Rutile titanium dioxide nanoparticles and ordered acicular
... titanium molar ratio of from 0.02 to 0.4, although best results have been observed when the ratio is from 0.02 to 0.2. The solution is simultaneously heated to a temperature in the range of from 60° C. to 80° C. with constant stirring. There ...
... titanium molar ratio of from 0.02 to 0.4, although best results have been observed when the ratio is from 0.02 to 0.2. The solution is simultaneously heated to a temperature in the range of from 60° C. to 80° C. with constant stirring. There ...
Chap 3 - HCC Learning Web
... Hint: See both 5th ed. and 6th ed. Section 9.3. In the formula, Ca(NO3)2, it indicates there are 1 Ca, 1x2 = 2 N and 3x2 = 6 O. Go to the periodic table and locate the atomic mass for each atom. Thus the formula mass of Ca(NO3)2 = 1x40 + 2x14 + 6x16 = 164. Note that if the unit given as amu (referri ...
... Hint: See both 5th ed. and 6th ed. Section 9.3. In the formula, Ca(NO3)2, it indicates there are 1 Ca, 1x2 = 2 N and 3x2 = 6 O. Go to the periodic table and locate the atomic mass for each atom. Thus the formula mass of Ca(NO3)2 = 1x40 + 2x14 + 6x16 = 164. Note that if the unit given as amu (referri ...
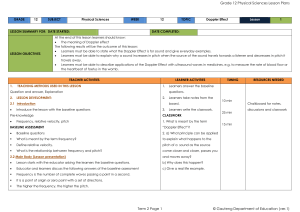
Physical Sciences Grade 12 Term 2
... The siren of a burglar alarm system has a frequency of 960 Hz. During a patrol, a security officer, travelling in his car, hears the siren of the alarm of a house and approaches the house at 2.1 Name the phenomenon that explains the change in the observed frequency. 2.2 Calculate the speed at which ...
... The siren of a burglar alarm system has a frequency of 960 Hz. During a patrol, a security officer, travelling in his car, hears the siren of the alarm of a house and approaches the house at 2.1 Name the phenomenon that explains the change in the observed frequency. 2.2 Calculate the speed at which ...
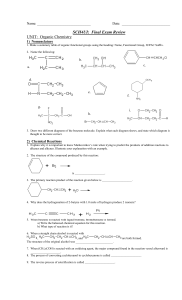
Name:
... According to these results, what would be the initial rate (in mol/(L·s)) if all three concentrations are: [BrO3-]=[Br-]=[H+]=0.20 mol/L? 2. Use the following diagram to answer the questions below. a) Is the reaction exothermic or endothermic? Explain. b) What letter represents the activation energy ...
... According to these results, what would be the initial rate (in mol/(L·s)) if all three concentrations are: [BrO3-]=[Br-]=[H+]=0.20 mol/L? 2. Use the following diagram to answer the questions below. a) Is the reaction exothermic or endothermic? Explain. b) What letter represents the activation energy ...
Chemistry II - Mr. Dougan`s Wonderful World of Chemistry
... reagents have been mixed. The endpoint of a titration for reactions of acids and bases is usually indicated by a third reagent, the indicator, which has an abrupt and distinctive color change at the hydrogen ion concentration which is present after neutralization has occurred. A good indicator of ch ...
... reagents have been mixed. The endpoint of a titration for reactions of acids and bases is usually indicated by a third reagent, the indicator, which has an abrupt and distinctive color change at the hydrogen ion concentration which is present after neutralization has occurred. A good indicator of ch ...
Equilibrium Part 2
... a system at equilibrium. The system attempts to remove added heat by using it up in the forward reaction (endothermic reaction). The equilibrium position shifts towards the right (products). The concentration of NO2 increases and the concentration of N2O4 decreases. We can also think of adding heat ...
... a system at equilibrium. The system attempts to remove added heat by using it up in the forward reaction (endothermic reaction). The equilibrium position shifts towards the right (products). The concentration of NO2 increases and the concentration of N2O4 decreases. We can also think of adding heat ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... Members of the SCHOLAR Forum may reproduce this publication in whole or in part for educational purposes within their establishment providing that no profit accrues at any stage, Any other use of the materials is governed by the general copyright statement that follows. All rights reserved. No part ...
... Members of the SCHOLAR Forum may reproduce this publication in whole or in part for educational purposes within their establishment providing that no profit accrues at any stage, Any other use of the materials is governed by the general copyright statement that follows. All rights reserved. No part ...
Electrochemistry
... The electrode potential and the EMF of the cell depend upon the nature of the electrode, temperature and the activities (concentrations) of the ions in solution. The variation of electrode and cell potential with concentration of ions in solution can be obtained from thermodynamic considerations. Fo ...
... The electrode potential and the EMF of the cell depend upon the nature of the electrode, temperature and the activities (concentrations) of the ions in solution. The variation of electrode and cell potential with concentration of ions in solution can be obtained from thermodynamic considerations. Fo ...
sample chapter
... The equation describing the precipitation of lead iodide on page 100 is called a molecular equation because the formulas of the compounds are written as though all species existed as molecules or whole units. A molecular equation is useful because it identifies the reagents (that is, lead nitrate an ...
... The equation describing the precipitation of lead iodide on page 100 is called a molecular equation because the formulas of the compounds are written as though all species existed as molecules or whole units. A molecular equation is useful because it identifies the reagents (that is, lead nitrate an ...
SQA CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
Exam 2 Key
... 10. At elevated temperatures, solid sodium chlorate (NaClO3, molar mass 106.44 g/mol) decomposes to produce sodium chloride and oxygen gas. In an experiment, a 0.8765 g sample of impure sodium chlorate was heated until the production of oxygen ceased. The O2 gas was collected over water. The collec ...
... 10. At elevated temperatures, solid sodium chlorate (NaClO3, molar mass 106.44 g/mol) decomposes to produce sodium chloride and oxygen gas. In an experiment, a 0.8765 g sample of impure sodium chlorate was heated until the production of oxygen ceased. The O2 gas was collected over water. The collec ...
Chapter One Hemilabile Ligands in Transition
... hard metal ions such as Cr3+ and Al3+ are low in electron density and require good σdonor ligands. Low oxidation state metals such as Ag+, Hg2+, which are soft metals, tend to form strong complexes with unsaturated or polarizable ligands, including I-, PPh3 or C2H4 which are all soft ligands.1 The s ...
... hard metal ions such as Cr3+ and Al3+ are low in electron density and require good σdonor ligands. Low oxidation state metals such as Ag+, Hg2+, which are soft metals, tend to form strong complexes with unsaturated or polarizable ligands, including I-, PPh3 or C2H4 which are all soft ligands.1 The s ...
RUMPLE-DISSERTATION-2014 - SMARTech Home
... Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty sure they have each forgotten more than I’ll ever learn, and they still know so muc ...
... Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty sure they have each forgotten more than I’ll ever learn, and they still know so muc ...
CHE 1402 Lab Manual
... the lecture notes. However, we cannot guarantee the synchronization of lectures and laboratory experiments. For this, certain experiments may come before or after the material that has been covered in the lecture. The lab manual contains background information and procedures for the experiments you ...
... the lecture notes. However, we cannot guarantee the synchronization of lectures and laboratory experiments. For this, certain experiments may come before or after the material that has been covered in the lecture. The lab manual contains background information and procedures for the experiments you ...