Name:______ Chemistry 114 First Hour Exam
... A probonding sigma orbital (electrons along a line connecting the atoms) hybridized from a 2p atomic orbital ...
... A probonding sigma orbital (electrons along a line connecting the atoms) hybridized from a 2p atomic orbital ...
Chapter 4 REVIEW
... 21. Ionic compounds and metals have different physical properties because of the different forces involved. For example, while sodium chloride and nickel have nearly identical molar masses, their melting points, conductivity, and solubility in water are quite different. (a) Explain the large differe ...
... 21. Ionic compounds and metals have different physical properties because of the different forces involved. For example, while sodium chloride and nickel have nearly identical molar masses, their melting points, conductivity, and solubility in water are quite different. (a) Explain the large differe ...
Intermolecular Forces
... - intermolecular bonds are broken when a molecular compound melts and boils 1. Hydrogen bonding -water is a highly polar molecule: large electronegativity difference and shape (bent) boiling points ...
... - intermolecular bonds are broken when a molecular compound melts and boils 1. Hydrogen bonding -water is a highly polar molecule: large electronegativity difference and shape (bent) boiling points ...
Unit 1 - Learning Objectives
... (iii) Isotopes Isotopes are atoms with the same atomic number but different mass numbers. Most elements exist as a mixture of isotopes. The relative atomic mass of an element is rarely a whole number. d) Bonding, structure and properties Bonding Atoms can be held together by bonds. In form ...
... (iii) Isotopes Isotopes are atoms with the same atomic number but different mass numbers. Most elements exist as a mixture of isotopes. The relative atomic mass of an element is rarely a whole number. d) Bonding, structure and properties Bonding Atoms can be held together by bonds. In form ...
6.1 Organizing the Periodic Table
... not strong as ionic or covalent bonds but they are strong enough to hold molecules together in a solid or a liquid ...
... not strong as ionic or covalent bonds but they are strong enough to hold molecules together in a solid or a liquid ...
Covalent Bonds - WordPress.com
... • An ionic bond is an attracted between ions of opposite charge in an ionic compound • An example is the transfer of an electron from sodium to chlorine • When sodium and chlorine interection with together.what’ll happen? • After the transfer of an electron, both atoms have charges • A charged atom ...
... • An ionic bond is an attracted between ions of opposite charge in an ionic compound • An example is the transfer of an electron from sodium to chlorine • When sodium and chlorine interection with together.what’ll happen? • After the transfer of an electron, both atoms have charges • A charged atom ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
AlBr3 E IO Ionic FU C O Cov Molec C IO Cov Molec Sn E N/A N/A
... combine in the ratio of small, whole numbers to form compounds. ...
... combine in the ratio of small, whole numbers to form compounds. ...
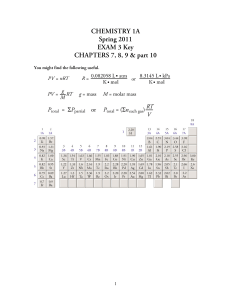
Exam 3 Key
... For each of the following, write the word, words, or number in each blank that best completes each sentence. (2 points each) 1. The condition of an atom that has at least one of its electrons in orbitals that do not represent the lowest possible potential energy is called a(n) excited state. 2. A(n ...
... For each of the following, write the word, words, or number in each blank that best completes each sentence. (2 points each) 1. The condition of an atom that has at least one of its electrons in orbitals that do not represent the lowest possible potential energy is called a(n) excited state. 2. A(n ...
What do you know about light?
... • The atomic number of an element provides information about its atomic structure. • For example, the atomic number of fluorine is 9, indicating that there must be 9 protons in the nucleus. ...
... • The atomic number of an element provides information about its atomic structure. • For example, the atomic number of fluorine is 9, indicating that there must be 9 protons in the nucleus. ...
Chemistry - El Camino College
... a. In ionic reactions, atoms give or take _________ to get a full outer electron orbital b. Oppositely charged ions are strongly attracted to each other, form _______ bonds, and are called ______ or electrolytes 2. _________ Bonds are strong chemical bonds between atoms that result from the _______ ...
... a. In ionic reactions, atoms give or take _________ to get a full outer electron orbital b. Oppositely charged ions are strongly attracted to each other, form _______ bonds, and are called ______ or electrolytes 2. _________ Bonds are strong chemical bonds between atoms that result from the _______ ...
Microsoft Word
... whenever possible, valence electrons in covalent compounds distribute so that each main-group element is surrounded by 8 electrons (except hydrogen which wants 2 electrons around it). ...
... whenever possible, valence electrons in covalent compounds distribute so that each main-group element is surrounded by 8 electrons (except hydrogen which wants 2 electrons around it). ...
Honors Unit 5 Practice Test
... a. stronger than the forces among formula units in ionic bonding. b. weaker than the forces among formula units in ionic bonding. c. approximately equal to the forces among formula units in ionic bonding. d. zero. Compared with nonmetals, the number of valence electrons in metals is generally a. sma ...
... a. stronger than the forces among formula units in ionic bonding. b. weaker than the forces among formula units in ionic bonding. c. approximately equal to the forces among formula units in ionic bonding. d. zero. Compared with nonmetals, the number of valence electrons in metals is generally a. sma ...
Atomic Radii Answers File
... A.R. increases going down a group. An extra shell is being added in successive elements and the electrons in the outer shell are “shielded” from the nucleus by the inner shells. There is a decreasing attractive pull on them from the nucleus. ...
... A.R. increases going down a group. An extra shell is being added in successive elements and the electrons in the outer shell are “shielded” from the nucleus by the inner shells. There is a decreasing attractive pull on them from the nucleus. ...
CHEMISTRY 113 EXAM 3(A)
... D. [Ar]3s1 10. The effective charge acting on the valence electrons of S is: A. +3 B. -2 C. +6 D. +1 11. The atomic radius of main-group elements generally increases: A. down the group B. across the period C. between metals and non-metals D. when the element is ionized 12. Which of the following at ...
... D. [Ar]3s1 10. The effective charge acting on the valence electrons of S is: A. +3 B. -2 C. +6 D. +1 11. The atomic radius of main-group elements generally increases: A. down the group B. across the period C. between metals and non-metals D. when the element is ionized 12. Which of the following at ...
Chemistry Study Guide
... 2. Which particles are located in the nucleus of the atom? Protons & neutrons 3. What is a molecule? Be able to identify one from a list. Smallest particles of a substance with the same properties of the substance (ex. H2O, O2, N2, CO2) 4. Use the periodic table to explain how elements form bonds. E ...
... 2. Which particles are located in the nucleus of the atom? Protons & neutrons 3. What is a molecule? Be able to identify one from a list. Smallest particles of a substance with the same properties of the substance (ex. H2O, O2, N2, CO2) 4. Use the periodic table to explain how elements form bonds. E ...
Chapter 2 Learning Objectives
... a. The threshold frequency b. The correlation between radiant intensity and the number of emitted electrons 3. Understand that the electrons of an atom behave as waves, resulting in quantum numbers 4. Know all four quantum numbers (n, l, ml, ms), and the dependency rules between them 5. Be able to u ...
... a. The threshold frequency b. The correlation between radiant intensity and the number of emitted electrons 3. Understand that the electrons of an atom behave as waves, resulting in quantum numbers 4. Know all four quantum numbers (n, l, ml, ms), and the dependency rules between them 5. Be able to u ...
$doc.title
... Note for He2 (4 electrons), Pauli principle means two e’s in antibonding state as well as bonding state so no overall energy saving (inert gases – no bond - no He2) Mid-periodic table elements (half-filled orbitals) tend to have strongest bonds (e.g. melting points. etc.) ...
... Note for He2 (4 electrons), Pauli principle means two e’s in antibonding state as well as bonding state so no overall energy saving (inert gases – no bond - no He2) Mid-periodic table elements (half-filled orbitals) tend to have strongest bonds (e.g. melting points. etc.) ...
Review for Exam 1
... Determine how many of each ion type is needed for an overall charge of zero. When the cation and anion have different charges, use the ion charges to determine the number of ions of each needed. ...
... Determine how many of each ion type is needed for an overall charge of zero. When the cation and anion have different charges, use the ion charges to determine the number of ions of each needed. ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.