Equilibrium - Cobb Learning
... Strike a match and it erupts instantaneously. Coal made from dead plants takes millions of years ...
... Strike a match and it erupts instantaneously. Coal made from dead plants takes millions of years ...
Ch 5.1 The Nature of Chemical Reactions
... • Understand parts to a chemical equation (reactants, products, yeild sign, double arrow) • Conservation of matter is expressed through balancing chemical equations • Describe difference between endothermic and exothermic reactions ...
... • Understand parts to a chemical equation (reactants, products, yeild sign, double arrow) • Conservation of matter is expressed through balancing chemical equations • Describe difference between endothermic and exothermic reactions ...
South Pasadena • AP Chemistry
... The chemical elements are the building blocks of matter, which can be understood in terms of arrangements of atoms. Molecules & elements Chemical analysis The mole Electron configuration Periodicity Quantum mechanical model Atomic models Mass spectrometry Light & matter Conservat ...
... The chemical elements are the building blocks of matter, which can be understood in terms of arrangements of atoms. Molecules & elements Chemical analysis The mole Electron configuration Periodicity Quantum mechanical model Atomic models Mass spectrometry Light & matter Conservat ...
CHM 101
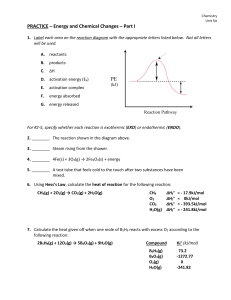
... The reactants in a chemical change have 487 kJ of energy. The change they undergo has a H = -157 kJ. The activation energy for the reaction is 570 kJ. a. Draw the energy vs reaction progress graph on the axes above paying attention to all values. Label a point that represents all products and one t ...
... The reactants in a chemical change have 487 kJ of energy. The change they undergo has a H = -157 kJ. The activation energy for the reaction is 570 kJ. a. Draw the energy vs reaction progress graph on the axes above paying attention to all values. Label a point that represents all products and one t ...
The following list of topics for an AP Chemistry course is intended to
... of vaporization and fusion; calorimetry 3. Second law; entropy; free energy of formation; free energy of reaction; dependence of change in free energy on enthalpy and entropy changes 4. Relationship of change in free energy to equilibrium constants and electrode potentials ...
... of vaporization and fusion; calorimetry 3. Second law; entropy; free energy of formation; free energy of reaction; dependence of change in free energy on enthalpy and entropy changes 4. Relationship of change in free energy to equilibrium constants and electrode potentials ...
AP Chemistry
... Descriptive and experimental laboratory exercises are frequently assigned to provide the maximum opportunity for students to learn a variety of skills and those facts, principles, and concepts of chemistry covered in lectures, reading, and discussion. In addition, the laboratory exercises are used t ...
... Descriptive and experimental laboratory exercises are frequently assigned to provide the maximum opportunity for students to learn a variety of skills and those facts, principles, and concepts of chemistry covered in lectures, reading, and discussion. In addition, the laboratory exercises are used t ...
Project Details PPT
... oP R E V E N T W A S T E B Y U S I N G A SOLVENTLESS PROCESS. oM I N I M I Z E A M O U N T S O F S O L V E N T S A N D REAGENTS USED. ...
... oP R E V E N T W A S T E B Y U S I N G A SOLVENTLESS PROCESS. oM I N I M I Z E A M O U N T S O F S O L V E N T S A N D REAGENTS USED. ...
Microbial Metabolism
... Modes of E Conservation-ATP • Fermentation: in which redox reaction ocurs WITHOUT a terminal electron acceptor (couple oxiation with subsequent reduction of an organic ...
... Modes of E Conservation-ATP • Fermentation: in which redox reaction ocurs WITHOUT a terminal electron acceptor (couple oxiation with subsequent reduction of an organic ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
... detach the loosest electron from atoms of that element. (e) All alkali metals have similar chemical properties. (f) Alkali earths have one electron in the outer shell. (g) Electronegativity is the amount of negative charge on an atom. (h) Ca has a valency of 2. (i) True ionic bonds are unknown and a ...
(s) If 5.00 moles of zinc is placed into 1.50 L... 34. solution,what is the mass of the hydrogen gas produced?
... Zn (s) + 2 HCI (aq) ~ Znc~ (s) +Hz(g) If 5.00 moles of zinc is placed into 1.50 L of a 3.00MHCI 34. solution,what is the mass of the hydrogen gas produced? (A) 0.750 g (D) 5.00 g (B) 2.25 g (E) 10.0 g (C) 4.50 g 32. How many grams of Zinc (atomic mass 65.0 g) are required to react completelywith 1.0 ...
... Zn (s) + 2 HCI (aq) ~ Znc~ (s) +Hz(g) If 5.00 moles of zinc is placed into 1.50 L of a 3.00MHCI 34. solution,what is the mass of the hydrogen gas produced? (A) 0.750 g (D) 5.00 g (B) 2.25 g (E) 10.0 g (C) 4.50 g 32. How many grams of Zinc (atomic mass 65.0 g) are required to react completelywith 1.0 ...
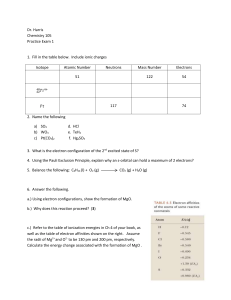
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 18. Refer to the activity series in chapter 10. For the single replacement reactions below, write the half reactions. Label the reducing and oxidizing agents. Show the net ionic equation. If no reaction occurs, write “no reaction”. a.) Titanium (IV) perchlorate (aq) + Lithium (s) b.) Magnesium (s) ...
... 18. Refer to the activity series in chapter 10. For the single replacement reactions below, write the half reactions. Label the reducing and oxidizing agents. Show the net ionic equation. If no reaction occurs, write “no reaction”. a.) Titanium (IV) perchlorate (aq) + Lithium (s) b.) Magnesium (s) ...
CHM 101
... How many grams of water will be produced if the reaction is conducted with 75.6 g of C5H12O and 95.5 g of oxygen? ...
... How many grams of water will be produced if the reaction is conducted with 75.6 g of C5H12O and 95.5 g of oxygen? ...