Topic 6 – Benefits and drawbacks of using radioactive materials
... burial must be geologically stable (i.e have a very low risk of earthquakes) Advantages of nuclear power: Nuclear power stations themselves don’t directly produce carbon dioxide nuclear power doesn’t contribute to global warming o (However, energy is needed to make some components of nuclear reacto ...
... burial must be geologically stable (i.e have a very low risk of earthquakes) Advantages of nuclear power: Nuclear power stations themselves don’t directly produce carbon dioxide nuclear power doesn’t contribute to global warming o (However, energy is needed to make some components of nuclear reacto ...
Chapter 9
... In a balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers are equal for the nuclei of the reactants and the ...
... In a balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers are equal for the nuclei of the reactants and the ...
Chapter 16 Notes - Mr. Julien`s Homepage
... Read Section 16.5 in your eText! I will describe the use of radioisotopes in medicine. A. Scans with radioisotopes. 1. A scanner is used once the injected or ingested radioisotope has had time to migrate to the target organ or system. 2. The radiologist uses the scanner to detect the gamma rays emit ...
... Read Section 16.5 in your eText! I will describe the use of radioisotopes in medicine. A. Scans with radioisotopes. 1. A scanner is used once the injected or ingested radioisotope has had time to migrate to the target organ or system. 2. The radiologist uses the scanner to detect the gamma rays emit ...
Chapter 21 - Richsingiser.com
... through the basement, one of the most effective means of eliminating the radiation danger is to add ventilation fans in the basement. ...
... through the basement, one of the most effective means of eliminating the radiation danger is to add ventilation fans in the basement. ...
Anti-TSHR The thyroid gland produces hormones thyroxine (T4) and
... multinodular goiter and to monitor the effectiveness of anti-thyroid therapy. The test is also done during the third trimester of pregnancy to predict neonatal Graves disease, because some women may continue producing Anti-TSHR even after clinical cure. Anti-TSHR are IgG antibodies, so they can cros ...
... multinodular goiter and to monitor the effectiveness of anti-thyroid therapy. The test is also done during the third trimester of pregnancy to predict neonatal Graves disease, because some women may continue producing Anti-TSHR even after clinical cure. Anti-TSHR are IgG antibodies, so they can cros ...
NUCLEAR CHEMISTRY
... The movie The China Syndrome came out right before the accident occurred at the Three Mile Island nuclear reactor. ...
... The movie The China Syndrome came out right before the accident occurred at the Three Mile Island nuclear reactor. ...
Nuclear Power Plant Notes
... travel many meters in air and many centimeters in human tissue. It readily penetrates most materials and is sometimes called "penetrating radiation." ...
... travel many meters in air and many centimeters in human tissue. It readily penetrates most materials and is sometimes called "penetrating radiation." ...
Nuclear Power, Uranium Mining and Public Health
... industry workers has been occurring since the 1940s. More than one million workers have been employed in this industry since its beginning. However, studies have often investigated small groups of workers making it difficult to estimate precisely the risks associated with low levels of exposure. Ris ...
... industry workers has been occurring since the 1940s. More than one million workers have been employed in this industry since its beginning. However, studies have often investigated small groups of workers making it difficult to estimate precisely the risks associated with low levels of exposure. Ris ...
Principles of Matter and Energy
... By determining ratios of the different isotopes of carbon and other elements in samples of biological origin and in rocks, scientists are able to determine with certainty when these materials formed. ...
... By determining ratios of the different isotopes of carbon and other elements in samples of biological origin and in rocks, scientists are able to determine with certainty when these materials formed. ...
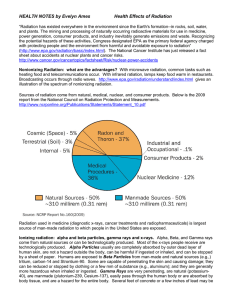
Health Effects of Radiation
... of skin. However, if alpha emitters have been inhaled, ingested (swallowed), or absorbed into the blood stream, sensitive living tissue can be exposed to alpha radiation. The resulting biological damage increases the risk of cancer; in particular, alpha radiation is known to cause lung cancer in hum ...
... of skin. However, if alpha emitters have been inhaled, ingested (swallowed), or absorbed into the blood stream, sensitive living tissue can be exposed to alpha radiation. The resulting biological damage increases the risk of cancer; in particular, alpha radiation is known to cause lung cancer in hum ...
NUCLEAR CHEMISTRY
... Most naturally occurring isotopes of elements up to atomic number 19 have stable nuclei. Elements with higher atomic number (20-83) consist of a mixture isotopes, some of which may have unstable nuclei. When the nucleus of an isotope is unstable, it is radioactive, which means that it will spontaneo ...
... Most naturally occurring isotopes of elements up to atomic number 19 have stable nuclei. Elements with higher atomic number (20-83) consist of a mixture isotopes, some of which may have unstable nuclei. When the nucleus of an isotope is unstable, it is radioactive, which means that it will spontaneo ...
I. Ch. 21.1 Nuclear Radiation
... The control of fission in a nuclear reactor involves two steps, neutron moderation and neutron absorption. a) Neutron Moderation is a process that ____________ ________________ neutrons so the reactor fuel (uranium-235 or plutonium-239) captures them to continue the chain reaction (water or graphite ...
... The control of fission in a nuclear reactor involves two steps, neutron moderation and neutron absorption. a) Neutron Moderation is a process that ____________ ________________ neutrons so the reactor fuel (uranium-235 or plutonium-239) captures them to continue the chain reaction (water or graphite ...
Honors Midterm - Stamford High School
... What is the charge on the helium nucleus above? +2 What is the charge on the whole atom of helium? neutral How much does a proton weigh? A neutron weigh? A electron weigh? ...
... What is the charge on the helium nucleus above? +2 What is the charge on the whole atom of helium? neutral How much does a proton weigh? A neutron weigh? A electron weigh? ...
Keeper is a fifteen year old, male neutered Patterdale Terrier. He
... Modern anaesthetics are generally very safe, although every anaesthetic carries an element of risk. Anaesthetics are usually required in older patients for surgical removal of tumours and dental work. Although the anaesthetic risk may be increased in older patients surgery is often essential and the ...
... Modern anaesthetics are generally very safe, although every anaesthetic carries an element of risk. Anaesthetics are usually required in older patients for surgical removal of tumours and dental work. Although the anaesthetic risk may be increased in older patients surgery is often essential and the ...
Hormones That Affect Metabolism
... – Growth hormone causes tissues to increase in number and/or size. In muscles, the cells increase in size by promoting protein synthesis and inhibiting protein breakdown. In bones, the cells elongate and the skeleton lengthens. – The growth hormone also promotes the breakdown of fats into fatty acid ...
... – Growth hormone causes tissues to increase in number and/or size. In muscles, the cells increase in size by promoting protein synthesis and inhibiting protein breakdown. In bones, the cells elongate and the skeleton lengthens. – The growth hormone also promotes the breakdown of fats into fatty acid ...
AP Study Guide Name__________________________ Chapter 45
... 4. Explain how a lack of iodine in the diet may result in goiter, an enlarged thyroid gland. ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ __________________ ...
... 4. Explain how a lack of iodine in the diet may result in goiter, an enlarged thyroid gland. ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ __________________ ...
2005 Nuclear FRQs - AP Chemistry Olympics
... and write the balanced nuclear reaction for that less. decay process. (c) The neutron/proton ratio in Sr-90 and Cs-137 is (c) Gamma rays are observed during the radioactive too large and they emit beta particles (converting decay of carbon-11. Why is it unnecessary to inneutrons into protons) to low ...
... and write the balanced nuclear reaction for that less. decay process. (c) The neutron/proton ratio in Sr-90 and Cs-137 is (c) Gamma rays are observed during the radioactive too large and they emit beta particles (converting decay of carbon-11. Why is it unnecessary to inneutrons into protons) to low ...
Rhenium isotopes in geochronology Stable isotope Relative atomic
... gamma rays (gamma radiation) – a stream of high-energy electromagnetic radiation given off by an atomic nucleus undergoing radioactive decay. The energies of gamma rays are higher than those of X-rays; thus, gamma rays have greater penetrating power. half-life (radioactive) – the time interval that ...
... gamma rays (gamma radiation) – a stream of high-energy electromagnetic radiation given off by an atomic nucleus undergoing radioactive decay. The energies of gamma rays are higher than those of X-rays; thus, gamma rays have greater penetrating power. half-life (radioactive) – the time interval that ...
Revision of Atomic Structure and Nuclide Notations Nuclide
... Since every radioisotope has a unique and constant half-life that fact can be used to find out how old an object containing that substance is. The radioactive element Carbon-14 is found in any object made of once living things. Carbon-14 is made in the upper atmosphere at a constant rate due to a ty ...
... Since every radioisotope has a unique and constant half-life that fact can be used to find out how old an object containing that substance is. The radioactive element Carbon-14 is found in any object made of once living things. Carbon-14 is made in the upper atmosphere at a constant rate due to a ty ...
2 α
... and create a nuclear reaction that produces neutrons. The neutrons bombard everything in the reactor, including the stable isotope, and the it absorbs the neutrons into its nucleus. Radioactive Iridium-192 is produced in a small-scale nuclear reactor by bombarding naturally occurring Iridium-191 wit ...
... and create a nuclear reaction that produces neutrons. The neutrons bombard everything in the reactor, including the stable isotope, and the it absorbs the neutrons into its nucleus. Radioactive Iridium-192 is produced in a small-scale nuclear reactor by bombarding naturally occurring Iridium-191 wit ...
By what process do most stars release energy? A. Electromagnetic
... Carbon-14 has a half-life of approximately 5,700 years. Analysis of the carbon in a piece of charred wood found in an excavation revealed that the carbon has 25 percent of the amount of carbon-14 that is found in the carbon of living trees. Which of the following is most nearly the age of the excava ...
... Carbon-14 has a half-life of approximately 5,700 years. Analysis of the carbon in a piece of charred wood found in an excavation revealed that the carbon has 25 percent of the amount of carbon-14 that is found in the carbon of living trees. Which of the following is most nearly the age of the excava ...
NUCLEAR CHEMISTRY
... Most naturally occurring isotopes of elements up to atomic number 19 have stable nuclei. Elements with higher atomic number (20-83) consist of a mixture isotopes, some of which may have unstable nuclei. When the nucleus of an isotope is unstable, it is radioactive, which means that it will spontaneo ...
... Most naturally occurring isotopes of elements up to atomic number 19 have stable nuclei. Elements with higher atomic number (20-83) consist of a mixture isotopes, some of which may have unstable nuclei. When the nucleus of an isotope is unstable, it is radioactive, which means that it will spontaneo ...
Chapter 3 Nuclear Radiation
... 9.3 RADIATION MEASUREMENT A Geiger counter • detects beta and gamma radiation. • uses ions produced by radiation to create an electrical current. ...
... 9.3 RADIATION MEASUREMENT A Geiger counter • detects beta and gamma radiation. • uses ions produced by radiation to create an electrical current. ...
File
... 3. An arsenic atom is more massive than a phosphorus atom is, and the elements are located in the same group. Which of the following statements about arsenic and phosphorus is true? A. Arsenic and phosphorus have the same number of valence electrons. B. The diameter of an arsenic atom is smaller tha ...
... 3. An arsenic atom is more massive than a phosphorus atom is, and the elements are located in the same group. Which of the following statements about arsenic and phosphorus is true? A. Arsenic and phosphorus have the same number of valence electrons. B. The diameter of an arsenic atom is smaller tha ...
Nuclear Fusion
... Half Life Calculations The half-life of beryllium-11 is 13.81 seconds. Let's say you start with 16 grams of 11Be. After 13.81s, you have 8 grams of that isotope left (the rest will have decayed to something else). After another 13.81s, you have 4 grams left; 13.81 seconds more, and you have 2 grams ...
... Half Life Calculations The half-life of beryllium-11 is 13.81 seconds. Let's say you start with 16 grams of 11Be. After 13.81s, you have 8 grams of that isotope left (the rest will have decayed to something else). After another 13.81s, you have 4 grams left; 13.81 seconds more, and you have 2 grams ...
Iodine-131
Iodine-131 (131I), also loosely and nonspecifically called radioiodine, is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuclear energy, medical diagnostic and treatment procedures, and natural gas production. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Fukushima nuclear crisis. This is because I-131 is a major uranium, plutonium fission product, comprising nearly 3% of the total products of fission (by weight). See fission product yield for a comparison with other radioactive fission products. I-131 is also a major fission product of uranium-233, produced from thorium.Due to its mode of beta decay, iodine-131 is notable for causing mutation and death in cells that it penetrates, and other cells up to several millimeters away. For this reason, high doses of the isotope are sometimes less dangerous than low doses, since they tend to kill thyroid tissues that would otherwise become cancerous as a result of the radiation. For example, children treated with moderate dose of I-131 for thyroid adenomas had a detectable increase in thyroid cancer, but children treated with a much higher dose did not. Likewise, most studies of very-high-dose I-131 for treatment of Graves disease have failed to find any increase in thyroid cancer, even though there is linear increase in thyroid cancer risk with I-131 absorption at moderate doses. Thus, iodine-131 is increasingly less employed in small doses in medical use (especially in children), but increasingly is used only in large and maximal treatment doses, as a way of killing targeted tissues. This is known as ""therapeutic use.""Iodine-131 can be ""seen"" by nuclear medicine imaging techniques (i.e., gamma cameras) whenever it is given for therapeutic use, since about 10% of its energy and radiation dose is via gamma radiation. However, since the other 90% of radiation (beta radiation) causes tissue damage without contributing to any ability to see or ""image"" the isotope, other less-damaging radioisotopes of iodine such as iodine-123 (see isotopes of iodine) are preferred in situations when only nuclear imaging is required. The isotope I-131 is still occasionally used for purely diagnostic (i.e., imaging) work, due to its low expense compared to other iodine radioisotopes. Very small medical imaging doses of I-131 have not shown any increase in thyroid cancer. The low-cost availability of I-131, in turn, is due to the relative ease of creating I-131 by neutron bombardment of natural tellurium in a nuclear reactor, then separating I-131 out by various simple methods (i.e., heating to drive off the volatile iodine). By contrast, other iodine radioisotopes are usually created by far more expensive techniques, starting with reactor radiation of expensive capsules of pressurized xenon gas.Iodine-131 is also one of the most commonly used gamma-emitting radioactive industrial tracer. Radioactive tracer isotopes are injected with hydraulic fracturing fluid to determine the injection profile and location of fractures created by hydraulic fracturing.Much smaller incidental doses of iodine-131 than those used in medical therapeutic procedures, are thought to be the major cause of increased thyroid cancers after accidental nuclear contamination. These cancers happen from residual tissue radiation damage caused by the I-131, and usually appear years after exposure, long after the I-131 has decayed.