Ans:- (i) Gluconic acid - Kendriya Vidyalaya No.2, Kribhco, Surat
... with increasing dilution the molar conductance increases rapidly because of greater extent of ionization at greater dilution. These are dissociated more or less completely at low dilutions. Their molar conductances therefore show slight increase with increasing dilution. At low conc, the interionic ...
... with increasing dilution the molar conductance increases rapidly because of greater extent of ionization at greater dilution. These are dissociated more or less completely at low dilutions. Their molar conductances therefore show slight increase with increasing dilution. At low conc, the interionic ...
Stoichiometry and the Mole - 2012 Book Archive
... grams in 1 mol of molecules of that compound. Sometimes these masses are called molar masses3 to emphasize the fact that they are the mass for 1 mol of things. (The term molar is the adjective form of mole and has nothing to do with teeth.) Here are some examples. The mass of a hydrogen atom is 1.00 ...
... grams in 1 mol of molecules of that compound. Sometimes these masses are called molar masses3 to emphasize the fact that they are the mass for 1 mol of things. (The term molar is the adjective form of mole and has nothing to do with teeth.) Here are some examples. The mass of a hydrogen atom is 1.00 ...
File - IB CHEM NINJA
... If aqueous thiocyanate ions are added to an aqueous solution of an iron(III) salt, then a blood–red colouration is observed owing to the formation of the complex ion shown. If the concentration of either the thiocyanate ion or the iron(III) ion is increased, then the intensity of the colouration inc ...
... If aqueous thiocyanate ions are added to an aqueous solution of an iron(III) salt, then a blood–red colouration is observed owing to the formation of the complex ion shown. If the concentration of either the thiocyanate ion or the iron(III) ion is increased, then the intensity of the colouration inc ...
Answer Key - mrkelleher
... 6. Gold has a low reactivity and therefore does not corrode over time. 7. In single-displacement reactions, if the activity of the free element is greater than that of the element in the compound, the reaction will take place. 8. Yes; because aluminum is above copper in the activity series, aluminum ...
... 6. Gold has a low reactivity and therefore does not corrode over time. 7. In single-displacement reactions, if the activity of the free element is greater than that of the element in the compound, the reaction will take place. 8. Yes; because aluminum is above copper in the activity series, aluminum ...
Organic and Bio-Molecular Chemistry
... organic compounds, a potentially infinite number of different molecules can be synthesized, by exploiting the great variety of chemical reactions that have been developed since the 21st Century, and progress in the techniques of purification and structural determination. On the other hand, the molec ...
... organic compounds, a potentially infinite number of different molecules can be synthesized, by exploiting the great variety of chemical reactions that have been developed since the 21st Century, and progress in the techniques of purification and structural determination. On the other hand, the molec ...
ION-SELECTIVE ELECTRODES - Clayton State University
... - Elements that are more powerful reducing agents than hydrogen show negative potentials - Elements that are less powerful reducing agents than hydrogen show positive potentials - Metals with more negative Eo are more active - More active metals displace less active metals from solution Fe will disp ...
... - Elements that are more powerful reducing agents than hydrogen show negative potentials - Elements that are less powerful reducing agents than hydrogen show positive potentials - Metals with more negative Eo are more active - More active metals displace less active metals from solution Fe will disp ...
LaBrake, Fundamentals Diagnostic Questions
... d) neutrons e) positrons 18. All of the following are statements from Dalton’s atomic hypothesis, except: a) All the atoms of a given element are identical. b) The atoms of different elements have different masses. c) All atoms are composed of electrons, protons, and neutrons. (correct) d) A compoun ...
... d) neutrons e) positrons 18. All of the following are statements from Dalton’s atomic hypothesis, except: a) All the atoms of a given element are identical. b) The atoms of different elements have different masses. c) All atoms are composed of electrons, protons, and neutrons. (correct) d) A compoun ...
PHOSPHORUS AND SULFUR COSMOCHEMISTRY
... of the document, preparation of a few figures, and provided much needed encouragement. I look forward to taking you to Hawai’i! The author also thanks Hansen’s Soda Company for their creation of the Monster Energy Drink, without which this dissertation would never have been completed. Unleash the Be ...
... of the document, preparation of a few figures, and provided much needed encouragement. I look forward to taking you to Hawai’i! The author also thanks Hansen’s Soda Company for their creation of the Monster Energy Drink, without which this dissertation would never have been completed. Unleash the Be ...
Chapter 22 - 2012 Book Archive
... Group 13 is the first group to span the dividing line between metals and nonmetals, so its chemistry is more diverse than that of groups 1 and 2, which include only metallic elements. Except for the lightest element (boron), the group 13 elements are all relatively electropositive; that is, they ten ...
... Group 13 is the first group to span the dividing line between metals and nonmetals, so its chemistry is more diverse than that of groups 1 and 2, which include only metallic elements. Except for the lightest element (boron), the group 13 elements are all relatively electropositive; that is, they ten ...
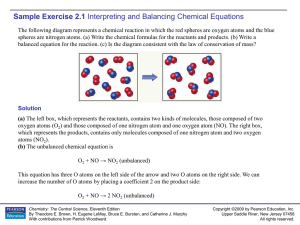
Sample Exercise 2.1
... Write the balanced equation for the reaction that occurs when methanol, CH 3OH(l), is burned in air. Solution When any compound containing C, H, and O is combusted, it reacts with the O 2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) → CO2(g) + H2O(g) In t ...
... Write the balanced equation for the reaction that occurs when methanol, CH 3OH(l), is burned in air. Solution When any compound containing C, H, and O is combusted, it reacts with the O 2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) → CO2(g) + H2O(g) In t ...
Chapter 4 - AP Chemistry with dr hart
... Practice Exercise 2 (4.6) Consider solutions in which 0.1 mol of each of the following compounds is dissolved in 1 L of water: Ca(NO3)2 (calcium nitrate), C6H12O6 (glucose), NaC2H3O2 (sodium acetate), and HC2H3O2 (acetic acid). Rank the solutions in order of increasing electrical conductivity, base ...
... Practice Exercise 2 (4.6) Consider solutions in which 0.1 mol of each of the following compounds is dissolved in 1 L of water: Ca(NO3)2 (calcium nitrate), C6H12O6 (glucose), NaC2H3O2 (sodium acetate), and HC2H3O2 (acetic acid). Rank the solutions in order of increasing electrical conductivity, base ...
GCSE Chemistry Specification Specification for exams from 2014 2014
... followed by Additional Science, or equally by Additional Applied Science. Similarly, Science B could lead to either Additional Science or Additional Applied Science. Our separate science GCSEs have common units with Science A and Additional Science, enabling co-teaching following single, double or t ...
... followed by Additional Science, or equally by Additional Applied Science. Similarly, Science B could lead to either Additional Science or Additional Applied Science. Our separate science GCSEs have common units with Science A and Additional Science, enabling co-teaching following single, double or t ...
Chapter 3 - Educator
... Once we know the formulas of the reactants and products in a reaction, we can write the unbalanced equation. We then balance the equation by determining the coefficients that provide equal numbers of each type of atom on each side of the equation. For most purposes, a balanced equation should contai ...
... Once we know the formulas of the reactants and products in a reaction, we can write the unbalanced equation. We then balance the equation by determining the coefficients that provide equal numbers of each type of atom on each side of the equation. For most purposes, a balanced equation should contai ...
101
... Not all redox reactions give off light, however. How can you recognize a redox reaction, and how can you identify the oxidizing and reducing agents? In section 10.1, you saw net ionic equations with monatomic elements, such as Cu and Zn, and with ions containing a single element, such as Cu2+ and Zn ...
... Not all redox reactions give off light, however. How can you recognize a redox reaction, and how can you identify the oxidizing and reducing agents? In section 10.1, you saw net ionic equations with monatomic elements, such as Cu and Zn, and with ions containing a single element, such as Cu2+ and Zn ...
HYBRID MULTIDENTATE PHOSPHINE
... Stille,9 Suzuki-Miyaura,10 and Sonogashira,11 are well-established methods for the construction of C-C bonds, and widely applied in the synthesis of complex molecules. Over the last few decades considerable large research effort has been placed on the design and synthesis of highly active transition ...
... Stille,9 Suzuki-Miyaura,10 and Sonogashira,11 are well-established methods for the construction of C-C bonds, and widely applied in the synthesis of complex molecules. Over the last few decades considerable large research effort has been placed on the design and synthesis of highly active transition ...
CHEMISTRY CET
... The statement that is NOT correct is 1) Energies of stationary states in hydrogen like atoms is inversely proportional to the square of the principal quantum number. 2) The radius of the first orbit of He is half that of the first orbit of hydrogen atom. 3) Angular quantum number signifies the shape ...
... The statement that is NOT correct is 1) Energies of stationary states in hydrogen like atoms is inversely proportional to the square of the principal quantum number. 2) The radius of the first orbit of He is half that of the first orbit of hydrogen atom. 3) Angular quantum number signifies the shape ...
analytical chemistry - Львівський національний медичний
... Group reactions use for selection from complex (complicated) mixes some substances. Substances with definite properties are united in special analytical groups. This reactions use for: a) detection the present analytical group; b) selection this analytical group from another during systematic path ( ...
... Group reactions use for selection from complex (complicated) mixes some substances. Substances with definite properties are united in special analytical groups. This reactions use for: a) detection the present analytical group; b) selection this analytical group from another during systematic path ( ...
kinetic characterisation of catalysts for methanol synthesis
... vigorous conditions - pressures ranging up to 30 MPa and temperatures up to 400°C (DR Patent, 1923). In order to reduce the costs of process the ICI group developed in the 1960s the low pressure methanol synthesis using a sulphur-free synthesis gas on Cu/ZnO catalyst (Lange, 2001). However, the copp ...
... vigorous conditions - pressures ranging up to 30 MPa and temperatures up to 400°C (DR Patent, 1923). In order to reduce the costs of process the ICI group developed in the 1960s the low pressure methanol synthesis using a sulphur-free synthesis gas on Cu/ZnO catalyst (Lange, 2001). However, the copp ...
Tro Chemistry a Molecular Approach, 3E
... defined in Chapter 6. Perhaps, just as a mechanical system proceeds in the direction of lowest potential energy, so a chemical system might proceed in the direction of lowest enthalpy. If this were the case, all exothermic reactions would be spontaneous and all endothermic reactions would not. Howev ...
... defined in Chapter 6. Perhaps, just as a mechanical system proceeds in the direction of lowest potential energy, so a chemical system might proceed in the direction of lowest enthalpy. If this were the case, all exothermic reactions would be spontaneous and all endothermic reactions would not. Howev ...
Chemical Vapor Deposition (CVD)
... coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different pr ...
... coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different pr ...
Visible Light Photoredox Catalysis with Transition
... Shortly thereafter, Stephenson and co-workers disclosed a photoredox reductive dehalogenation of activated alkyl halides mediated by the same catalyst.13 The combined efforts of these three research groups have helped to initiate a renewed interest in this field, prompting a diversity of studies into ...
... Shortly thereafter, Stephenson and co-workers disclosed a photoredox reductive dehalogenation of activated alkyl halides mediated by the same catalyst.13 The combined efforts of these three research groups have helped to initiate a renewed interest in this field, prompting a diversity of studies into ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... determined using Stuart smp 10 melting point apparatus and are uncorrected. The purity of dyes was determined by thin- layer chromatography (TLC) using silica gel-G coated Al-plates and spots were visualized under uv radiation. IR spectra were recorded on FTIR spectrophotometer Perkin Elmer RX-1 usi ...
... determined using Stuart smp 10 melting point apparatus and are uncorrected. The purity of dyes was determined by thin- layer chromatography (TLC) using silica gel-G coated Al-plates and spots were visualized under uv radiation. IR spectra were recorded on FTIR spectrophotometer Perkin Elmer RX-1 usi ...
- Kendriya Vidyalaya Jamuna Colliery
... 5. In terms of band theory what is the difference between a conductor, an insulator and a semiconductor? The energy gap between the valence band and conduction band in an insulator is very large while in a conductor, the energy gap is very small or there is overlapping between valence band and condu ...
... 5. In terms of band theory what is the difference between a conductor, an insulator and a semiconductor? The energy gap between the valence band and conduction band in an insulator is very large while in a conductor, the energy gap is very small or there is overlapping between valence band and condu ...