Chapter
... • two general types of bonding between atoms found in compounds, ionic and covalent • ionic bonds result when electrons have been transferred between atoms, resulting in oppositely charged ions that attract each other – generally found when metal atoms bonded to nonmetal atoms ...
... • two general types of bonding between atoms found in compounds, ionic and covalent • ionic bonds result when electrons have been transferred between atoms, resulting in oppositely charged ions that attract each other – generally found when metal atoms bonded to nonmetal atoms ...
Amine‐Directed Hydrogen‐Bonded Two‐Dimensional
... first choice in manufacturing sensors,[10] electronics[9] or switches at the single-molecule level.[5, 11–14] One possible strategy can be employing pH, due to applications in designing experiments in aqueous solutions under potential control,[15–18] as a knob to manipulate intermolecular hydrogen b ...
... first choice in manufacturing sensors,[10] electronics[9] or switches at the single-molecule level.[5, 11–14] One possible strategy can be employing pH, due to applications in designing experiments in aqueous solutions under potential control,[15–18] as a knob to manipulate intermolecular hydrogen b ...
M.Sc. Part-I Chemistry - North Maharashtra University
... Infrared spectroscopy: simple harmonic oscillator, vibrational spectra of diatomic molecules, anharmonic oscillator, diatomic vibrating rotator, vibration-rotation spectrum of diatomic molecule applying Born-Oppenheimer approximation, vibration-rotation spectra of linear polyatomic molecules and sym ...
... Infrared spectroscopy: simple harmonic oscillator, vibrational spectra of diatomic molecules, anharmonic oscillator, diatomic vibrating rotator, vibration-rotation spectrum of diatomic molecule applying Born-Oppenheimer approximation, vibration-rotation spectra of linear polyatomic molecules and sym ...
[HMIM][Br9]: a Room-temperature Ionic Liquid Based on a
... the S30 SevenEasy instrument from Mettler Toledo using a platinum electrode InLab710 with a cell constant of 0.8096 cm−1 in a cell filled with 3.5 mL of the sample. NMR data were recorded at room temperature on a Bruker DPX 200 spectrometer using the software T OPSPIN 1.3. The samples of the investi ...
... the S30 SevenEasy instrument from Mettler Toledo using a platinum electrode InLab710 with a cell constant of 0.8096 cm−1 in a cell filled with 3.5 mL of the sample. NMR data were recorded at room temperature on a Bruker DPX 200 spectrometer using the software T OPSPIN 1.3. The samples of the investi ...
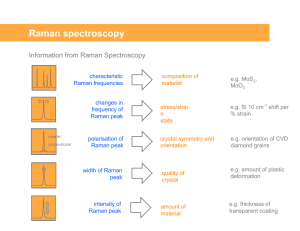
Raman spectroscopy
... 2. Dark Charge rate: How long can you integrate before the binned CCD pixels generate a charge equivalent to the read noise? At the integration time that the dark charge signal contributes to the noise either through shot noise or uniformity of response, it must be subtracted. 3. Uniformity of respo ...
... 2. Dark Charge rate: How long can you integrate before the binned CCD pixels generate a charge equivalent to the read noise? At the integration time that the dark charge signal contributes to the noise either through shot noise or uniformity of response, it must be subtracted. 3. Uniformity of respo ...
Calculations with Chemical Formulas and Equations
... – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been Stoichiometry determined ...
... – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been Stoichiometry determined ...
Mathematical Skills Handbook
... more reliable and powerful than using formula triangles, which may be misremembered and can only be used for formulae that comprise three variables. A good understanding of units developed early on in the course will also stand learners in good stead for determining units of rate and equilibrium con ...
... more reliable and powerful than using formula triangles, which may be misremembered and can only be used for formulae that comprise three variables. A good understanding of units developed early on in the course will also stand learners in good stead for determining units of rate and equilibrium con ...
J. Am. Chem. SOC. 1993,115, 7685-7695
... A similar reaction occurs with the symmetric arene 2,6dimethoxynaphthalene. The single product shows bound 72resonances at 6 3.023 (td, J = 7.6, 1.9 Hz) and 3.664 (td, J = 7.8, 2.4 Hz) and a 31Pdoublet at 6 0.50 ( J = 201 Hz), as well as two distinct methoxy resonances. The formation of the 3,4-q2-2 ...
... A similar reaction occurs with the symmetric arene 2,6dimethoxynaphthalene. The single product shows bound 72resonances at 6 3.023 (td, J = 7.6, 1.9 Hz) and 3.664 (td, J = 7.8, 2.4 Hz) and a 31Pdoublet at 6 0.50 ( J = 201 Hz), as well as two distinct methoxy resonances. The formation of the 3,4-q2-2 ...
electrical energy and capacitance
... Example 1. A compound is discovered with a 58.12 g/mol molar mass. Its empirical formula is C2H5. What is the molecular formula of this compound? 1A. (1) C = 12.01 amu (2) H = 1.01 amu (3) C2 + H5 (4) C2H5 = 2(12.01 amu) + 5(1.01 amu) (5) EF = C2H5 = 29.07 g/mol (6) MF = 58.12 g/mol (7) MF = n(EF) ( ...
... Example 1. A compound is discovered with a 58.12 g/mol molar mass. Its empirical formula is C2H5. What is the molecular formula of this compound? 1A. (1) C = 12.01 amu (2) H = 1.01 amu (3) C2 + H5 (4) C2H5 = 2(12.01 amu) + 5(1.01 amu) (5) EF = C2H5 = 29.07 g/mol (6) MF = 58.12 g/mol (7) MF = n(EF) ( ...
O 2 - Montville.net
... 1. What are the reactants in this chemical equation? 2. What are the products in this chemical equation? 3. Are there the same number of atoms on both sides of the equation? a. Where any atoms destroyed or created? b. Was the Law of Conservation of Matter maintained? ...
... 1. What are the reactants in this chemical equation? 2. What are the products in this chemical equation? 3. Are there the same number of atoms on both sides of the equation? a. Where any atoms destroyed or created? b. Was the Law of Conservation of Matter maintained? ...
Stoichiometry - Taylor County Schools
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
Practice Test_final_161_F2015
... 12. Which of the following conclusions regarding Rutherford’s gold foil experiment is not consistent with the observations? A) The nucleus occupies only a small portion of the space of an atom. B) Most alpha particles travel straight through the gold foil. C) The nucleus occupies a large amount of t ...
... 12. Which of the following conclusions regarding Rutherford’s gold foil experiment is not consistent with the observations? A) The nucleus occupies only a small portion of the space of an atom. B) Most alpha particles travel straight through the gold foil. C) The nucleus occupies a large amount of t ...
Stoichiometry
... – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been determined Stoichiometry ...
... – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been determined Stoichiometry ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
What is equilibrium?
... is different from the moles of gaseous products. • If the number of moles is the same on both sides of the balanced equation, changes in pressure and volume have no effect on the equilibrium. ...
... is different from the moles of gaseous products. • If the number of moles is the same on both sides of the balanced equation, changes in pressure and volume have no effect on the equilibrium. ...
B.Sc. (Hons.) CHEMISTRY THREE-YEARS FULL
... enthalpy, H, relation between heat capacities, calculations of q, w, U and H for reversible, irreversible and free expansion of gases (ideal and van der Waals) under isothermal and adiabatic conditions. Unit-II Thermochemistry-II : Heats of reactions: standard states; enthalpy of formation of molecu ...
... enthalpy, H, relation between heat capacities, calculations of q, w, U and H for reversible, irreversible and free expansion of gases (ideal and van der Waals) under isothermal and adiabatic conditions. Unit-II Thermochemistry-II : Heats of reactions: standard states; enthalpy of formation of molecu ...
Phase behavior of clathrate hydrates: a model for single and
... beyond the 9rst shell or cage (Klauda & Sandler, 2002). To include the e)ect of lattice distortion by guests, the water locations for sI guests were obtained from X-ray di)raction measurements of methane (Gutt et al., 2000), carbon dioxide (Ikeda et al., 2000), and ethylene oxide (McMullen & Je)rey, ...
... beyond the 9rst shell or cage (Klauda & Sandler, 2002). To include the e)ect of lattice distortion by guests, the water locations for sI guests were obtained from X-ray di)raction measurements of methane (Gutt et al., 2000), carbon dioxide (Ikeda et al., 2000), and ethylene oxide (McMullen & Je)rey, ...
quantitative_chemistry
... In addition to being naturally present in the body, adrenaline is administered as a drug to stimulate the heart, to alleviate allergic reactions, and even to help break up fat cells during liposuction. As you might expect, control over the amounts administered is vital. To make, use, or detect speci ...
... In addition to being naturally present in the body, adrenaline is administered as a drug to stimulate the heart, to alleviate allergic reactions, and even to help break up fat cells during liposuction. As you might expect, control over the amounts administered is vital. To make, use, or detect speci ...
equilibrium - TeacherWeb
... The density of a pure liquid or solid is a constant at any given temperature. CO2(g) + H2(g) ↔ CO(g) + H2O(l) ...
... The density of a pure liquid or solid is a constant at any given temperature. CO2(g) + H2(g) ↔ CO(g) + H2O(l) ...
Peter Ertl - American Chemical Society
... be classified in some way. For applications in drug design, a classification based on substituent physicochemical properties is more useful than a simple classification based on structural similarity. In this study, we used substituent size, hydrophobicity, electronic properties (donating/accepting ...
... be classified in some way. For applications in drug design, a classification based on substituent physicochemical properties is more useful than a simple classification based on structural similarity. In this study, we used substituent size, hydrophobicity, electronic properties (donating/accepting ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
... Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical ...
supplementary information
... studies have been limited to Au, Ag and Cu substrates because these free-electron-like metals have appropriate values of the dielectric constant to fully support the effective electron oscillation, also known as the localized surface plasmon resonance (LSPR), which generates a large electromagnetic ...
... studies have been limited to Au, Ag and Cu substrates because these free-electron-like metals have appropriate values of the dielectric constant to fully support the effective electron oscillation, also known as the localized surface plasmon resonance (LSPR), which generates a large electromagnetic ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.


![[HMIM][Br9]: a Room-temperature Ionic Liquid Based on a](http://s1.studyres.com/store/data/016911324_1-ac5688316a1e3a6c1ba364df016e5832-300x300.png)