Solubility and Complex-ion Equilibria
... determine whether precipitation will occur. • One form of kidney stones is calcium phosphate, Ca3(PO4)2, which has a Ksp of 1.0 × 10−26. A sample of urine contains 1.0 × 10−3 M Ca2+ and 1.0 × 10−8 M PO43− ion. • Calculate Qc and predict whether Ca3(PO4)2 will precipitate. ...
... determine whether precipitation will occur. • One form of kidney stones is calcium phosphate, Ca3(PO4)2, which has a Ksp of 1.0 × 10−26. A sample of urine contains 1.0 × 10−3 M Ca2+ and 1.0 × 10−8 M PO43− ion. • Calculate Qc and predict whether Ca3(PO4)2 will precipitate. ...
First-Principles Theoretical Study of Molecular HCl Adsorption on a
... it is not known whether HCl exists in an ionic or molecular form at (or near) the ice surface. Spectroscopic,24-26 and other,27 evidence indicates an ionic form of HCl at low temperatures (less than 180 K) and partial pressures where ionic hydrates of HCl are known to be thermodynamically stable. Ho ...
... it is not known whether HCl exists in an ionic or molecular form at (or near) the ice surface. Spectroscopic,24-26 and other,27 evidence indicates an ionic form of HCl at low temperatures (less than 180 K) and partial pressures where ionic hydrates of HCl are known to be thermodynamically stable. Ho ...
Stoichiometry and the Mole - 2012 Book Archive
... Although the number of things in a mole is known to eight decimal places, it is usually fine to use only two or three decimal places in calculations. The numerical value of things in a mole is often called Avogadro’s number (NA), which is also known as the Avogadro constant, after Amadeo Avogadro, a ...
... Although the number of things in a mole is known to eight decimal places, it is usually fine to use only two or three decimal places in calculations. The numerical value of things in a mole is often called Avogadro’s number (NA), which is also known as the Avogadro constant, after Amadeo Avogadro, a ...
Document
... Suppose you want to ‘whip’ a batch of hydrogen iodide, following the balanced chemical equation: ...
... Suppose you want to ‘whip’ a batch of hydrogen iodide, following the balanced chemical equation: ...
CYPRUS
... Course lectures are in Greek and the students must take a final exam for each course. The final grade results from a combination of homework grades, intermediate (mid-term) exams, literature projects or laboratory reports. There are no prerequisite courses, but in a series of related courses (e.g., ...
... Course lectures are in Greek and the students must take a final exam for each course. The final grade results from a combination of homework grades, intermediate (mid-term) exams, literature projects or laboratory reports. There are no prerequisite courses, but in a series of related courses (e.g., ...
Stoichiometry and the Mole
... Curiously, this chemical reaction question is very similar to the pound cake question. Both of them involve relating a quantity of one substance to a quantity of another substance or substances. The relating of one chemical substance to another using a balanced chemical reaction is called stoichiome ...
... Curiously, this chemical reaction question is very similar to the pound cake question. Both of them involve relating a quantity of one substance to a quantity of another substance or substances. The relating of one chemical substance to another using a balanced chemical reaction is called stoichiome ...
Fulltext: english,
... far as processes involving 1:1 electrolytes are concerned, transfer Gibbs energies provide invaluable information regarding the differences in solvation of these electrolytes from one solvent to another.62−66 Even more relevant are data for single-ion values.67,68 Although these are based on an extr ...
... far as processes involving 1:1 electrolytes are concerned, transfer Gibbs energies provide invaluable information regarding the differences in solvation of these electrolytes from one solvent to another.62−66 Even more relevant are data for single-ion values.67,68 Although these are based on an extr ...
Basic Stoichometry
... In reality, reactants don't have to react in perfect whole-numbers of moles. In a two-reactant synthesis reaction, usually one reactant gets entirely used up (and determines how much product is made), even if that means using fractions of a mole of reactant. For instance, when solid, metallic alumin ...
... In reality, reactants don't have to react in perfect whole-numbers of moles. In a two-reactant synthesis reaction, usually one reactant gets entirely used up (and determines how much product is made), even if that means using fractions of a mole of reactant. For instance, when solid, metallic alumin ...
Beverley John C. Beverley IE 500/PHI 598: Ontological Engineering
... As indicated above, Thermodynamics is the study of energy, but more specifically, it is the study of energy inhering in a Thermodynamic System. Such systems will be formally defined below in the Classes section, but the intuition underlying the concept is easily grasped: they are arbitrary regions o ...
... As indicated above, Thermodynamics is the study of energy, but more specifically, it is the study of energy inhering in a Thermodynamic System. Such systems will be formally defined below in the Classes section, but the intuition underlying the concept is easily grasped: they are arbitrary regions o ...
"Fundamentals of Rotation--Vibration Spectra" in
... potential Veff (R, l) is degenerate with excited states arising from lower values of l (l − 1, l − 2, . . . 0), leading to the well-known “Coulomb degeneracy” (slightly lifted in reality). For diatomic molecules, no such degeneracy arises because V (R) is qualitatively very different from the Coulom ...
... potential Veff (R, l) is degenerate with excited states arising from lower values of l (l − 1, l − 2, . . . 0), leading to the well-known “Coulomb degeneracy” (slightly lifted in reality). For diatomic molecules, no such degeneracy arises because V (R) is qualitatively very different from the Coulom ...
Noncovalent interactions of molecules with single walled carbon
... energy.25,26 However, addition of an atom to the interior of the sidewall would add strain energy to the nanotube. Theoretical modelling has shown that the interior of a SWNT is more inert than the exterior of a nanotube to the addition of atomic nitrogen,27 atomic carbon,28 atomic fluorine,26 atomi ...
... energy.25,26 However, addition of an atom to the interior of the sidewall would add strain energy to the nanotube. Theoretical modelling has shown that the interior of a SWNT is more inert than the exterior of a nanotube to the addition of atomic nitrogen,27 atomic carbon,28 atomic fluorine,26 atomi ...
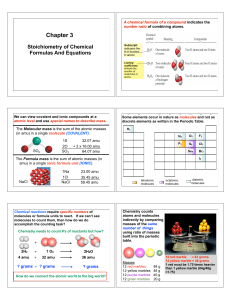
Chapter 3 2014
... Al2(SO4)3 2 atoms of Al and 3 molecules of (SO4)2- = 1 formula unit Al2(SO4)3 2 moles of Al and 3 moles of (SO4)2- = 1 formula unit Al2(SO4)3 1 formula unit Al2(SO4)3 = 342.17 amu Al2(SO4)3 1 mole Al2(SO4)3 = 342.17 g Al2(SO4)3 1 mole Al2(SO4)3 = 6.022 x 1023 formula units Al2(SO4)3 1 mole Al2(SO4)3 ...
... Al2(SO4)3 2 atoms of Al and 3 molecules of (SO4)2- = 1 formula unit Al2(SO4)3 2 moles of Al and 3 moles of (SO4)2- = 1 formula unit Al2(SO4)3 1 formula unit Al2(SO4)3 = 342.17 amu Al2(SO4)3 1 mole Al2(SO4)3 = 342.17 g Al2(SO4)3 1 mole Al2(SO4)3 = 6.022 x 1023 formula units Al2(SO4)3 1 mole Al2(SO4)3 ...
PCCPwww
... reported up to now refer to over-exchanged samples4 prepared via conventional ion exchange with aqueous solutions of cupric precursors. As a result of this procedure, samples containing mixtures of copper ions in different aggregation and oxidation states are usually obtained. In order to avoid the h ...
... reported up to now refer to over-exchanged samples4 prepared via conventional ion exchange with aqueous solutions of cupric precursors. As a result of this procedure, samples containing mixtures of copper ions in different aggregation and oxidation states are usually obtained. In order to avoid the h ...
Interaction of the C-terminal peptide from pigeon cytochrome C with
... Results on the application of SERS in peptides have been published by PodstawkaProniewicz et al. [18]; L-valine phosphonate dipeptides were studied by Fourier-transform infrared (FT-IR) spectroscopy, Fourier-transform Raman spectroscopy (FT-RS) and SERS. The band assignment was performed on the basi ...
... Results on the application of SERS in peptides have been published by PodstawkaProniewicz et al. [18]; L-valine phosphonate dipeptides were studied by Fourier-transform infrared (FT-IR) spectroscopy, Fourier-transform Raman spectroscopy (FT-RS) and SERS. The band assignment was performed on the basi ...
Cyclam ``capa` POT.4` to ``capa` POT.3` denticity change
... form NO carrier systems with an additional possibility of targeting improvement by the judicious choice of the biomolecule (an antibody, for instance). With this aim, our first results were achieved with trans-[RuCl(L)(1-(3-propylammonium)cyclam)]n+ (L ) Cl-, H2O (tfms ) trifluoromethanesulfonate))2 ...
... form NO carrier systems with an additional possibility of targeting improvement by the judicious choice of the biomolecule (an antibody, for instance). With this aim, our first results were achieved with trans-[RuCl(L)(1-(3-propylammonium)cyclam)]n+ (L ) Cl-, H2O (tfms ) trifluoromethanesulfonate))2 ...
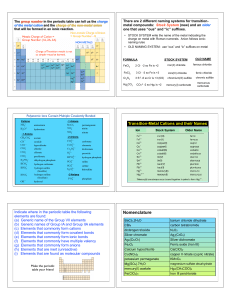
Chapter 3 Sem 2 2013-14
... reduced whole-number ratio of the atoms in a compound. A molecular formula shows the whole number ratio of an actual known molecule. ...
... reduced whole-number ratio of the atoms in a compound. A molecular formula shows the whole number ratio of an actual known molecule. ...
Metal Complexes Containing Natural and Artificial Radioactive
... DMSO-solvated thorium(IV) ion is nine-coordinate in both solution and the solid state with average Th-O bond lengths of 2.45 Å. On the contrary, the dmso-solvated lanthanoid(III) ions are eight-coordinate. Actinide carboxylates have been extremely widely studied. Thus, the results on the optical abs ...
... DMSO-solvated thorium(IV) ion is nine-coordinate in both solution and the solid state with average Th-O bond lengths of 2.45 Å. On the contrary, the dmso-solvated lanthanoid(III) ions are eight-coordinate. Actinide carboxylates have been extremely widely studied. Thus, the results on the optical abs ...
Stoichiometry
... If you have 100 g of X2Y there would be 60g of X and 40g of Y For XY3 since it has only one X atom you can think of that as ½(60g) or 30 g of X and since there’s three Y atoms you can think of that as 3(40g) or 120g of Y. So in XY3 there’s 30g of X for every 120g of Y – so %Y = (120g/150g)100 = 80% ...
... If you have 100 g of X2Y there would be 60g of X and 40g of Y For XY3 since it has only one X atom you can think of that as ½(60g) or 30 g of X and since there’s three Y atoms you can think of that as 3(40g) or 120g of Y. So in XY3 there’s 30g of X for every 120g of Y – so %Y = (120g/150g)100 = 80% ...
Chapter 3 2013
... 2. The red numbers are the number of moles of atoms in lactic acid. This is what we use in the formula C3.33 H6.66 O3.33 C3.33 H6.66 O3.33 ...
... 2. The red numbers are the number of moles of atoms in lactic acid. This is what we use in the formula C3.33 H6.66 O3.33 C3.33 H6.66 O3.33 ...
PowerPoint Presentation - Chemical Equilibrium
... The relatively small value for K indicates that, compared to 1.0, X would not be large enough to include in the calculation. The equilibrium expression could be simplified to K = X2/1.0. A quick test of this hypothesis could be made by using the 4.2 10–3 value as X and checking the right side of t ...
... The relatively small value for K indicates that, compared to 1.0, X would not be large enough to include in the calculation. The equilibrium expression could be simplified to K = X2/1.0. A quick test of this hypothesis could be made by using the 4.2 10–3 value as X and checking the right side of t ...
CHEMISTRY
... Introduces the student to current analytical methods and to cultivate sound experimental technique. Laboratory work includes ion exchange separations, complexometric and potentiometric acid-base titrations, and absorption spectrophotometry. Corequisite: CHEM 0250. ...
... Introduces the student to current analytical methods and to cultivate sound experimental technique. Laboratory work includes ion exchange separations, complexometric and potentiometric acid-base titrations, and absorption spectrophotometry. Corequisite: CHEM 0250. ...
Equilibrium Expression (Keq)
... Ex: Write Keq expression for: NaCl(s) + H2SO4(l) ↔ HCl(g) + NaHSO4(s) Keq = ...
... Ex: Write Keq expression for: NaCl(s) + H2SO4(l) ↔ HCl(g) + NaHSO4(s) Keq = ...
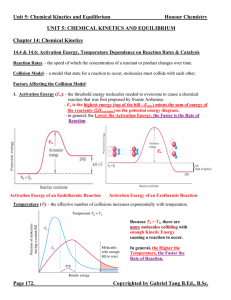
Unit 5: Chemical Kinetics and Equilibrium
... 1. When K >> 1, the equilibrium system favours the products. There are more products than reactants at the state of equilibrium. ([C]eq and [D]eq or PC, eq and PD, eq >> [A]eq and [B]eq or PA, eq and PB, eq) 2. When K << 1, the equilibrium system favours the reactants. There are less products than r ...
... 1. When K >> 1, the equilibrium system favours the products. There are more products than reactants at the state of equilibrium. ([C]eq and [D]eq or PC, eq and PD, eq >> [A]eq and [B]eq or PA, eq and PB, eq) 2. When K << 1, the equilibrium system favours the reactants. There are less products than r ...
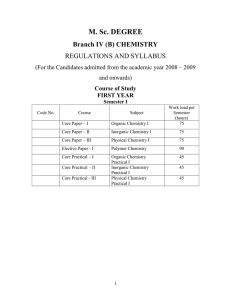
M.Sc. Chemistry - Periyar University
... PHYSICAL CHEMISTRY – I (75 Hours) Unit – I Classical Thermodynamics – I (15 Hours) Maxwell’s relations and thermodynamic equations of state – applications in the evaluation of Cp – Cv for solids and for vanderwaals gases, Cp – Cv in terms of coefficient of expansion and coefficient of compressibilit ...
... PHYSICAL CHEMISTRY – I (75 Hours) Unit – I Classical Thermodynamics – I (15 Hours) Maxwell’s relations and thermodynamic equations of state – applications in the evaluation of Cp – Cv for solids and for vanderwaals gases, Cp – Cv in terms of coefficient of expansion and coefficient of compressibilit ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.