
Ch 3 Student.pptx
... • Molecular compounds form between two nonmetals – The atoms in molecular compounds don’t form ions – they share electrons. For this reason the same combination of elements can form a number of different molecular compounds. ...
... • Molecular compounds form between two nonmetals – The atoms in molecular compounds don’t form ions – they share electrons. For this reason the same combination of elements can form a number of different molecular compounds. ...
Chapter 3: Calculations with Chemical Formulas
... • Stoichiometry: Relates the moles of products and reactants to each other and to measurable quantities involved in a chemical reaction. ...
... • Stoichiometry: Relates the moles of products and reactants to each other and to measurable quantities involved in a chemical reaction. ...
Core organic chemistry
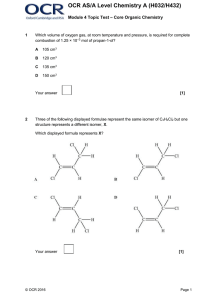
... Either name OR structure should be correct for two compounds. AND Attempts to explain formation of one type of isomer. The information about isomerism is basic and communicated in an unstructured way. The relationship to the compounds drawn/named may not be clear. ...
... Either name OR structure should be correct for two compounds. AND Attempts to explain formation of one type of isomer. The information about isomerism is basic and communicated in an unstructured way. The relationship to the compounds drawn/named may not be clear. ...
chapter_14 Equilibr
... 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium ...
... 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium ...
Avogadro`s Number, Moles and Molar Mass
... feasible. The volume that this many atoms or molecules would occupy is negligible. Reasonable masses of elements for scientists to work with are those that are based on the average atomic mass for an element. It was determined by the Italian scientist, Avogadro, that the number of atoms in exactly 1 ...
... feasible. The volume that this many atoms or molecules would occupy is negligible. Reasonable masses of elements for scientists to work with are those that are based on the average atomic mass for an element. It was determined by the Italian scientist, Avogadro, that the number of atoms in exactly 1 ...
CO 2 - TrimbleChemistry
... • Molecular compounds - molecules containing atoms from two or more different elements • Covalent bonds - the force holding the atoms together in a molecular compound by the sharing of electrons ...
... • Molecular compounds - molecules containing atoms from two or more different elements • Covalent bonds - the force holding the atoms together in a molecular compound by the sharing of electrons ...
Mole
... Mole Ratio In a balanced equation, the ration between the numbers of moles of any two substances. ...
... Mole Ratio In a balanced equation, the ration between the numbers of moles of any two substances. ...
Solubility and Solubility Equilibrium
... Solubility and Solubility Equilibrium: Student Review Notes Now, equilibrium is this idea of reversibility in chemical reactions. It might seem kind of oxymoronic to talk about equilibrium for insoluble species, but we can talk about equilibrium of insoluble molecules because, to some small extent, ...
... Solubility and Solubility Equilibrium: Student Review Notes Now, equilibrium is this idea of reversibility in chemical reactions. It might seem kind of oxymoronic to talk about equilibrium for insoluble species, but we can talk about equilibrium of insoluble molecules because, to some small extent, ...
Moles Class Packet Unit 2
... The empirical formula of a compound is the simplest whole-number ratio of atoms of the elements in a compound. It may be different from the molecular formula, which is the actual ratio of atoms in a molecule of that compound. (3.3d) In all chemical reactions there is a conservation of mass, energy, ...
... The empirical formula of a compound is the simplest whole-number ratio of atoms of the elements in a compound. It may be different from the molecular formula, which is the actual ratio of atoms in a molecule of that compound. (3.3d) In all chemical reactions there is a conservation of mass, energy, ...
Document
... 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium ...
... 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium ...
chap15pptlecture_chapte.ppt [Read-Only]
... 2NO2 (g) 2NO (g) + O2 (g) is 158 at 1000K. What is the equilibrium pressure of O2 if the PNO2 = 0.400 atm and PNO = 0.270 atm? ...
... 2NO2 (g) 2NO (g) + O2 (g) is 158 at 1000K. What is the equilibrium pressure of O2 if the PNO2 = 0.400 atm and PNO = 0.270 atm? ...
Chapter
... Types of Formula Structural Formula • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess a ...
... Types of Formula Structural Formula • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess a ...
Chapter 11 Chemical Calculations
... Divide the g/mol fo reach element by the atomic mass 36.00 g/mol C /12 g/atom = 3 atoms/mole = C3 6 g/molH / 1g/atom H = 6 atoms/mol = H6 Molecular formula = C3H6 Key calculation Given % composition and molar mass be able to determine the molecular formula. This can be done by two different procedur ...
... Divide the g/mol fo reach element by the atomic mass 36.00 g/mol C /12 g/atom = 3 atoms/mole = C3 6 g/molH / 1g/atom H = 6 atoms/mol = H6 Molecular formula = C3H6 Key calculation Given % composition and molar mass be able to determine the molecular formula. This can be done by two different procedur ...
Electronic structure of molecular van der Waals complexes with
... to describe the tunneling through various flat-lying alkyl derivatives on HOPG under typically used experimental conditions.4–16 Electrons from a metallic STM-tip tunnel through a molecule into the conducting substrate, or vice versa. A schematic energy diagram is displayed in Fig. 1. The workfuncti ...
... to describe the tunneling through various flat-lying alkyl derivatives on HOPG under typically used experimental conditions.4–16 Electrons from a metallic STM-tip tunnel through a molecule into the conducting substrate, or vice versa. A schematic energy diagram is displayed in Fig. 1. The workfuncti ...
Enthalpy - Net Texts
... the energy taken by the vaporizing molecules in order for the temperature to remain constant. This heat required to vaporize the liquid is called enthalpy, or often, heat of vaporization. For the vaporization of one mole of water the enthalpy is given as: ΔH = 44.0 kJ at 298K When a solid melts, the ...
... the energy taken by the vaporizing molecules in order for the temperature to remain constant. This heat required to vaporize the liquid is called enthalpy, or often, heat of vaporization. For the vaporization of one mole of water the enthalpy is given as: ΔH = 44.0 kJ at 298K When a solid melts, the ...
File
... How many molecules of NaCl are there if you have 25 grams of NaCl? If 12.02 grams of Ni(CO)4 yielded 2.53 g Ni when you performed the experiment, determine the percent yield. ◦ Ni(CO)4 (g) ...
... How many molecules of NaCl are there if you have 25 grams of NaCl? If 12.02 grams of Ni(CO)4 yielded 2.53 g Ni when you performed the experiment, determine the percent yield. ◦ Ni(CO)4 (g) ...
Writing Equilibrium Cons... and Liquids - Chemwiki
... We are going to look at a general case with the equation: ...
... We are going to look at a general case with the equation: ...
Introduction to X-ray Absorption Spectroscopy, Extended X
... Ø Monochromation by diffraction of a set of parallel Si crystals, following Bragg’s law n l = 2 d sin q Ø Wavelength l selected by varying angle q of Si crystal with respect to beam Ø Higher order reflections (n > 1) are rejected by making the crystals slightly non-parallel Ø Transmitted intensity I ...
... Ø Monochromation by diffraction of a set of parallel Si crystals, following Bragg’s law n l = 2 d sin q Ø Wavelength l selected by varying angle q of Si crystal with respect to beam Ø Higher order reflections (n > 1) are rejected by making the crystals slightly non-parallel Ø Transmitted intensity I ...
Dynamic Multi-Component Covalent Assembly for the Binding of
... structure 4 because the 3Å MS scavenge water, thereby driving the equilibrium to alcohol incorporation. Therefore, the proposed sequence involves compound 2 formed from 2-PA, DPA, and Zn(OTf)2, dissociating from Zn(II), followed by water elimination catalyzed by the BrØnsted acid to give iminium 3, ...
... structure 4 because the 3Å MS scavenge water, thereby driving the equilibrium to alcohol incorporation. Therefore, the proposed sequence involves compound 2 formed from 2-PA, DPA, and Zn(OTf)2, dissociating from Zn(II), followed by water elimination catalyzed by the BrØnsted acid to give iminium 3, ...
Bioorganic chemistry-a scientific endeavour in continuous
... realized only recently that our research interests do not end with organic chemistry -- structural, synthetic, or kinetic -- but that they now involve biological material. Structure guides us as to the potential details of how active biological partners may interact. Synthesis provides us with compo ...
... realized only recently that our research interests do not end with organic chemistry -- structural, synthetic, or kinetic -- but that they now involve biological material. Structure guides us as to the potential details of how active biological partners may interact. Synthesis provides us with compo ...
Photogeneration of Hydride Donors and Their Use Toward CO2
... Na2S2O4 in D2O did not afford any stereospecific products in the deuterization at C8 of pbn. The stereoselective formation of Δ(S)-[1•DD]2+ and Λ-(R)-[1•DD]2+, which are enantiomers with the same 1H NMR spectra, clearly Photoproduct of [1]2+ in H2O (a) and (c); in D2O (b) and (d). H8 is located abov ...
... Na2S2O4 in D2O did not afford any stereospecific products in the deuterization at C8 of pbn. The stereoselective formation of Δ(S)-[1•DD]2+ and Λ-(R)-[1•DD]2+, which are enantiomers with the same 1H NMR spectra, clearly Photoproduct of [1]2+ in H2O (a) and (c); in D2O (b) and (d). H8 is located abov ...
2 CHEMICAL ARITHMATICS W MODULE - 1
... In your previous classes, you have studied how to write chemical formula of a sustance. For example, water is represented by H2O, carbon dioxide is represented by CO2, methane is represented by CH4, dinitrogen penta oxide is represented by N2O5, and so on. You are aware, formula for a molecule uses ...
... In your previous classes, you have studied how to write chemical formula of a sustance. For example, water is represented by H2O, carbon dioxide is represented by CO2, methane is represented by CH4, dinitrogen penta oxide is represented by N2O5, and so on. You are aware, formula for a molecule uses ...
Molecular Dynamics Simulation of the
... AMBER does not contain parameters for the sulfate fragment of taurine, the necessary values are adopted from Gaff (Table S1).23 The atomic charges of the BS needed to calculate the electrostatic contribution to the energy are derived by applying the RESP procedure24,25 where the charges are fit to the ...
... AMBER does not contain parameters for the sulfate fragment of taurine, the necessary values are adopted from Gaff (Table S1).23 The atomic charges of the BS needed to calculate the electrostatic contribution to the energy are derived by applying the RESP procedure24,25 where the charges are fit to the ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.









![chap15pptlecture_chapte.ppt [Read-Only]](http://s1.studyres.com/store/data/015369082_1-00cbf06a2d468a4ae1c963f5ca674e31-300x300.png)












