies were found to be radioactive. If one litre of milk contaminated with iodine-131 gives a count rate of 400 counts/second, how long will it take for the count rate ...
Grade 10S Physics T3W5 material
... How do we determine the half-life from the graph of activity versus time? 2. What are the factors that affect the rate of radioactive decay? 3. Give some examples of the uses of radioactivity A slightly radioactive sample of fertilizer is put in the soil. As the plant absorbs the fertilizer, the pla ...
... How do we determine the half-life from the graph of activity versus time? 2. What are the factors that affect the rate of radioactive decay? 3. Give some examples of the uses of radioactivity A slightly radioactive sample of fertilizer is put in the soil. As the plant absorbs the fertilizer, the pla ...
Isotope Half-Life Radiation Emitted
... a. Sterilize food (kill microorganisms) b. detect leaks in pipes c. Medical uses (imaging, detecting disease, tracing Blood circulation, treating cancer ...
... a. Sterilize food (kill microorganisms) b. detect leaks in pipes c. Medical uses (imaging, detecting disease, tracing Blood circulation, treating cancer ...
Introduction to Radiation and Radioactivity
... – The A/Z ratio is greater than 2 (or the N to P ratio is increasingly greater than 1) ...
... – The A/Z ratio is greater than 2 (or the N to P ratio is increasingly greater than 1) ...
Iodine-131
Iodine-131 (131I), also loosely and nonspecifically called radioiodine, is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuclear energy, medical diagnostic and treatment procedures, and natural gas production. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Fukushima nuclear crisis. This is because I-131 is a major uranium, plutonium fission product, comprising nearly 3% of the total products of fission (by weight). See fission product yield for a comparison with other radioactive fission products. I-131 is also a major fission product of uranium-233, produced from thorium.Due to its mode of beta decay, iodine-131 is notable for causing mutation and death in cells that it penetrates, and other cells up to several millimeters away. For this reason, high doses of the isotope are sometimes less dangerous than low doses, since they tend to kill thyroid tissues that would otherwise become cancerous as a result of the radiation. For example, children treated with moderate dose of I-131 for thyroid adenomas had a detectable increase in thyroid cancer, but children treated with a much higher dose did not. Likewise, most studies of very-high-dose I-131 for treatment of Graves disease have failed to find any increase in thyroid cancer, even though there is linear increase in thyroid cancer risk with I-131 absorption at moderate doses. Thus, iodine-131 is increasingly less employed in small doses in medical use (especially in children), but increasingly is used only in large and maximal treatment doses, as a way of killing targeted tissues. This is known as ""therapeutic use.""Iodine-131 can be ""seen"" by nuclear medicine imaging techniques (i.e., gamma cameras) whenever it is given for therapeutic use, since about 10% of its energy and radiation dose is via gamma radiation. However, since the other 90% of radiation (beta radiation) causes tissue damage without contributing to any ability to see or ""image"" the isotope, other less-damaging radioisotopes of iodine such as iodine-123 (see isotopes of iodine) are preferred in situations when only nuclear imaging is required. The isotope I-131 is still occasionally used for purely diagnostic (i.e., imaging) work, due to its low expense compared to other iodine radioisotopes. Very small medical imaging doses of I-131 have not shown any increase in thyroid cancer. The low-cost availability of I-131, in turn, is due to the relative ease of creating I-131 by neutron bombardment of natural tellurium in a nuclear reactor, then separating I-131 out by various simple methods (i.e., heating to drive off the volatile iodine). By contrast, other iodine radioisotopes are usually created by far more expensive techniques, starting with reactor radiation of expensive capsules of pressurized xenon gas.Iodine-131 is also one of the most commonly used gamma-emitting radioactive industrial tracer. Radioactive tracer isotopes are injected with hydraulic fracturing fluid to determine the injection profile and location of fractures created by hydraulic fracturing.Much smaller incidental doses of iodine-131 than those used in medical therapeutic procedures, are thought to be the major cause of increased thyroid cancers after accidental nuclear contamination. These cancers happen from residual tissue radiation damage caused by the I-131, and usually appear years after exposure, long after the I-131 has decayed.