Endocrine problems after treatment for cancer
... Thyroid deficiency or hypothyroidism Causes: Surgical removal of thyroid (thyroid cancer), radiation of the thyroid ...
... Thyroid deficiency or hypothyroidism Causes: Surgical removal of thyroid (thyroid cancer), radiation of the thyroid ...
Thyroid Temperture Test
... If the thyroid is overactive, your temperature will show 1-2 degrees above normal. If the thyroid is underactive your temperature will show 1-2 degrees below normal. The normal underarm temperature is between 36.6 and 36.8 degrees. If your temperature is consistently below this level, blood tests fo ...
... If the thyroid is overactive, your temperature will show 1-2 degrees above normal. If the thyroid is underactive your temperature will show 1-2 degrees below normal. The normal underarm temperature is between 36.6 and 36.8 degrees. If your temperature is consistently below this level, blood tests fo ...
Worksheet
... 8. Look at Figure 2 of the Friesema et al. paper. Why are PCR products on gels shown for patient 1 (P1) and patient 4 (P4), while DNA sequencing data are shown for patients 2, 3, and 5? ...
... 8. Look at Figure 2 of the Friesema et al. paper. Why are PCR products on gels shown for patient 1 (P1) and patient 4 (P4), while DNA sequencing data are shown for patients 2, 3, and 5? ...
Foldable - Georgetown ISD
... Nuclear Reactions (*produce far more energy compared to chemical): ...
... Nuclear Reactions (*produce far more energy compared to chemical): ...
Hypothyroidism is a deficiency in thyroid gland secretion of the
... Synthroid) once a day provides normal thyroid function and growth. The dose, depending upon the child’s age, ranges between 50 µg (0.05 mg) and 200 µg (0.2 mg) although some infants require slightly lower doses. Sometimes, a child may make T4 but can’t make T3 thus will need a medication that combin ...
... Synthroid) once a day provides normal thyroid function and growth. The dose, depending upon the child’s age, ranges between 50 µg (0.05 mg) and 200 µg (0.2 mg) although some infants require slightly lower doses. Sometimes, a child may make T4 but can’t make T3 thus will need a medication that combin ...
Chapter 19 Radioactive Material An Isotope is an element with a
... the radioactive isotope and then tracing that isotope in the person’s circulator system. Chemotherapy-‐ using radiation to kill cancerous cells in a person’s body Nuclear Energy: -‐Fission-‐ the process ...
... the radioactive isotope and then tracing that isotope in the person’s circulator system. Chemotherapy-‐ using radiation to kill cancerous cells in a person’s body Nuclear Energy: -‐Fission-‐ the process ...
2. Thyroid Gland T 4 and T 3
... disproportionate growth, short body with thick tongue and neck • treatment - reversed by iodine supplements or hormone replacement therapy ...
... disproportionate growth, short body with thick tongue and neck • treatment - reversed by iodine supplements or hormone replacement therapy ...
Chapter 1
... • Based on the fact that high-energy gamma rays cause damage to biological molecules • Tumor cells are more susceptible than normal cells • Example: cobalt-60 • Gamma radiation can cure cancer, but can also cause cancer ...
... • Based on the fact that high-energy gamma rays cause damage to biological molecules • Tumor cells are more susceptible than normal cells • Example: cobalt-60 • Gamma radiation can cure cancer, but can also cause cancer ...
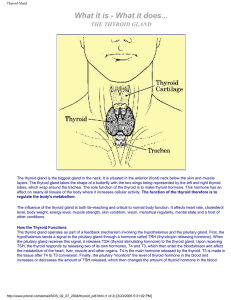
Thyroid Gland
... layers. The thyroid gland takes the shape of a butterfly with the two wings being represented by the left and right thyroid lobes, which wrap around the trachea. The sole function of the thyroid is to make thyroid hormone. This hormone has an effect on nearly all tissues of the body where it increas ...
... layers. The thyroid gland takes the shape of a butterfly with the two wings being represented by the left and right thyroid lobes, which wrap around the trachea. The sole function of the thyroid is to make thyroid hormone. This hormone has an effect on nearly all tissues of the body where it increas ...
Endocrine System Anatomy
... • Acromegaly = enlargement of the extremities, from excessive amounts of growth hormone. • Adrenalitis = inflammation of the adrenal gland. • Hypercalcemia = excessive calcium in the blood. • Hyperglycemia = excessive sugar in the blood. • Hyperkalemia = excessive potassium in blood. • Hyperthyroidi ...
... • Acromegaly = enlargement of the extremities, from excessive amounts of growth hormone. • Adrenalitis = inflammation of the adrenal gland. • Hypercalcemia = excessive calcium in the blood. • Hyperglycemia = excessive sugar in the blood. • Hyperkalemia = excessive potassium in blood. • Hyperthyroidi ...
Homeostatic Dysfunction
... causes Pernicious anaemia (stem cell divisions don’t occur) Vitamin K Produced by bacteria in large intestine. Lack causes reduced blood clotting ability. Homeostasis compromised if excessive blood is lost. Energy balance (intake and use) is a complex but essential element of homeostasis. ...
... causes Pernicious anaemia (stem cell divisions don’t occur) Vitamin K Produced by bacteria in large intestine. Lack causes reduced blood clotting ability. Homeostasis compromised if excessive blood is lost. Energy balance (intake and use) is a complex but essential element of homeostasis. ...
Is it Fat? Or could it be PHAT?
... Have you tried to lose weight? Have you increased your exercise? Reduced your calories? You've done all this and yet the weight stays on. You're tired and have only a portion of your former energy and stamina. It could be that you are at the mercy of an imbalance of PHAT. There is a complex and yet ...
... Have you tried to lose weight? Have you increased your exercise? Reduced your calories? You've done all this and yet the weight stays on. You're tired and have only a portion of your former energy and stamina. It could be that you are at the mercy of an imbalance of PHAT. There is a complex and yet ...
File
... •Aluminum foil will stop a beta particle •Can damage human cells if released inside the body ...
... •Aluminum foil will stop a beta particle •Can damage human cells if released inside the body ...
Review of Nuclear Chemistry
... we will breathe radon into our lungs, where it can cause harm. Radon is a uranium daughter, meaning that it is formed by the natural decay of U-238. On average, exposure from radiation from nuclear power plants and global nuclear fall out is small, when compared to natural sources. Radiation has bee ...
... we will breathe radon into our lungs, where it can cause harm. Radon is a uranium daughter, meaning that it is formed by the natural decay of U-238. On average, exposure from radiation from nuclear power plants and global nuclear fall out is small, when compared to natural sources. Radiation has bee ...
Radioactivity and Nuclear Reactions
... material required so that each fission reaction produces approximately one more fission reaction. • Nuclear power plants use control rods made of nonfissionable material that can absorb neutrons. • The control rods are moved in and out of the fissionable material to control the rate of the chain rea ...
... material required so that each fission reaction produces approximately one more fission reaction. • Nuclear power plants use control rods made of nonfissionable material that can absorb neutrons. • The control rods are moved in and out of the fissionable material to control the rate of the chain rea ...
Document
... When radiation (electromagnetic or particles) is absorbed by any material, energy is absorbed by the material (photon energy or kinetic energy). The physical unit of absorbed radiation dose is the gray (Gy), equal to 1 J per kg of mass. Dose rate is the rate of dosage, the dose per unit time interva ...
... When radiation (electromagnetic or particles) is absorbed by any material, energy is absorbed by the material (photon energy or kinetic energy). The physical unit of absorbed radiation dose is the gray (Gy), equal to 1 J per kg of mass. Dose rate is the rate of dosage, the dose per unit time interva ...
NUCLEAR CHEMISTRY
... 100 assemblies = reactor core Spent fuel rods are high-level nuclear waste (highly radioactive) All nuclear power plants have holding tanks for spent rods 12 meter deep and filled with water – cools rods and acts as a radiation shield Spend a decade or more in tank Limited time for plant operation d ...
... 100 assemblies = reactor core Spent fuel rods are high-level nuclear waste (highly radioactive) All nuclear power plants have holding tanks for spent rods 12 meter deep and filled with water – cools rods and acts as a radiation shield Spend a decade or more in tank Limited time for plant operation d ...
Santee Education Complex Chemistry Mini Assessment 11
... b. 0n1 + 13Al27 → 11Na24 + 2He4 c. 13Al27 + 2He4 → 15P30 +0n1 d. 7N14 + 2He4 →1H1 + 8O17 14) A process in which a very heavy nucleus splits into more stable nuclei of intermediate mass is called: a. nuclear fission. b. a chain reaction. c. nuclear fusion. d. radiocarbon dating. 15) An electron emitt ...
... b. 0n1 + 13Al27 → 11Na24 + 2He4 c. 13Al27 + 2He4 → 15P30 +0n1 d. 7N14 + 2He4 →1H1 + 8O17 14) A process in which a very heavy nucleus splits into more stable nuclei of intermediate mass is called: a. nuclear fission. b. a chain reaction. c. nuclear fusion. d. radiocarbon dating. 15) An electron emitt ...
Radioactivity Notes Day 1 and 2 Apr 23 and Apr 24
... o When these nuclei lose energy and break apart, decay occurs. ...
... o When these nuclei lose energy and break apart, decay occurs. ...
Discussion Topics
... Key concept: if the medication dosage is correct, there should be few, if any, adverse effects. The most common adverse effects result from cardiac stimulation and CNS stimulation. 2a. Students’ answers should include the following: Students should use Box 37.4 as a guide to answer this questi ...
... Key concept: if the medication dosage is correct, there should be few, if any, adverse effects. The most common adverse effects result from cardiac stimulation and CNS stimulation. 2a. Students’ answers should include the following: Students should use Box 37.4 as a guide to answer this questi ...
The discovery of the natural radioactive decay of uranium in 1896 by
... different atomic weights owing to variations in the number of neutrons. Atoms of the same element with differing atomic weights are called isotopes. Radioactive decay is a spontaneous process in which an isotope (the parent) loses particles from its nucleus to form an isotope of a new element (the d ...
... different atomic weights owing to variations in the number of neutrons. Atoms of the same element with differing atomic weights are called isotopes. Radioactive decay is a spontaneous process in which an isotope (the parent) loses particles from its nucleus to form an isotope of a new element (the d ...
Endocrine3
... Many common symptoms of hyperthyroidism are similar to those of adrenergic excess. ...
... Many common symptoms of hyperthyroidism are similar to those of adrenergic excess. ...
Chapter 9: Nuclear Changes
... affected cells and are used to locate tumors 3) _______________ - radioactive tracers in water - follow path and rate of absorption into ground and plants - locate with sensors 4) ___________ ___________ - Ex.- shooting beams of gamma rays at tumors - Ex.- kill defective bone marrow with nuclear rad ...
... affected cells and are used to locate tumors 3) _______________ - radioactive tracers in water - follow path and rate of absorption into ground and plants - locate with sensors 4) ___________ ___________ - Ex.- shooting beams of gamma rays at tumors - Ex.- kill defective bone marrow with nuclear rad ...
Iodine-131
Iodine-131 (131I), also loosely and nonspecifically called radioiodine, is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuclear energy, medical diagnostic and treatment procedures, and natural gas production. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Fukushima nuclear crisis. This is because I-131 is a major uranium, plutonium fission product, comprising nearly 3% of the total products of fission (by weight). See fission product yield for a comparison with other radioactive fission products. I-131 is also a major fission product of uranium-233, produced from thorium.Due to its mode of beta decay, iodine-131 is notable for causing mutation and death in cells that it penetrates, and other cells up to several millimeters away. For this reason, high doses of the isotope are sometimes less dangerous than low doses, since they tend to kill thyroid tissues that would otherwise become cancerous as a result of the radiation. For example, children treated with moderate dose of I-131 for thyroid adenomas had a detectable increase in thyroid cancer, but children treated with a much higher dose did not. Likewise, most studies of very-high-dose I-131 for treatment of Graves disease have failed to find any increase in thyroid cancer, even though there is linear increase in thyroid cancer risk with I-131 absorption at moderate doses. Thus, iodine-131 is increasingly less employed in small doses in medical use (especially in children), but increasingly is used only in large and maximal treatment doses, as a way of killing targeted tissues. This is known as ""therapeutic use.""Iodine-131 can be ""seen"" by nuclear medicine imaging techniques (i.e., gamma cameras) whenever it is given for therapeutic use, since about 10% of its energy and radiation dose is via gamma radiation. However, since the other 90% of radiation (beta radiation) causes tissue damage without contributing to any ability to see or ""image"" the isotope, other less-damaging radioisotopes of iodine such as iodine-123 (see isotopes of iodine) are preferred in situations when only nuclear imaging is required. The isotope I-131 is still occasionally used for purely diagnostic (i.e., imaging) work, due to its low expense compared to other iodine radioisotopes. Very small medical imaging doses of I-131 have not shown any increase in thyroid cancer. The low-cost availability of I-131, in turn, is due to the relative ease of creating I-131 by neutron bombardment of natural tellurium in a nuclear reactor, then separating I-131 out by various simple methods (i.e., heating to drive off the volatile iodine). By contrast, other iodine radioisotopes are usually created by far more expensive techniques, starting with reactor radiation of expensive capsules of pressurized xenon gas.Iodine-131 is also one of the most commonly used gamma-emitting radioactive industrial tracer. Radioactive tracer isotopes are injected with hydraulic fracturing fluid to determine the injection profile and location of fractures created by hydraulic fracturing.Much smaller incidental doses of iodine-131 than those used in medical therapeutic procedures, are thought to be the major cause of increased thyroid cancers after accidental nuclear contamination. These cancers happen from residual tissue radiation damage caused by the I-131, and usually appear years after exposure, long after the I-131 has decayed.