Unit 7 Reaction Rates and Equilibrium Notes
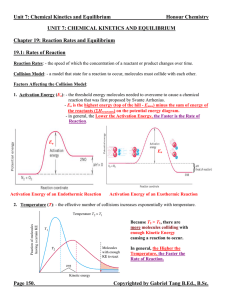
... Le Châtelier’s Principle: - a qualitative method to predict the shift on an equilibrium system if it is disturbed by means of changing concentration, pressure and temperature. - the equilibrium will shift in the direction that minimizes the change imposed on the system. 1. Effects of a Change in Con ...
... Le Châtelier’s Principle: - a qualitative method to predict the shift on an equilibrium system if it is disturbed by means of changing concentration, pressure and temperature. - the equilibrium will shift in the direction that minimizes the change imposed on the system. 1. Effects of a Change in Con ...
5 pancakes 2 eggs
... 15. 1 molecule of tetraphosphorus decoxide contains: how many moles? How many grams? How many P atoms? How many O atoms? How many total atoms? 26. A 7.52 g sample of ajoene (garlic odor) was found to contain 3.09 g S, 0.453 g H, 0.513 g O and the rest, C. Calculate the percent composition. 39. Ethan ...
... 15. 1 molecule of tetraphosphorus decoxide contains: how many moles? How many grams? How many P atoms? How many O atoms? How many total atoms? 26. A 7.52 g sample of ajoene (garlic odor) was found to contain 3.09 g S, 0.453 g H, 0.513 g O and the rest, C. Calculate the percent composition. 39. Ethan ...
The Mole - Bakersfield College
... 23.72% sulfur, and 47.35% oxygen. The experimental molar mass of 270.0 g/mol. What are the empirical and molecular formulas of potassium persulfate? To find the molecular formula: Calculate the MM of KSO4 = (1 mol K x 39.10 g/mol K) + (1 mol S x 32.07 g/mol S) + (4 mol O x 16.00 g/mol O) = 135.17 g/ ...
... 23.72% sulfur, and 47.35% oxygen. The experimental molar mass of 270.0 g/mol. What are the empirical and molecular formulas of potassium persulfate? To find the molecular formula: Calculate the MM of KSO4 = (1 mol K x 39.10 g/mol K) + (1 mol S x 32.07 g/mol S) + (4 mol O x 16.00 g/mol O) = 135.17 g/ ...
13- and 14-membered macrocyclic ligands containing
... The spectroscopic equilibrium measurements were carried out using 10.00 mL of the ligand solutions. Following each addition of titrant, the pH was measured and a sample of solution was placed in a 5 mm NMR tube adapted with an internal capillary tube containing D2O for locking and H3PO4 for referenc ...
... The spectroscopic equilibrium measurements were carried out using 10.00 mL of the ligand solutions. Following each addition of titrant, the pH was measured and a sample of solution was placed in a 5 mm NMR tube adapted with an internal capillary tube containing D2O for locking and H3PO4 for referenc ...
C H A P T E R
... 1. Count out exactly 200 small beads. Using a stopwatch, record the amount of time it takes you to count them. 2. Your teacher will tell you the approximate number of small beads in 1 g. Knowing that number, calculate the mass of 200 small beads. Record the mass that you have calculated. 3. Use a ba ...
... 1. Count out exactly 200 small beads. Using a stopwatch, record the amount of time it takes you to count them. 2. Your teacher will tell you the approximate number of small beads in 1 g. Knowing that number, calculate the mass of 200 small beads. Record the mass that you have calculated. 3. Use a ba ...
SQA CfE Higher Chemistry Unit 3: Chemistry in society
... appropriate. Chemical calculations • Balanced equations show the mole ratio(s) of reactants and products. Using the balanced equation and the gram formula masses (GFM), mass to mass calculations can be performed. • The quantity of a reactant or product can also be expressed in terms of moles. The co ...
... appropriate. Chemical calculations • Balanced equations show the mole ratio(s) of reactants and products. Using the balanced equation and the gram formula masses (GFM), mass to mass calculations can be performed. • The quantity of a reactant or product can also be expressed in terms of moles. The co ...
Basic Stoichometry
... In chemistry, reactions proceed with very specific recipes. The study of these recipes is stoichiometry. When the reactants are present in the correct amounts, the reaction will produce products. What happens if there are more or less of some of the reactants present? Vocabulary: Before you begin, p ...
... In chemistry, reactions proceed with very specific recipes. The study of these recipes is stoichiometry. When the reactants are present in the correct amounts, the reaction will produce products. What happens if there are more or less of some of the reactants present? Vocabulary: Before you begin, p ...
Chemistry 12 Worksheet 2-3 Calculations Involving the
... a) In an equilibrium mixture the following concentrations were found: [A] = 0.45M, [B] = 0.63M and [C] = 0.30M. Calculate the value of the equilibrium constant for this reaction. Answer ____________________ b) At the same temperature, another equilibrium mixture is analyzed and it is found that [B] ...
... a) In an equilibrium mixture the following concentrations were found: [A] = 0.45M, [B] = 0.63M and [C] = 0.30M. Calculate the value of the equilibrium constant for this reaction. Answer ____________________ b) At the same temperature, another equilibrium mixture is analyzed and it is found that [B] ...
Honors Chemistry
... 4a. Use %composition to calculate the empirical formula for a compound given the elements present. 4b. Use the molar mass of the compound and the empirical formula to determine the molecular formula. ...
... 4a. Use %composition to calculate the empirical formula for a compound given the elements present. 4b. Use the molar mass of the compound and the empirical formula to determine the molecular formula. ...
Unit 10 complete 2016-2017
... 4a. Use %composition to calculate the empirical formula for a compound given the elements present. 4b. Use the molar mass of the compound and the empirical formula to determine the molecular formula. ...
... 4a. Use %composition to calculate the empirical formula for a compound given the elements present. 4b. Use the molar mass of the compound and the empirical formula to determine the molecular formula. ...
Solubility
... [CrO42-] = 0.01M, [Ag+] = 1.0x10-5 (calculated previously) Ksp = [Ag+][Br-] = (1.0x10-5M)[Br-] = 5.0x10-13 [Br-] = 5.0x10-8M So if we stop the addition of AgNO3 just before Ag2CrO4 starts to precipitate... [Br-] drops from 0.01M to 5.0x10-8M. What % of Br- is left? 5.0x10-8M / 1.0x10-2M *100% ...
... [CrO42-] = 0.01M, [Ag+] = 1.0x10-5 (calculated previously) Ksp = [Ag+][Br-] = (1.0x10-5M)[Br-] = 5.0x10-13 [Br-] = 5.0x10-8M So if we stop the addition of AgNO3 just before Ag2CrO4 starts to precipitate... [Br-] drops from 0.01M to 5.0x10-8M. What % of Br- is left? 5.0x10-8M / 1.0x10-2M *100% ...
THE DIFFUSION MECHANISM OF HYDROCARBONS IN... Jirong Xiao B.S., East China Institute of Chemical Technology
... interplay between the guest molecule and host lattice, and is mainly responsible for the different diffusivities of various compounds in ZSM-5. For some molecules in 5.A, the intracrystalline partitioning is important. The diffusion coefficients of benzene, toluene, and 2-methylbutane in ZSM-5 are ...
... interplay between the guest molecule and host lattice, and is mainly responsible for the different diffusivities of various compounds in ZSM-5. For some molecules in 5.A, the intracrystalline partitioning is important. The diffusion coefficients of benzene, toluene, and 2-methylbutane in ZSM-5 are ...
syllabus for two‐year four‐semester course in chemistry 2014
... Spectrophotometric, ion exchange and complexometric estimations. Identification of single organic liquid with one or more functional groups. Numerical, kinetic and equilibrium experiments. ...
... Spectrophotometric, ion exchange and complexometric estimations. Identification of single organic liquid with one or more functional groups. Numerical, kinetic and equilibrium experiments. ...
M - coercingmolecules
... Chang, R. 2002. Chemistry 7th ed. Singapore: McGraw-Hill. Silberberg, M. 2010. Principles of General Chemistry. 2nd ed. New York: McGraw-Hill. ...
... Chang, R. 2002. Chemistry 7th ed. Singapore: McGraw-Hill. Silberberg, M. 2010. Principles of General Chemistry. 2nd ed. New York: McGraw-Hill. ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
Chapter 3 Stoichiometry: Calculations with Chemical
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
Calculations with Chemical Formulas and Equations
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... • By definition, this is the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
K c
... experimental conditions may disturb the balance and shift the equilibrium position so that more or less of the desired product is formed. • In this section we will study 5 factor which can effect chemical equilibrium namely : concentration, pressure, volume, temperature, and catalyst. • Le Châtelier ...
... experimental conditions may disturb the balance and shift the equilibrium position so that more or less of the desired product is formed. • In this section we will study 5 factor which can effect chemical equilibrium namely : concentration, pressure, volume, temperature, and catalyst. • Le Châtelier ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... analyzed through combustion in a chamber like this – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been ...
... analyzed through combustion in a chamber like this – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been ...
Sample Exercise 15.1 Writing Equilibrium
... Plan For equilibrium to be achieved, it must be possible for both the forward process and the reverse process to occur. For the forward process to occur, there must be some calcium carbonate present. For the reverse process to occur, there must be both calcium oxide and carbon dioxide. In both cases ...
... Plan For equilibrium to be achieved, it must be possible for both the forward process and the reverse process to occur. For the forward process to occur, there must be some calcium carbonate present. For the reverse process to occur, there must be both calcium oxide and carbon dioxide. In both cases ...
Interaction of the Adenine-Thymine Watson
... four-body, etc. contributions. However, we do not see any practical reason to do so for our purposes. According to the perturbational theory of molecular interactions,14b the ∆E3 term includes first-order exchange and induction (SCF-deformation) nonadditivities at the SCF level. The induction term i ...
... four-body, etc. contributions. However, we do not see any practical reason to do so for our purposes. According to the perturbational theory of molecular interactions,14b the ∆E3 term includes first-order exchange and induction (SCF-deformation) nonadditivities at the SCF level. The induction term i ...
Synthesis and thermal decarbonylation of W(CO)5 complexes
... the random phosphazene copolymers {[NP(O2C12H8)]0.5[NP(O–C6H4–CO2Prn)(O–C6H4–L)]0.5}n [L = CN (1), PPh2 (2)]. A similar sequential substitution but using 4-hydroxypyridine in the third step afforded the polymer {[NP(O2C12H8)]0.6[NP(O–C6H4–CO2Prn)(O–C5H4N)]0.4}n (3) (Scheme 1). The average Mw (of the ...
... the random phosphazene copolymers {[NP(O2C12H8)]0.5[NP(O–C6H4–CO2Prn)(O–C6H4–L)]0.5}n [L = CN (1), PPh2 (2)]. A similar sequential substitution but using 4-hydroxypyridine in the third step afforded the polymer {[NP(O2C12H8)]0.6[NP(O–C6H4–CO2Prn)(O–C5H4N)]0.4}n (3) (Scheme 1). The average Mw (of the ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.