The only sure evidence that a chemical reaction has occured is
... ____ is a compound that increases the number of hydrogen ions (H+) when dissolved in water. Which of the following would taste sour? When all the molecules of a compound break apart in water to make hydrogen ions (H+), you have a ____. When few molecules of a compound break apart in water to make hy ...
... ____ is a compound that increases the number of hydrogen ions (H+) when dissolved in water. Which of the following would taste sour? When all the molecules of a compound break apart in water to make hydrogen ions (H+), you have a ____. When few molecules of a compound break apart in water to make hy ...
Types of Chemical Reactions
... of a combination of carbon and hydrogen) to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. This reaction can be expressed as: ...
... of a combination of carbon and hydrogen) to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. This reaction can be expressed as: ...
Ahmed Fazary_Click Chemistry
... combinatorial libraries through heteroatom links…” The Huisgen 1,3-dipolar cycloaddition of azides and alkynes is regarded as the ‘cream of the crop’ of concerted reactions…” Lober, S.; Rodriguez-Loaiza, P.; Gmeiner, P. Org. Lett. 2003, 5, 1753. ...
... combinatorial libraries through heteroatom links…” The Huisgen 1,3-dipolar cycloaddition of azides and alkynes is regarded as the ‘cream of the crop’ of concerted reactions…” Lober, S.; Rodriguez-Loaiza, P.; Gmeiner, P. Org. Lett. 2003, 5, 1753. ...
Test 4
... Molar entropy of fusion The heat energy that must be added to a solid to make it a liquid when it is at its melting temperature. Gibbs Free energy ÄG =ÄH-TÄS; The energy available to do work. entropy favored reaction A reaction where the driving energy term is the increase in entropy. (+ÄSrxn) ...
... Molar entropy of fusion The heat energy that must be added to a solid to make it a liquid when it is at its melting temperature. Gibbs Free energy ÄG =ÄH-TÄS; The energy available to do work. entropy favored reaction A reaction where the driving energy term is the increase in entropy. (+ÄSrxn) ...
Lesson 6
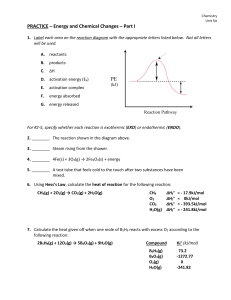
... 4. The substances present before a reaction are called the ________________. 5. The substances present after a reaction are called the _________________. 6. List the three main indicators that a chemical reaction has occurred. 7. Are there any other indicators of a chemical reaction? 8. What evidenc ...
... 4. The substances present before a reaction are called the ________________. 5. The substances present after a reaction are called the _________________. 6. List the three main indicators that a chemical reaction has occurred. 7. Are there any other indicators of a chemical reaction? 8. What evidenc ...
What are reactions? - UTLNET Secure Site
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
Year 9 Homework Task 9E-5 Reactions 5-7
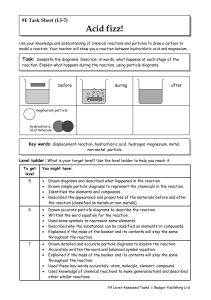
... Use your knowledge and understanding of chemical reactions and particles to draw a cartoon to model a reaction. Your teacher will show you a reaction between hydrochloric acid and magnesium. ...
... Use your knowledge and understanding of chemical reactions and particles to draw a cartoon to model a reaction. Your teacher will show you a reaction between hydrochloric acid and magnesium. ...
Reactions
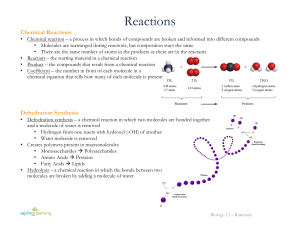
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
A.P. Chemistry Complexation Reactions
... (B) in a compound A single element must be more reactive to replace another element. ...
... (B) in a compound A single element must be more reactive to replace another element. ...