Final Exam - Seattle Central College
... CHEM 162: Final Exam Study Guide Chapter 16: Chemical Equilibrium equilibrium: state where the forward and reverse reactions or processes occur at the same rate – Know that concentrations are not changing at equilibrium, but they need not be equal to one another. – Be able to indicate when equilibri ...
... CHEM 162: Final Exam Study Guide Chapter 16: Chemical Equilibrium equilibrium: state where the forward and reverse reactions or processes occur at the same rate – Know that concentrations are not changing at equilibrium, but they need not be equal to one another. – Be able to indicate when equilibri ...
2011-2012 ACAD REVIEW SHEET Chapter 16
... law of mass action equilibrium constant equilibrium expression law of chemical equilibrium ...
... law of mass action equilibrium constant equilibrium expression law of chemical equilibrium ...
syllabus for screening test (mcq type)
... Nature of liquid state, surface tension, capillary rise, spreading of liquid over other surface, temperature dependence of surface tension. Measurement of surface tension, viscosity of liquids, origin of viscosity of gases and liquids, determination of viscosity coefficient, Poiseuille’s equation, t ...
... Nature of liquid state, surface tension, capillary rise, spreading of liquid over other surface, temperature dependence of surface tension. Measurement of surface tension, viscosity of liquids, origin of viscosity of gases and liquids, determination of viscosity coefficient, Poiseuille’s equation, t ...
Document
... solely on the state of the system. Once the state is set, all the state functions will have a definite value. And the state function difference between two different states only depends on the initial and final state of a process. ...
... solely on the state of the system. Once the state is set, all the state functions will have a definite value. And the state function difference between two different states only depends on the initial and final state of a process. ...
Review Unit 8 Test (Chp 15,17)
... stays constant before and after equilibrium is reached. A is half true, but “decreases”, decreases to become a constant nonzero rate at equilibrium. not “increases.” decreases to become zero at equilibrium. Greater pressure of reactant initially (Q = 0/1.00 = 0) so forward rate is faster due to grea ...
... stays constant before and after equilibrium is reached. A is half true, but “decreases”, decreases to become a constant nonzero rate at equilibrium. not “increases.” decreases to become zero at equilibrium. Greater pressure of reactant initially (Q = 0/1.00 = 0) so forward rate is faster due to grea ...
A Thumbnail Review of Regents Chemistry
... COMPACT REGENTS REVIEW 2010-2011 In SATURATED solutions, the RATES of dissolving and crystallizing are EQUAL LECHATELIER’S PRINCIPLE: a system that loses equilibrium due to stress will shift to re-establish equilibrium STRESSES: change in temperature / change in concentration / change in pressure C ...
... COMPACT REGENTS REVIEW 2010-2011 In SATURATED solutions, the RATES of dissolving and crystallizing are EQUAL LECHATELIER’S PRINCIPLE: a system that loses equilibrium due to stress will shift to re-establish equilibrium STRESSES: change in temperature / change in concentration / change in pressure C ...
Chemistry 434 - St. Francis Xavier University
... sysG < 0 - spontaneous process sysG > 0 - non-spontaneous process (note that this process would be spontaneous in the reverse direction) sysG = 0 - system is in equilibrium ...
... sysG < 0 - spontaneous process sysG > 0 - non-spontaneous process (note that this process would be spontaneous in the reverse direction) sysG = 0 - system is in equilibrium ...
Chemical equilibrium, redox and pE
... e.g. Fe (II) to Fe (III) - increase in charge on charged species e.g. Co2+ to Co3+ Reduction is the reverse Oxidation-reduction reactions occur together One component is oxidised, the other is reduced Component that is reduced is the oxidising agent Component that is oxidised is the reducing agent ...
... e.g. Fe (II) to Fe (III) - increase in charge on charged species e.g. Co2+ to Co3+ Reduction is the reverse Oxidation-reduction reactions occur together One component is oxidised, the other is reduced Component that is reduced is the oxidising agent Component that is oxidised is the reducing agent ...
Chemical Reactions & Balancing Equations
... This equation is already balanced – same number of atoms of each type of element on each side What if it isn’t balanced already? ...
... This equation is already balanced – same number of atoms of each type of element on each side What if it isn’t balanced already? ...
2014-15 FINAL REVIEW Nomenclature: Chemical Name Chemical
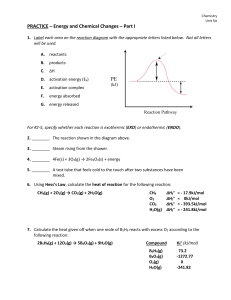
... 4. What final temperature will 120.00 g of benzene at 7.00oC have after it absorbs 2.20 kJ of heat? The Cp of benzene is 1.74 J/goC. Equilibrium: 1. List 3 stressors that would shift this reaction to the left: ...
... 4. What final temperature will 120.00 g of benzene at 7.00oC have after it absorbs 2.20 kJ of heat? The Cp of benzene is 1.74 J/goC. Equilibrium: 1. List 3 stressors that would shift this reaction to the left: ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.