Which scientist developed the quantum mechanical model of the
... What color of visible light has the shortest wavelength? A) B) C) D) ...
... What color of visible light has the shortest wavelength? A) B) C) D) ...
ELECTRONIC STRUCTURE OF ATOMS
... For EMR, the product of the frequency() and the wavelength() equals the speed of light: c = x Wavelength is the distance from node to node. Frequency is the number of wavelengths that pass a given point in a period of time. When solids are heated , they emit radiation i.e. the red glow o ...
... For EMR, the product of the frequency() and the wavelength() equals the speed of light: c = x Wavelength is the distance from node to node. Frequency is the number of wavelengths that pass a given point in a period of time. When solids are heated , they emit radiation i.e. the red glow o ...
Questions and Answers - hrsbstaff.ednet.ns.ca
... with the most and least energy? Explain. 2. Does your stove emit energy when the burner is not turned on? Explain. 3. A single photon is ejected from a light source with a frequency of 2.0 x 1014 Hz. How much energy does it possess? 4. What is the maximum kinetic energy of a photoelectron that has b ...
... with the most and least energy? Explain. 2. Does your stove emit energy when the burner is not turned on? Explain. 3. A single photon is ejected from a light source with a frequency of 2.0 x 1014 Hz. How much energy does it possess? 4. What is the maximum kinetic energy of a photoelectron that has b ...
Honors Midterm Review – 2015-16
... (with any great degree of certainty) both the location and velocity of an electron) _________ responsible for the planetary model of the atom, where electrons traveled in distinct paths around the nucleus _________ responsible for the equation which determines the exact amount of energy needed for e ...
... (with any great degree of certainty) both the location and velocity of an electron) _________ responsible for the planetary model of the atom, where electrons traveled in distinct paths around the nucleus _________ responsible for the equation which determines the exact amount of energy needed for e ...
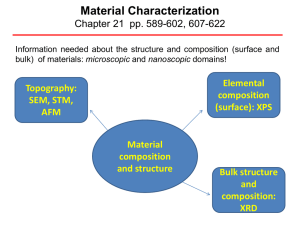
Material Characterization
... Detection limits for most of the elements are in the parts per thousand (ppt) (0.1%) range. Detections limits of parts per million (ppm) (0.0001%) are possible, but require special conditions: concentration at top surface or very long collection time (overnight). XPS is routinely used to analyze ...
... Detection limits for most of the elements are in the parts per thousand (ppt) (0.1%) range. Detections limits of parts per million (ppm) (0.0001%) are possible, but require special conditions: concentration at top surface or very long collection time (overnight). XPS is routinely used to analyze ...
AP Chemistry Chapter 7 Review Packet
... atoms falling from 3p to 3s orbitals. The wavelength of one orange-yellow line in the spectrum of sodium is 589 nm. a. Write the electron configuration for the ground state of sodium. b. Write the electron configuration of the excited state of the sodium atom that is involved in this change in energ ...
... atoms falling from 3p to 3s orbitals. The wavelength of one orange-yellow line in the spectrum of sodium is 589 nm. a. Write the electron configuration for the ground state of sodium. b. Write the electron configuration of the excited state of the sodium atom that is involved in this change in energ ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... Introduction to Geochemistry – Pre-lecture Quiz (1) True or False? (a) The diameter of an atom is less than the diameter of its nucleus. (b) The relative atomic mass of an atom is the mass of an atom relative to an atom of 12C. (c) p-orbitals can contain a maximum of 10 electrons. (d) The first ioni ...
... Introduction to Geochemistry – Pre-lecture Quiz (1) True or False? (a) The diameter of an atom is less than the diameter of its nucleus. (b) The relative atomic mass of an atom is the mass of an atom relative to an atom of 12C. (c) p-orbitals can contain a maximum of 10 electrons. (d) The first ioni ...
Homework 3
... 1. Explain the terms wavelength and amplitude. If a photon has a frequency of 9 1010 Hz what is its wavelength? Which region of the electromagnetic spectrum does this correspond to? ...
... 1. Explain the terms wavelength and amplitude. If a photon has a frequency of 9 1010 Hz what is its wavelength? Which region of the electromagnetic spectrum does this correspond to? ...
The Address of the Electrons
... electron has a set of four numbers, called quantum numbers Each electron has a different set of numbers ¡ No ...
... electron has a set of four numbers, called quantum numbers Each electron has a different set of numbers ¡ No ...
Review Sheet
... Kinetic vs. potential energy Enthalpy (H) Measuring heat (q) Specific heat capacity Endothermic vs. exothermic Stoichiometry using energy (using enthalpy of reaction and balanced chemical reactions) Calculating H using Hess’s Law, Enthalpy Diagrams, and/or H° of formations Calorimetry calculations ...
... Kinetic vs. potential energy Enthalpy (H) Measuring heat (q) Specific heat capacity Endothermic vs. exothermic Stoichiometry using energy (using enthalpy of reaction and balanced chemical reactions) Calculating H using Hess’s Law, Enthalpy Diagrams, and/or H° of formations Calorimetry calculations ...
The end
... b/ One milliwatt of light of wavelength 4,560A is incident on a caesium surface. Calculate the electron current liberated and the minimum stopping voltage necessary to reduce this current to zero. Work function of caesium is 1.93 volts. Assume a quantum efficiency of 0.5 % (that means only 0,5 % of ...
... b/ One milliwatt of light of wavelength 4,560A is incident on a caesium surface. Calculate the electron current liberated and the minimum stopping voltage necessary to reduce this current to zero. Work function of caesium is 1.93 volts. Assume a quantum efficiency of 0.5 % (that means only 0,5 % of ...
HW 8
... Which of the following provided evidence that the electrons in atoms are arranged in distinct energy levels? 1. the results of the Millikan oil-drop experiment 2. the existence of elements with noninteger values for atomic weights 3. the scattering of α particles by a metal foil 4. the deflection of ...
... Which of the following provided evidence that the electrons in atoms are arranged in distinct energy levels? 1. the results of the Millikan oil-drop experiment 2. the existence of elements with noninteger values for atomic weights 3. the scattering of α particles by a metal foil 4. the deflection of ...
X-ray photoelectron spectroscopy

X-ray photoelectron spectroscopy (XPS) is a surface-sensitive quantitative spectroscopic technique that measures the elemental composition at the parts per thousand range, empirical formula, chemical state and electronic state of the elements that exist within a material. XPS spectra are obtained by irradiating a material with a beam of X-rays while simultaneously measuring the kinetic energy and number of electrons that escape from the top 0 to 10 nm of the material being analyzed. XPS requires high vacuum (P ~ 10−8 millibar) or ultra-high vacuum (UHV; P < 10−9 millibar) conditions, although a current area of development is ambient-pressure XPS, in which samples are analyzed at pressures of a few tens of millibar.XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its as-received state, or after some treatment, for example: fracturing, cutting or scraping in air or UHV to expose the bulk chemistry, ion beam etching to clean off some or all of the surface contamination (with mild ion etching) or to intentionally expose deeper layers of the sample (with more extensive ion etching) in depth-profiling XPS, exposure to heat to study the changes due to heating, exposure to reactive gases or solutions, exposure to ion beam implant, exposure to ultraviolet light.XPS is also known as ESCA (Electron Spectroscopy for Chemical Analysis), an abbreviation introduced by Kai Siegbahn's research group to emphasize the chemical (rather than merely elemental) information that the technique provides.In principle XPS detects all elements. In practice, using typical laboratory-scale X-ray sources, XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot easily detect hydrogen (Z = 1) or helium (Z = 2).Detection limits for most of the elements (on a modern instrument) are in the parts per thousand range. Detection limits of parts per million (ppm) are possible, but require special conditions: concentration at top surface or very long collection time (overnight).XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, viscous oils, glues, ion-modified materials and many others.XPS is less routinely used to analyze the hydrated forms of some of the above materials by freezing the samples in their hydrated state in an ultra pure environment, and allowing or causing multilayers of ice to sublime away prior to analysis. Such hydrated XPS analysis allows hydrated sample structures, which may be different from vacuum-dehydrated sample structures, to be studied in their more relevant as-used hydrated structure. Many bio-materials such as hydrogels are examples of such samples.