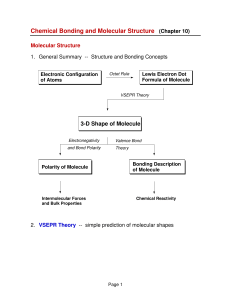
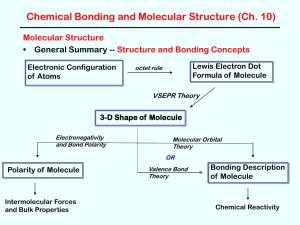
Bond
... A group of atoms held together by covalent bonds is called a molecule. The properties of a molecule, including its role in nature, depends primarily on its molecular structure, or shape. Molecular shape contributes toward determining a compound’s boiling point, freezing point, viscosity, solubility, ...
... A group of atoms held together by covalent bonds is called a molecule. The properties of a molecule, including its role in nature, depends primarily on its molecular structure, or shape. Molecular shape contributes toward determining a compound’s boiling point, freezing point, viscosity, solubility, ...
3-D Shape of Molecule
... 2. Molecular Orbitals for simple diatomic molecules (H2 and He2) in H2 the 1s atomic orbitals on the two H atoms are combined into: a bonding MO -- σ1s and an antibonding MO -- σ*1s MO energy level diagram for H2 (only the bonding MO is filled): ...
... 2. Molecular Orbitals for simple diatomic molecules (H2 and He2) in H2 the 1s atomic orbitals on the two H atoms are combined into: a bonding MO -- σ1s and an antibonding MO -- σ*1s MO energy level diagram for H2 (only the bonding MO is filled): ...
4 - College of Arts and Sciences
... The fat stored in the hump of a camel is a source of both energy and water. Calculate the grams produced by metabolism of 5.599 kg of the fat (C57H110O6) stored in the hump ...
... The fat stored in the hump of a camel is a source of both energy and water. Calculate the grams produced by metabolism of 5.599 kg of the fat (C57H110O6) stored in the hump ...
+ 2 HCL(aq) CaCl2(aq) + H2O(l) + CO2(g)
... KEY Vocabulary Cation: An ion with a positive charge Anion: An ion with a negative charge Covalent Bond: A bond between two non-metals where a pair of electrons are shared. Ionic Bond: A bond between a non-metal and a metal where electrons are lost or gained. Subscript: A number that represents how ...
... KEY Vocabulary Cation: An ion with a positive charge Anion: An ion with a negative charge Covalent Bond: A bond between two non-metals where a pair of electrons are shared. Ionic Bond: A bond between a non-metal and a metal where electrons are lost or gained. Subscript: A number that represents how ...
Chemistry: The Basics
... • The 1st orbital of an atom (closest to the nucleus) can hold 2 electrons • The 2nd orbital of an atom can hold up to 8 ...
... • The 1st orbital of an atom (closest to the nucleus) can hold 2 electrons • The 2nd orbital of an atom can hold up to 8 ...
Chemistry Standards Review
... 37. In the reaction, 2 Mg + O2 2 MgO, if 100.0 g of magnesium reacts with 50.0 g of oxygen, what mass of product is produced? Gases and Their Properties 38. What is the kinetic molecular theory? 39. How do gases create pressure, use KMT to support your answer. 40. Explain diffusion, use KMT to sup ...
... 37. In the reaction, 2 Mg + O2 2 MgO, if 100.0 g of magnesium reacts with 50.0 g of oxygen, what mass of product is produced? Gases and Their Properties 38. What is the kinetic molecular theory? 39. How do gases create pressure, use KMT to support your answer. 40. Explain diffusion, use KMT to sup ...
Chapter1011
... • Polar or Non-Polar? – In very symmetrical structures (e.g. CO2 or CF4), the individual bond dipoles effectively cancel each other and the molecule is ...
... • Polar or Non-Polar? – In very symmetrical structures (e.g. CO2 or CF4), the individual bond dipoles effectively cancel each other and the molecule is ...
ATOMIC MASS
... isotopes (nuclear decay) Energy (ability to do work); energy levels (electron states of potential energy) ...
... isotopes (nuclear decay) Energy (ability to do work); energy levels (electron states of potential energy) ...
Molecular Geometry Lab
... d. How many hybrid orbitals are nonbonding in SnCl2? 3. Answer the following question that relate to VSEPR theory: a. How many nonbonding electron pairs are on N in NH3? b. How many nonbonding electron pairs are on O in H2O? 4. Answer the following question that relate to polarity: a. Are the Sn-Cl ...
... d. How many hybrid orbitals are nonbonding in SnCl2? 3. Answer the following question that relate to VSEPR theory: a. How many nonbonding electron pairs are on N in NH3? b. How many nonbonding electron pairs are on O in H2O? 4. Answer the following question that relate to polarity: a. Are the Sn-Cl ...
atoms-chemical
... • Atoms with incomplete valence shells interact by either sharing or transferring valence electrons. • These interactions typically result in the atoms remaining close together, held by an attractions called chemical bonds. ...
... • Atoms with incomplete valence shells interact by either sharing or transferring valence electrons. • These interactions typically result in the atoms remaining close together, held by an attractions called chemical bonds. ...
Basic Chemistry - Biology with Radjewski
... undergo chemical reactions to fill their outer shells. • They can attain stability by sharing electrons with other atoms (covalent bond) or by losing or gaining electrons (ionic bond) • The atoms are then bonded together into molecules. • Octet rule—atoms with at least two electron shells form stabl ...
... undergo chemical reactions to fill their outer shells. • They can attain stability by sharing electrons with other atoms (covalent bond) or by losing or gaining electrons (ionic bond) • The atoms are then bonded together into molecules. • Octet rule—atoms with at least two electron shells form stabl ...
Intro Biochemistry/Ecology
... Because they have the same number of protons, all isotopes of an element have the same chemical properties. The main types of chemical bonds are covalent bonds and ionic bonds. Section 2-2: Properties of Water A water molecule is polar, because there is an uneven distribution of electrons between th ...
... Because they have the same number of protons, all isotopes of an element have the same chemical properties. The main types of chemical bonds are covalent bonds and ionic bonds. Section 2-2: Properties of Water A water molecule is polar, because there is an uneven distribution of electrons between th ...
Chemistry Fall Final Review 2012-2013 Alchemy Unit
... 1. Using the periodic table, where are the metals and nonmetals? What is hydrogen? Metals are in the left side of the periodic table. Nonmetals are on the right side of the periodic table. Hydrogen is an nonmetal. 2. Where are the alkali, alkaline earth, transition metals, halogens, and noble gases? ...
... 1. Using the periodic table, where are the metals and nonmetals? What is hydrogen? Metals are in the left side of the periodic table. Nonmetals are on the right side of the periodic table. Hydrogen is an nonmetal. 2. Where are the alkali, alkaline earth, transition metals, halogens, and noble gases? ...
2 Types of Chemical Bonds
... 1. Ionic Bond – gain or lose valence electrons • This is a chemical bond formed by the attraction between positive (+) and negative (-) ions. What types of elements form Ionic Bonds? Metal elements: • Lose valence electrons to form (+) ions • Easier to lose than gain to get 8 Non Metal elements: • G ...
... 1. Ionic Bond – gain or lose valence electrons • This is a chemical bond formed by the attraction between positive (+) and negative (-) ions. What types of elements form Ionic Bonds? Metal elements: • Lose valence electrons to form (+) ions • Easier to lose than gain to get 8 Non Metal elements: • G ...
Mid-Term OR Study Guide
... dots, stars, different color dots, etc.), and put loops around shared electron pairs. (A) Is the bond type between a phosphorus atom and a fluorine atom ionic, polar covalent, or nonpolar covalent? Show any relevant calculations/reasoning that you use to determine this. (B) Write the two possible Le ...
... dots, stars, different color dots, etc.), and put loops around shared electron pairs. (A) Is the bond type between a phosphorus atom and a fluorine atom ionic, polar covalent, or nonpolar covalent? Show any relevant calculations/reasoning that you use to determine this. (B) Write the two possible Le ...
CHEM 1A: VSEPR Theory Practice Visualizing Shapes
... CHEM 1A: VSEPR Theory Now that we have an understanding of covalent bonding and how atoms share electrons to form molecules and polyatomic ions, we will use Lewis dot structures to predict electronic and molecular geometries. In turn, the geometries of the molecules will influence many different phy ...
... CHEM 1A: VSEPR Theory Now that we have an understanding of covalent bonding and how atoms share electrons to form molecules and polyatomic ions, we will use Lewis dot structures to predict electronic and molecular geometries. In turn, the geometries of the molecules will influence many different phy ...
The Chemistry of Biology
... • Electrons fill the orbitals and shells in pairs starting with the shell nearest the nucleus • Each element, then, has a unique pattern of orbitals and shells ...
... • Electrons fill the orbitals and shells in pairs starting with the shell nearest the nucleus • Each element, then, has a unique pattern of orbitals and shells ...
Chemical Bonding Quiz
... Study Guide: Chemical Bonding Quiz Students should be able to understand and apply the following Chemical Bonding concepts: ...
... Study Guide: Chemical Bonding Quiz Students should be able to understand and apply the following Chemical Bonding concepts: ...
Chapter 9: Molecular Geometry and Bonding Theories
... Chapter 9: Molecular Geometry and Bonding Theories ...
... Chapter 9: Molecular Geometry and Bonding Theories ...
TEST REVIEW S Valence Electrons TEST REVIEW SHEET 2017
... NOTE: If an element has <4 valence electrons it will give them away during an ionic bond and become a positive ion. If >4, it will take them and become a negative ion For the most part…. metals will give away their valence electrons and nonmetals will take enough valence electrons to fill their oute ...
... NOTE: If an element has <4 valence electrons it will give them away during an ionic bond and become a positive ion. If >4, it will take them and become a negative ion For the most part…. metals will give away their valence electrons and nonmetals will take enough valence electrons to fill their oute ...
Ch. 2 The Chemical Basis of Life
... electrons, atoms interact in order to have eight electrons in their outermost energy level (valence shell) Stable (unreactive) elements have a full valence shell Unstable (reactive) elements lack a full valence shell and will gain, lose, or share electrons with other atoms to achieve ...
... electrons, atoms interact in order to have eight electrons in their outermost energy level (valence shell) Stable (unreactive) elements have a full valence shell Unstable (reactive) elements lack a full valence shell and will gain, lose, or share electrons with other atoms to achieve ...