Signs of Reaction - Calderglen High School
... Substances made of two or more different types of atom joined together. Calderglen High School ...
... Substances made of two or more different types of atom joined together. Calderglen High School ...
Photoremovable Protecting Groups
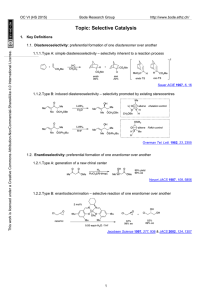
... Based on these observations Corrie and Trentham4 re-examined the photochemistry of several substituted benzoin phosphates. They found that the 3′,5′-dimethoxybenzoin cage was best suited among those investigated for the release of Pi. The formation of 3′,5′-dimethoxy-2-phenylbenzofuran (11) occurred ...
... Based on these observations Corrie and Trentham4 re-examined the photochemistry of several substituted benzoin phosphates. They found that the 3′,5′-dimethoxybenzoin cage was best suited among those investigated for the release of Pi. The formation of 3′,5′-dimethoxy-2-phenylbenzofuran (11) occurred ...
PowerPoint 演示文稿
... in his early work, to test the theory. Studies of aliphatic substitutions and eliminations, often with his long-time collaborator E. D. Hughes, led to I ncorporation into the standard language of chemistry of such words as nucleophile, electrophile, inductive and mesomeric (resonance) effects, and s ...
... in his early work, to test the theory. Studies of aliphatic substitutions and eliminations, often with his long-time collaborator E. D. Hughes, led to I ncorporation into the standard language of chemistry of such words as nucleophile, electrophile, inductive and mesomeric (resonance) effects, and s ...
Penny Commons - Chemistry Education Association
... The pracs, exercises and demonstrations are all found in old editions of Pearson Heinemann TRAB or in the Student Workbook. For the present Study Design they are provided as pdfs with support materials etc for Lab technicians at pearsonplaces.com.au SW refer to the Heinemann Student Workbook 2 – ...
... The pracs, exercises and demonstrations are all found in old editions of Pearson Heinemann TRAB or in the Student Workbook. For the present Study Design they are provided as pdfs with support materials etc for Lab technicians at pearsonplaces.com.au SW refer to the Heinemann Student Workbook 2 – ...
3.Redox
... resulting saturated solution is 1.17 g / mL. Calculate (a) the mole fraction of KCl in the solution, (b) the Molarity of the solution. 10. Suppose a 8.75 M aqueous CH3OH solution has a density of 0.789 g / mL. Calculate the mole fraction of CH3OH in the solution.. 11. A solution prepared by dissolv ...
... resulting saturated solution is 1.17 g / mL. Calculate (a) the mole fraction of KCl in the solution, (b) the Molarity of the solution. 10. Suppose a 8.75 M aqueous CH3OH solution has a density of 0.789 g / mL. Calculate the mole fraction of CH3OH in the solution.. 11. A solution prepared by dissolv ...
Equilibrium Booklet - mrstorie
... a) What is the effect on [HI] if a small amount of H2 is added? b) What is the effect on [HI] if the pressure of the system is increased? c) What is the effect on [HI] if the temperature is increased? d) What is the effect on [HI] if a catalyst is added? 4. For the reaction: CO(g) + 2 H2(g) CH3OH ...
... a) What is the effect on [HI] if a small amount of H2 is added? b) What is the effect on [HI] if the pressure of the system is increased? c) What is the effect on [HI] if the temperature is increased? d) What is the effect on [HI] if a catalyst is added? 4. For the reaction: CO(g) + 2 H2(g) CH3OH ...
Chapter 22 and 23 Study Guide
... You do not have to do this assignment. You will not receive points for doing it. You should practice these questions until you can do them WITHOUT your book, notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions ar ...
... You do not have to do this assignment. You will not receive points for doing it. You should practice these questions until you can do them WITHOUT your book, notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions ar ...
Unit 5: Chemical Kinetics and Equilibrium
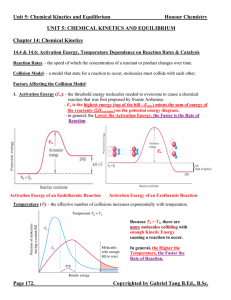
... Equilibrium Position: - the concentrations or pressures of all chemical species at equilibrium state. - depends strongly on the Initial Concentrations of the chemical species. (In contrast, K does NOT depend on initial concentrations, only on temperature and the specific reaction.) - since there all ...
... Equilibrium Position: - the concentrations or pressures of all chemical species at equilibrium state. - depends strongly on the Initial Concentrations of the chemical species. (In contrast, K does NOT depend on initial concentrations, only on temperature and the specific reaction.) - since there all ...
Packet 1 - Kentucky Community and Technical College System
... Step 3 Use the solubility rules to decide whether a solid forms and, if so, to predict the identity of the solid. CHE 170 Packet 4 - 50 ...
... Step 3 Use the solubility rules to decide whether a solid forms and, if so, to predict the identity of the solid. CHE 170 Packet 4 - 50 ...
lecture 6 oxidative addition
... OXIDATIVE ADDITION – SN2 Mechanism (non-concerted) • In all oxidative additions, a pair of electrons from the metal is used to break the A−B bond in the reagent. • In the SN2 pathway, adopted for polarized A‐B substrates such as alkyl halides, the metal electron pair of LnM directly attacks the A–B ...
... OXIDATIVE ADDITION – SN2 Mechanism (non-concerted) • In all oxidative additions, a pair of electrons from the metal is used to break the A−B bond in the reagent. • In the SN2 pathway, adopted for polarized A‐B substrates such as alkyl halides, the metal electron pair of LnM directly attacks the A–B ...