MIDTERM REVIEW GAME 16-17
... 5) The product of the frequency and the wavelength is the: 1. Speed of the wave 2. Number of waves passing a point per ...
... 5) The product of the frequency and the wavelength is the: 1. Speed of the wave 2. Number of waves passing a point per ...
Valence Electrons and Chemical Bonding
... eight electrons in their outer energy level or, in the case of elements 1-5, two in their outer shell level. ...
... eight electrons in their outer energy level or, in the case of elements 1-5, two in their outer shell level. ...
AP Chapter 9 Molecular Shapes
... π bonds • π bonds must lie in the same plane, therefore, the presence of π bonds makes the molecule slightly rigid. ...
... π bonds • π bonds must lie in the same plane, therefore, the presence of π bonds makes the molecule slightly rigid. ...
File - Science With BLT
... c. changes the formula so that it no longer represents that compound. d. has no effect on the formula. Which formula does NOT represent a molecule? a. H2O (water) c. CO2 (carbon dioxide) b. NH3 (ammonia) d. NaCl (table salt) What is the formula for zinc fluoride? a. ZnF c. Zn2F b. ZnF2 d. Zn2F3 What ...
... c. changes the formula so that it no longer represents that compound. d. has no effect on the formula. Which formula does NOT represent a molecule? a. H2O (water) c. CO2 (carbon dioxide) b. NH3 (ammonia) d. NaCl (table salt) What is the formula for zinc fluoride? a. ZnF c. Zn2F b. ZnF2 d. Zn2F3 What ...
Chemistry I Honors – Semester Exam Review – Fall 2000
... 36. Are the following properties characteristics of ionic, covalent, or metallic bonding? a. These bonds are formed by delocalized electrons in an “electron sea.” b. These bonds involve a transfer of electrons. c. Substances containing these bonds are malleable and have very high melting points. d. ...
... 36. Are the following properties characteristics of ionic, covalent, or metallic bonding? a. These bonds are formed by delocalized electrons in an “electron sea.” b. These bonds involve a transfer of electrons. c. Substances containing these bonds are malleable and have very high melting points. d. ...
Course Syllabus - Honors Chemistry
... b. Chemical bonds between atoms in molecules such as H2, CH4, NH3, H2CCH2, N2, Cl2, and many large biological molecules are covalent. c. Salt crystals, such as NaCl, are repeating patterns of positive and negative ions held together by electrostatic attraction. d. Atoms and molecules in liquids move ...
... b. Chemical bonds between atoms in molecules such as H2, CH4, NH3, H2CCH2, N2, Cl2, and many large biological molecules are covalent. c. Salt crystals, such as NaCl, are repeating patterns of positive and negative ions held together by electrostatic attraction. d. Atoms and molecules in liquids move ...
Vocabulary Notes
... A solid where the atoms are arranged in a predictable repeating pattern. Diamond forms in a crystal structure as does NaCl. ...
... A solid where the atoms are arranged in a predictable repeating pattern. Diamond forms in a crystal structure as does NaCl. ...
Semester Exam Review - Teach-n-Learn-Chem
... 36. Are the following properties characteristics of ionic, covalent, or metallic bonding? a. These bonds are formed by delocalized electrons in an “electron sea.” b. These bonds involve a transfer of electrons. c. Substances containing these bonds are malleable and have very high melting points. d. ...
... 36. Are the following properties characteristics of ionic, covalent, or metallic bonding? a. These bonds are formed by delocalized electrons in an “electron sea.” b. These bonds involve a transfer of electrons. c. Substances containing these bonds are malleable and have very high melting points. d. ...
File - Romona Olton
... electronegativity difference b/w elements (atoms) is zero or relatively small. In covalent compounds the bonds b/w atoms within a molecule (intramolecular bonding) are relatively strong BUT the attractive forces between molecules (intermolecular forces) are relatively weak. Hence covalent compou ...
... electronegativity difference b/w elements (atoms) is zero or relatively small. In covalent compounds the bonds b/w atoms within a molecule (intramolecular bonding) are relatively strong BUT the attractive forces between molecules (intermolecular forces) are relatively weak. Hence covalent compou ...
Chemistry I Honors – Semester Exam Review – Fall 2000
... 36. Are the following properties characteristics of ionic, covalent, or metallic bonding? a. These bonds are formed by delocalized electrons in an “electron sea.” b. These bonds involve a transfer of electrons. c. Substances containing these bonds are malleable and have very high melting points. d. ...
... 36. Are the following properties characteristics of ionic, covalent, or metallic bonding? a. These bonds are formed by delocalized electrons in an “electron sea.” b. These bonds involve a transfer of electrons. c. Substances containing these bonds are malleable and have very high melting points. d. ...
Chapt9
... When multiple bonds are present, a single Lewis structure may not adequately describe the compound or ion -- occurs whenever there is a "choice" of where to put a multiple bond. e.g., the HCO2- ion is a "resonance hybrid" of two "contributing resonance structures" ...
... When multiple bonds are present, a single Lewis structure may not adequately describe the compound or ion -- occurs whenever there is a "choice" of where to put a multiple bond. e.g., the HCO2- ion is a "resonance hybrid" of two "contributing resonance structures" ...
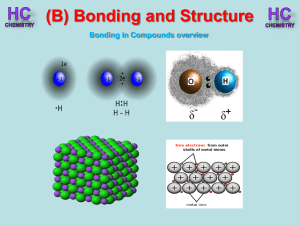
Lesson 1 - Bonding in compounds overview
... - non-metals gain electrons to form negative ions - electrons are transferred from metals to non-metals ...
... - non-metals gain electrons to form negative ions - electrons are transferred from metals to non-metals ...
BASIC CHEMISTRY
... Draw Carbon Nitrogen atomic number: 7 atomic mass: 14 Carbon atomic number 6 atomic mass: 12 ...
... Draw Carbon Nitrogen atomic number: 7 atomic mass: 14 Carbon atomic number 6 atomic mass: 12 ...
stable structure - Rothschild Science
... 4 things can happen when elements come together to form compounds? ...
... 4 things can happen when elements come together to form compounds? ...
Chapter 9 Notes - UIC Department of Chemistry
... (originating in their charges.) Lewis structures for ionic compounds are written by putting two Lewis ionic symbols together. F− Lewis structures for ions: Mg2+ Ionic compounds-put the ions together as in a formula: MgF2 Lewis Structures for Covalent Compounds 1) Nonmetals tend to share electrons in ...
... (originating in their charges.) Lewis structures for ionic compounds are written by putting two Lewis ionic symbols together. F− Lewis structures for ions: Mg2+ Ionic compounds-put the ions together as in a formula: MgF2 Lewis Structures for Covalent Compounds 1) Nonmetals tend to share electrons in ...
Chapter 2: You must understand chemistry to understand life (and to
... C. since atoms have the same number of electrons as protons, they meet this need to have a full valence shell by sharing, giving up, or acquiring electrons from other atoms; this forms chemical bonds 1. a chemical bond is a reduced energy state 2. bond energy is the amount of energy required to brea ...
... C. since atoms have the same number of electrons as protons, they meet this need to have a full valence shell by sharing, giving up, or acquiring electrons from other atoms; this forms chemical bonds 1. a chemical bond is a reduced energy state 2. bond energy is the amount of energy required to brea ...
Slayt 1
... charges of the ions in an ionic compound. The result of this bond is a neutral compound. Metals and non-metals usually combine by forming ionic bonds. http://www.mhhe.com/physsci/chemistry/animations/chang_7e_esp/bom1s2_11.swf ...
... charges of the ions in an ionic compound. The result of this bond is a neutral compound. Metals and non-metals usually combine by forming ionic bonds. http://www.mhhe.com/physsci/chemistry/animations/chang_7e_esp/bom1s2_11.swf ...
Notes
... • Identify the three subatomic particles found in atoms. • Explain how all of the isotopes of an element are similar and how they are different. • Explain how compounds are different from their component elements. • Describe the two main types of chemical bonds ...
... • Identify the three subatomic particles found in atoms. • Explain how all of the isotopes of an element are similar and how they are different. • Explain how compounds are different from their component elements. • Describe the two main types of chemical bonds ...
Unit chemical bonds
... • Two ions have opposite electrical charges therefore attracted to each other – Holds Ionic Bonds together • Weak bond between elements ...
... • Two ions have opposite electrical charges therefore attracted to each other – Holds Ionic Bonds together • Weak bond between elements ...
Chemical Bond - Cobb Learning
... Properties of Ionic compounds – Ionic compounds are usually solids at room temperature Melting and Boiling Points – Ionic compounds have much higher melting points and boiling points than covalent compounds –– Ionic compounds typically melt at several hundred degrees Celsius •They exist in a crysta ...
... Properties of Ionic compounds – Ionic compounds are usually solids at room temperature Melting and Boiling Points – Ionic compounds have much higher melting points and boiling points than covalent compounds –– Ionic compounds typically melt at several hundred degrees Celsius •They exist in a crysta ...
Chemical Bonding Quiz
... Study Guide: Chemical Bonding Quiz Students should be able to understand and apply the following Chemical Bonding concepts: ...
... Study Guide: Chemical Bonding Quiz Students should be able to understand and apply the following Chemical Bonding concepts: ...
MatterPP4
... elements that are chemically combined. Most compounds have totally different properties from the elements of which they are composed. Chemical bonds are the forces that hold the elements together in a compound creating a state of stability. ...
... elements that are chemically combined. Most compounds have totally different properties from the elements of which they are composed. Chemical bonds are the forces that hold the elements together in a compound creating a state of stability. ...
Chapter 5 Notes: The Structure of Matter
... This makes its charge 1+ because there are more + charges ...
... This makes its charge 1+ because there are more + charges ...
1. Define each of the following terms: a.Alkaline earth metals
... an example of a chemical change, also heat and light are produced and the change is difficult to reverse all characteristics of a chemical change.) ...
... an example of a chemical change, also heat and light are produced and the change is difficult to reverse all characteristics of a chemical change.) ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.