Nuclear Chemistry - Mona Shores Blogs
... from the atmosphere, but when they die they stop absorbing it and the levels do not change. • From this point levels start to decrease as the radioactive carbon decays. • The levels in the object are then compared with levels in the atmosphere. ...
... from the atmosphere, but when they die they stop absorbing it and the levels do not change. • From this point levels start to decrease as the radioactive carbon decays. • The levels in the object are then compared with levels in the atmosphere. ...
product information neo-mercazole
... months, and up to 2 years of treatment may be required. If thyroidectomy is intended, it can be carried out once the euthyroid state is achieved with NEO-MERCAZOLE, which is then discontinued. Changeover from thiouracils: When treatment with one of the thiouracils is replaced by NEO-MERCAZOLE therap ...
... months, and up to 2 years of treatment may be required. If thyroidectomy is intended, it can be carried out once the euthyroid state is achieved with NEO-MERCAZOLE, which is then discontinued. Changeover from thiouracils: When treatment with one of the thiouracils is replaced by NEO-MERCAZOLE therap ...
Nuclear Decay
... Fusion is a process where nuclei collide and join together to form a heavier atom, usually deuterium and tritium. When this happens a considerable amount of energy gets released at extremely high temperatures: nearly 150 million degrees Celsius. At extreme temperatures, electrons are separated from ...
... Fusion is a process where nuclei collide and join together to form a heavier atom, usually deuterium and tritium. When this happens a considerable amount of energy gets released at extremely high temperatures: nearly 150 million degrees Celsius. At extreme temperatures, electrons are separated from ...
CHM 123-Chapter 2.7
... What type of radiation occurs when carbon-11 decays to boron -11? How has the number of protons and neutrons in the nucleus changed? ...
... What type of radiation occurs when carbon-11 decays to boron -11? How has the number of protons and neutrons in the nucleus changed? ...
Ch.7 Summary Notes
... the half-life of the parent and the effective dating range the isotope can be used for. ...
... the half-life of the parent and the effective dating range the isotope can be used for. ...
Thyroid-Adrenal Fatigue Syndrome!
... If your battery is dead, then no matter how much you step on the gas pedal, you just won’t move. Conversely, if your gas pedal is stuck then you wont be able to get fuel to the engine which then needs to be ignited by a spark from the battery—simple combustion engine mechanics! As a car needs fuel, ...
... If your battery is dead, then no matter how much you step on the gas pedal, you just won’t move. Conversely, if your gas pedal is stuck then you wont be able to get fuel to the engine which then needs to be ignited by a spark from the battery—simple combustion engine mechanics! As a car needs fuel, ...
What do these food items have in common?
... technique in which food products are exposed briefly to highenergy radiation to control insects, mold, bacteria, and spoilage. ...
... technique in which food products are exposed briefly to highenergy radiation to control insects, mold, bacteria, and spoilage. ...
Thyroid, pituitary.and adrenal glands.etc
... • All hormones exert their effect at low blood concentrations • Receptors on or within target tissues are needed for all hormones to exert an effect • Most hormones (except for thyroid and adrenal medullary hormones) are not stored to any great extent and must be produced as needed • Hormones in the ...
... • All hormones exert their effect at low blood concentrations • Receptors on or within target tissues are needed for all hormones to exert an effect • Most hormones (except for thyroid and adrenal medullary hormones) are not stored to any great extent and must be produced as needed • Hormones in the ...
qPCR performed on all samples - York College of Pennsylvania
... -All stages across the timespan mean ΔCT values were statistically the same following an ANOVA((F1.071)=0.8959, P=0.4234) -Removal from thyroid hormone did not have a significant difference at stage 35 (P=0.5377, df=2) following a paired t-test ...
... -All stages across the timespan mean ΔCT values were statistically the same following an ANOVA((F1.071)=0.8959, P=0.4234) -Removal from thyroid hormone did not have a significant difference at stage 35 (P=0.5377, df=2) following a paired t-test ...
30.1 Radioactivity The atom is the smallest unit of achemical
... There are two types of the beta decay. The one is the βdecay and the other is the β+ decay. a- β• neutron decays into a proton • it has the same charge as electron • it has the same mass as electron • it can penetrate with few meters in air. 2 or 3 cm of wood are enough to protect oneself. ...
... There are two types of the beta decay. The one is the βdecay and the other is the β+ decay. a- β• neutron decays into a proton • it has the same charge as electron • it has the same mass as electron • it can penetrate with few meters in air. 2 or 3 cm of wood are enough to protect oneself. ...
malignant hyperthermia
... blood will release pyrogens which have a direct effect on the anterior hypothalamus, causing body temperature to rise, much like raising the temperature setting on a thermostat. ...
... blood will release pyrogens which have a direct effect on the anterior hypothalamus, causing body temperature to rise, much like raising the temperature setting on a thermostat. ...
1.6--NOTES--Detecting Radiation Nuclear Rxtns
... fission and fusion. c. Explain the process half-life as related to radioactive decay. d. Describe nuclear energy, its practical application as an alternative energy source, and its potential problems. ...
... fission and fusion. c. Explain the process half-life as related to radioactive decay. d. Describe nuclear energy, its practical application as an alternative energy source, and its potential problems. ...
Thyroid Stimulating Hormone Receptor Antibody Testing
... Graves disease is an autoimmune disorder, caused by the production of thyroid autoantibodies that activate the TSH-receptor. The antibodies stimulate thyroid hormone production that is not inhibited by the usual feedback mechanism, leading to hyperthyroidism and thyrotoxicosis. Graves disease is the ...
... Graves disease is an autoimmune disorder, caused by the production of thyroid autoantibodies that activate the TSH-receptor. The antibodies stimulate thyroid hormone production that is not inhibited by the usual feedback mechanism, leading to hyperthyroidism and thyrotoxicosis. Graves disease is the ...
File - Ms M - EARL MARRIOTT SECONDARY
... The release of alpha particles is called alpha decay. Alpha particles are slow and penetrate materials much less than the other forms of radiation. A sheet of paper will stop an alpha particle. ...
... The release of alpha particles is called alpha decay. Alpha particles are slow and penetrate materials much less than the other forms of radiation. A sheet of paper will stop an alpha particle. ...
Radiation Tutorial Questions
... radiation can be used from outside the body to treat cancerous cells deep inside the body. 2. Gamma radiation is used to sterilize articles after they have been pre-packed in plastic packets. a. Why is the gamma radiation still effective on these articles? b. Why could syringes not be made of plasti ...
... radiation can be used from outside the body to treat cancerous cells deep inside the body. 2. Gamma radiation is used to sterilize articles after they have been pre-packed in plastic packets. a. Why is the gamma radiation still effective on these articles? b. Why could syringes not be made of plasti ...
Nuclear Chem Notes - Warren County Schools
... • A PET scan detects abnormalities in living tissues without disrupting the tissue. ...
... • A PET scan detects abnormalities in living tissues without disrupting the tissue. ...
AP Chem
... 3. Describe how alpha, beta, and gamma rays each behave when passed through an electric field. Draw a picture. 4. Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? ...
... 3. Describe how alpha, beta, and gamma rays each behave when passed through an electric field. Draw a picture. 4. Why is it not possible to eliminate the hazard of nuclear waste by the process of incineration? ...
1 Intro to Nuclear Chemistry
... • A sample of 3x107 Radon atoms are trapped in a basement that is sealed. The half-life of Radon is 3.83 days. How many radon atoms are left after 31 days? ...
... • A sample of 3x107 Radon atoms are trapped in a basement that is sealed. The half-life of Radon is 3.83 days. How many radon atoms are left after 31 days? ...
Radioactivity
... • Gamma rays are high frequency radiation – they have no mass or charge. • Gamma emission does not change the atomic number or mass number of the atom, and ...
... • Gamma rays are high frequency radiation – they have no mass or charge. • Gamma emission does not change the atomic number or mass number of the atom, and ...
Chapter 25 Nuclear Chemistry
... Nuclear power is good because the fuel lasts longer than fossil fuels with no emissions of CO2(greenhouse gas) or SO2(acid rain gas). 1 kg of Uranium-235 releases more energy than 20,000 tons of dynamite (that's 40 million pounds!) Nuclear power is bad because the nuclei left after fission are radio ...
... Nuclear power is good because the fuel lasts longer than fossil fuels with no emissions of CO2(greenhouse gas) or SO2(acid rain gas). 1 kg of Uranium-235 releases more energy than 20,000 tons of dynamite (that's 40 million pounds!) Nuclear power is bad because the nuclei left after fission are radio ...
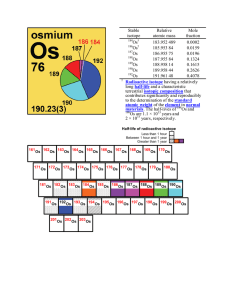
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... gamma rays (gamma radiation) – a stream of high-energy electromagnetic radiation given off by an atomic nucleus undergoing radioactive decay. The energies of gamma rays are higher than those of X-rays; thus, gamma rays have greater penetrating power. half-life (radioactive) – the time interval that ...
... gamma rays (gamma radiation) – a stream of high-energy electromagnetic radiation given off by an atomic nucleus undergoing radioactive decay. The energies of gamma rays are higher than those of X-rays; thus, gamma rays have greater penetrating power. half-life (radioactive) – the time interval that ...
Nuclear Chemistry
... We are exposed to low doses of radiation, including gamma radiation, every day without ill effects. •Radioactive decay heats the interior of Earth. •Radiation occurs in all of our surroundings: air, water, soil. •Cosmic radiation reaches us every day. ...
... We are exposed to low doses of radiation, including gamma radiation, every day without ill effects. •Radioactive decay heats the interior of Earth. •Radiation occurs in all of our surroundings: air, water, soil. •Cosmic radiation reaches us every day. ...
particle - Uplift North Hills
... of only – e so they are less reactive. The b -particle travels about 1 m in air before it is absorbed. It can be stopped by a few mm of Al or other metal ...
... of only – e so they are less reactive. The b -particle travels about 1 m in air before it is absorbed. It can be stopped by a few mm of Al or other metal ...
Endocrinology – Shanice Richardson
... FBC/U&E – low Na+ and high K+ due to lack of mineralcorticoid (aldosterone) Glucose – reduced due to a lack of cortisol Short synacthen (ACTH) test - Adrenal insufficiency is generally excluded if: The 30 minute increment in serum cortisol is >200 nmol/L The maximum serum cortisol level is >550 nmol ...
... FBC/U&E – low Na+ and high K+ due to lack of mineralcorticoid (aldosterone) Glucose – reduced due to a lack of cortisol Short synacthen (ACTH) test - Adrenal insufficiency is generally excluded if: The 30 minute increment in serum cortisol is >200 nmol/L The maximum serum cortisol level is >550 nmol ...
Iodine-131
Iodine-131 (131I), also loosely and nonspecifically called radioiodine, is an important radioisotope of iodine discovered by Glenn Seaborg and John Livingood in 1938 at the University of California, Berkeley. It has a radioactive decay half-life of about eight days. It is associated with nuclear energy, medical diagnostic and treatment procedures, and natural gas production. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Fukushima nuclear crisis. This is because I-131 is a major uranium, plutonium fission product, comprising nearly 3% of the total products of fission (by weight). See fission product yield for a comparison with other radioactive fission products. I-131 is also a major fission product of uranium-233, produced from thorium.Due to its mode of beta decay, iodine-131 is notable for causing mutation and death in cells that it penetrates, and other cells up to several millimeters away. For this reason, high doses of the isotope are sometimes less dangerous than low doses, since they tend to kill thyroid tissues that would otherwise become cancerous as a result of the radiation. For example, children treated with moderate dose of I-131 for thyroid adenomas had a detectable increase in thyroid cancer, but children treated with a much higher dose did not. Likewise, most studies of very-high-dose I-131 for treatment of Graves disease have failed to find any increase in thyroid cancer, even though there is linear increase in thyroid cancer risk with I-131 absorption at moderate doses. Thus, iodine-131 is increasingly less employed in small doses in medical use (especially in children), but increasingly is used only in large and maximal treatment doses, as a way of killing targeted tissues. This is known as ""therapeutic use.""Iodine-131 can be ""seen"" by nuclear medicine imaging techniques (i.e., gamma cameras) whenever it is given for therapeutic use, since about 10% of its energy and radiation dose is via gamma radiation. However, since the other 90% of radiation (beta radiation) causes tissue damage without contributing to any ability to see or ""image"" the isotope, other less-damaging radioisotopes of iodine such as iodine-123 (see isotopes of iodine) are preferred in situations when only nuclear imaging is required. The isotope I-131 is still occasionally used for purely diagnostic (i.e., imaging) work, due to its low expense compared to other iodine radioisotopes. Very small medical imaging doses of I-131 have not shown any increase in thyroid cancer. The low-cost availability of I-131, in turn, is due to the relative ease of creating I-131 by neutron bombardment of natural tellurium in a nuclear reactor, then separating I-131 out by various simple methods (i.e., heating to drive off the volatile iodine). By contrast, other iodine radioisotopes are usually created by far more expensive techniques, starting with reactor radiation of expensive capsules of pressurized xenon gas.Iodine-131 is also one of the most commonly used gamma-emitting radioactive industrial tracer. Radioactive tracer isotopes are injected with hydraulic fracturing fluid to determine the injection profile and location of fractures created by hydraulic fracturing.Much smaller incidental doses of iodine-131 than those used in medical therapeutic procedures, are thought to be the major cause of increased thyroid cancers after accidental nuclear contamination. These cancers happen from residual tissue radiation damage caused by the I-131, and usually appear years after exposure, long after the I-131 has decayed.