Rubidium
... Rubidium is not made by the same method as sodium as might have been expected. This is because the rubidium metal, once formed by electrolysis of liquid rubidium chloride (RbCl), is too soluble in the molten salt. cathode: Rb+(l) + e- Rb (l) anode: Cl-(l) 1/2Cl2 (g) + e- ...
... Rubidium is not made by the same method as sodium as might have been expected. This is because the rubidium metal, once formed by electrolysis of liquid rubidium chloride (RbCl), is too soluble in the molten salt. cathode: Rb+(l) + e- Rb (l) anode: Cl-(l) 1/2Cl2 (g) + e- ...
PART 3-ICHO 11-15
... sodium orthophosphate, barium hydroxide, lead nitrate, potassium hydroxide, aluminium sulphate, sodium carbonate. Using only these solutions as reagents, determine in which of the numbered test tubes each of the above given substances, is found. Draw up a plan of the analysis and write equations of ...
... sodium orthophosphate, barium hydroxide, lead nitrate, potassium hydroxide, aluminium sulphate, sodium carbonate. Using only these solutions as reagents, determine in which of the numbered test tubes each of the above given substances, is found. Draw up a plan of the analysis and write equations of ...
chemistry - Textbooks Online
... method - Calculations using densities and specific gravities - Calculation of formula weight - Understanding Avogadro’s number - Mole concept-mole fraction of the solvent and solute - Conversion of grams into moles and moles into grams Calculation of empirical formula from quantitative analysis and ...
... method - Calculations using densities and specific gravities - Calculation of formula weight - Understanding Avogadro’s number - Mole concept-mole fraction of the solvent and solute - Conversion of grams into moles and moles into grams Calculation of empirical formula from quantitative analysis and ...
Supporting Information - Royal Society of Chemistry
... required to achieve about 50% inhibition of the enzyme (2.5 M) in the presence of 75 M substrate (p-nitrophenyl acetate) were first determined (25 mM HEPES buffer, pH = 7.0; absorbance followed at 450 nm). The kinetic data were analyzed via the double reciprocal plots of the initial rates of the e ...
... required to achieve about 50% inhibition of the enzyme (2.5 M) in the presence of 75 M substrate (p-nitrophenyl acetate) were first determined (25 mM HEPES buffer, pH = 7.0; absorbance followed at 450 nm). The kinetic data were analyzed via the double reciprocal plots of the initial rates of the e ...
Chapter 1: Chemistry: The Study of Change
... 24. Oxidation of a hydrocarbon gave a product composed of carbon, hydrogen, and oxygen. The product that was purified and sent off for elemental analysis giving the following mass percents: 68.85% C and 4.95% H. Determine the empirical formula of this compound. (Section: 3.6) Ans: C7H6O2 25. Acetyle ...
... 24. Oxidation of a hydrocarbon gave a product composed of carbon, hydrogen, and oxygen. The product that was purified and sent off for elemental analysis giving the following mass percents: 68.85% C and 4.95% H. Determine the empirical formula of this compound. (Section: 3.6) Ans: C7H6O2 25. Acetyle ...
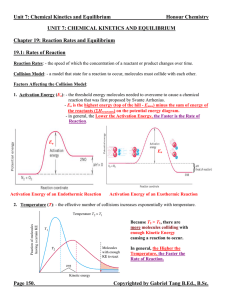
Unit 7 Reaction Rates and Equilibrium Notes
... Equilibrium Expression: - an expression relating the concentrations or pressures of the reactants and products when they are at the state of equilibrium. - it takes the form of the individual products raised to the power of their respective coefficients divided by the individual reactants raised to ...
... Equilibrium Expression: - an expression relating the concentrations or pressures of the reactants and products when they are at the state of equilibrium. - it takes the form of the individual products raised to the power of their respective coefficients divided by the individual reactants raised to ...
chapter_14 Equilibr
... do not appear in the equilibrium constant expressions. 3. The equilibrium constant is a dimensionless quantity. 4. In quoting a value for the equilibrium constant, you must specify the balanced equation and the temperature. 5. If a reaction can be expressed as a sum of two or more reactions, the equ ...
... do not appear in the equilibrium constant expressions. 3. The equilibrium constant is a dimensionless quantity. 4. In quoting a value for the equilibrium constant, you must specify the balanced equation and the temperature. 5. If a reaction can be expressed as a sum of two or more reactions, the equ ...
Chemistry – 5071
... This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need of students to develop skills that will be of long term value in an increasi ...
... This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need of students to develop skills that will be of long term value in an increasi ...
9/10/10 1 Chemistry 121: Atomic and Molecular Chemistry
... • Different properties explained by assuming atoms are not the same type. The second hypothesis: • To form a certain compound, need atoms of right elements, and proportion. • Extension of a law published in 1799 by Joseph Proust • ie., analyze samples of carbon dioxide gas different sources, find th ...
... • Different properties explained by assuming atoms are not the same type. The second hypothesis: • To form a certain compound, need atoms of right elements, and proportion. • Extension of a law published in 1799 by Joseph Proust • ie., analyze samples of carbon dioxide gas different sources, find th ...
- Mendeley Data
... changing the quantity of Schiff base complex also checked on the epoxidation of cyclooctene (Table 6). Different quantity of Schiff base complex (0.005, 0.01 and 0.02 mmol) were used in the epoxidation of cyclooctene in the CHCl3 and in the presence of TBHP However, the maximum epoxidation yield wa ...
... changing the quantity of Schiff base complex also checked on the epoxidation of cyclooctene (Table 6). Different quantity of Schiff base complex (0.005, 0.01 and 0.02 mmol) were used in the epoxidation of cyclooctene in the CHCl3 and in the presence of TBHP However, the maximum epoxidation yield wa ...
Exam 980415 - NTOU-Chem
... 22) The pH of acid rain may be as low as 2.80. What is the H3O+ in such acidic rain? A) 1.6 B) 630 C) 6.3 × 10- 12 D) 1.6 × 10-3 Answer: D 23) Which of the following salts is most soluble in pure water? A) ZnS, Ksp = 1.6 × 10-24 B) CuS, Ksp = 8.7 × 10-36 C) PbI2 , Ksp = 7.1 × 10-9 D) HgS, Ksp = 2.0 ...
... 22) The pH of acid rain may be as low as 2.80. What is the H3O+ in such acidic rain? A) 1.6 B) 630 C) 6.3 × 10- 12 D) 1.6 × 10-3 Answer: D 23) Which of the following salts is most soluble in pure water? A) ZnS, Ksp = 1.6 × 10-24 B) CuS, Ksp = 8.7 × 10-36 C) PbI2 , Ksp = 7.1 × 10-9 D) HgS, Ksp = 2.0 ...
2.6 M - Thierry Karsenti
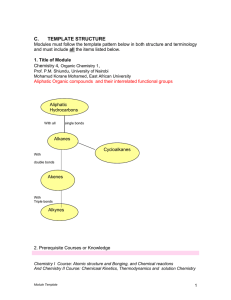
... contain rings have he formula CnH2n+2. An alkane in the shape of a ring is called a cycloalkane. Cycloalkanes have the formula CnH2n. ..4. An alkene is a compound that has at least one double bond. A straight-chain alkene with one double bond has the formula CnH2n. The functional group for an alkene ...
... contain rings have he formula CnH2n+2. An alkane in the shape of a ring is called a cycloalkane. Cycloalkanes have the formula CnH2n. ..4. An alkene is a compound that has at least one double bond. A straight-chain alkene with one double bond has the formula CnH2n. The functional group for an alkene ...
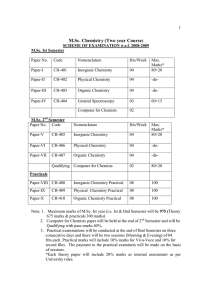
M.Sc. Chemistry (Two year Course)
... mobility of ions, ionic drift velocity and its relation with current density, Einstein relation between the absolute mobility and diffusion coefficient, the Stokes- Einstein relation , the Nernst -Einstein equation, Waldens rule, the Rate- Process approach to ionic migration , the Rate process equat ...
... mobility of ions, ionic drift velocity and its relation with current density, Einstein relation between the absolute mobility and diffusion coefficient, the Stokes- Einstein relation , the Nernst -Einstein equation, Waldens rule, the Rate- Process approach to ionic migration , the Rate process equat ...
Homework1-4-Answers
... 24. Oxidation of a hydrocarbon gave a product composed of carbon, hydrogen, and oxygen. The product that was purified and sent off for elemental analysis giving the following mass percents: 68.85% C and 4.95% H. Determine the empirical formula of this compound. (Section: 3.6) Ans: C7H6O2 25. Acetyle ...
... 24. Oxidation of a hydrocarbon gave a product composed of carbon, hydrogen, and oxygen. The product that was purified and sent off for elemental analysis giving the following mass percents: 68.85% C and 4.95% H. Determine the empirical formula of this compound. (Section: 3.6) Ans: C7H6O2 25. Acetyle ...
Document
... Any property that only depends on object’s current state or condition Independence from method, path or mechanism by which change occurs is important feature of all state functions Some State functions, E, P, t, and V : ...
... Any property that only depends on object’s current state or condition Independence from method, path or mechanism by which change occurs is important feature of all state functions Some State functions, E, P, t, and V : ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... • The molar quantities indicated by the coefficients in a balanced equation are called stoichiometrically equivalent quantities. • Stoichiometric factors (or molar ratios) may be used to convert between quantities of reactants and products in a reaction. • It is important to realize that the stoichi ...
... • The molar quantities indicated by the coefficients in a balanced equation are called stoichiometrically equivalent quantities. • Stoichiometric factors (or molar ratios) may be used to convert between quantities of reactants and products in a reaction. • It is important to realize that the stoichi ...
sample problem - KFUPM Resources
... More practical aspect of Gibb’s free energy • The standard free energy (ΔGrxn) of a system is the change in free energy when reactants in their standard states are converted to products in their standard states. • Standard states are: ...
... More practical aspect of Gibb’s free energy • The standard free energy (ΔGrxn) of a system is the change in free energy when reactants in their standard states are converted to products in their standard states. • Standard states are: ...
Mole-Volume Conversion Assignment
... 8. Get two pieces of filter paper. Weigh both together and record the mass: ______________________ g 9. With the pieces of filter paper together, fold them to make a cone (you may want to wet the filter paper). Place the empty beaker under the funnel. Swirl the mixture & pour it into the funnel. Add ...
... 8. Get two pieces of filter paper. Weigh both together and record the mass: ______________________ g 9. With the pieces of filter paper together, fold them to make a cone (you may want to wet the filter paper). Place the empty beaker under the funnel. Swirl the mixture & pour it into the funnel. Add ...
pH and ORP Applications Cyanide Waste Treatment
... Gold ORP electrodes give more reliable measurement than platinum for this application. Platinum may catalyze some additional reactions at its surface and is more subject to coating than gold. The solubility of gold in cyanide solutions does not present a problem, since it is in contact primarily wit ...
... Gold ORP electrodes give more reliable measurement than platinum for this application. Platinum may catalyze some additional reactions at its surface and is more subject to coating than gold. The solubility of gold in cyanide solutions does not present a problem, since it is in contact primarily wit ...
chemical equilibrium type 1
... technique that is used to determine whether a reaction it truly at equilibrium is to approach equilibrium starting with reactants in one experiment and starting with products in another. If the same value of the reaction quotient is observed when the concentrations stop changing in both experiments, ...
... technique that is used to determine whether a reaction it truly at equilibrium is to approach equilibrium starting with reactants in one experiment and starting with products in another. If the same value of the reaction quotient is observed when the concentrations stop changing in both experiments, ...
Student Study Guide Chemistry 534
... function in the upper atmosphere by preventing harmful ultraviolet radiation from reaching the Earth. Air conditioning increase our comfort in hot climatic conditions and one of the gases that has been used for cooling is freon (a chlorinated fluorocarbon or CFC). This gas is effective for cooling b ...
... function in the upper atmosphere by preventing harmful ultraviolet radiation from reaching the Earth. Air conditioning increase our comfort in hot climatic conditions and one of the gases that has been used for cooling is freon (a chlorinated fluorocarbon or CFC). This gas is effective for cooling b ...