What do you know about light?
... • On its own the atomic number does not tell us the number of neutrons in an element. In order to determine the number of neutrons we need the mass number. ...
... • On its own the atomic number does not tell us the number of neutrons in an element. In order to determine the number of neutrons we need the mass number. ...
File
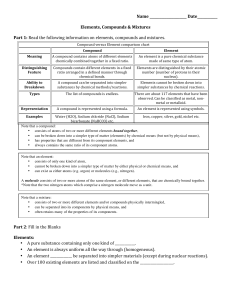
... that occurs without any chemical reaction. Substances in the mixture retain their own properties and may be physically separated from one another. • Molecule-a chemically bonded cluster of atoms. • Periodic Table of Elements-originally developed by Dimitri Mendeleev and ordered by the cycle of the e ...
... that occurs without any chemical reaction. Substances in the mixture retain their own properties and may be physically separated from one another. • Molecule-a chemically bonded cluster of atoms. • Periodic Table of Elements-originally developed by Dimitri Mendeleev and ordered by the cycle of the e ...
NOTES: 2.1 - Intro to Chemistry
... ATOM: smallest unit of matter that retains the physical and chemical properties of its element ● three subatomic particles: ...
... ATOM: smallest unit of matter that retains the physical and chemical properties of its element ● three subatomic particles: ...
Chem 1411 Chapt2
... ‘ide’. Also, one uses prefixes (mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca) to indicate the number of atoms of each element. If there is only one atom of the more electropositive element, do not use the prefix ‘mono’. Example: CO carbon monoxide; CO2 carbon dioxide NO2 nitrogen dioxi ...
... ‘ide’. Also, one uses prefixes (mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca) to indicate the number of atoms of each element. If there is only one atom of the more electropositive element, do not use the prefix ‘mono’. Example: CO carbon monoxide; CO2 carbon dioxide NO2 nitrogen dioxi ...
Compound vs Element chart
... • The atoms are _________________ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. • A compound is always homogeneous (uniform). • Compounds ___________________ be separated by physical means. Separating a compound requires a che ...
... • The atoms are _________________ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. • A compound is always homogeneous (uniform). • Compounds ___________________ be separated by physical means. Separating a compound requires a che ...
bonding notes for votech
... how many atoms of an element are in a compound Applies to the element to its immediate left If there is no subscript, it means there is only one atom of that element. What is the number of atoms in each of these compounds: H2O CO2 H2SO4 CO ...
... how many atoms of an element are in a compound Applies to the element to its immediate left If there is no subscript, it means there is only one atom of that element. What is the number of atoms in each of these compounds: H2O CO2 H2SO4 CO ...
Slide 1 - Effingham County Schools
... • Aristotle was wrong. However, his theory persisted for 2000 years. ...
... • Aristotle was wrong. However, his theory persisted for 2000 years. ...
8.P.1.1Homework for Website
... top to bottom B. The atomic size of the elements increases from left to right and increases from top to bottom C. The atomic size of the elements decreases from left to right and decreases from top to bottom 16. Which element in large amounts is poisonous to humans? A. arsenic B. carbon dioxide C. s ...
... top to bottom B. The atomic size of the elements increases from left to right and increases from top to bottom C. The atomic size of the elements decreases from left to right and decreases from top to bottom 16. Which element in large amounts is poisonous to humans? A. arsenic B. carbon dioxide C. s ...
Chemistry for Changing Times
... • Elements might combine in more than one set of proportions – Each set makes up a new compound ...
... • Elements might combine in more than one set of proportions – Each set makes up a new compound ...
+ 2 HCL(aq) CaCl2(aq) + H2O(l) + CO2(g)
... Compound: A substance made of the combined atoms of two or more elements. Chemical Formula: States what elements a compound contains and the exact number of atoms of these elements. Oxidation Number: positive or negative number on the periodic table that indicates how many electrons an element has g ...
... Compound: A substance made of the combined atoms of two or more elements. Chemical Formula: States what elements a compound contains and the exact number of atoms of these elements. Oxidation Number: positive or negative number on the periodic table that indicates how many electrons an element has g ...
Chemical reactions revision
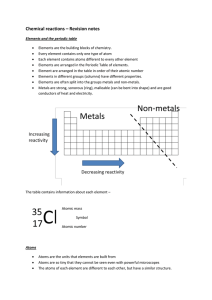
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
Chapter 8: Chemical Reactions and Physical Changes
... • Mass number: total protons and neutrons in an atom’s nucleus • Atomic mass: the average mass of a sample of atoms of that element found in nature • Periodic table: chart that arranges elements by atomic number into rows and columns according to similarities in their properties ...
... • Mass number: total protons and neutrons in an atom’s nucleus • Atomic mass: the average mass of a sample of atoms of that element found in nature • Periodic table: chart that arranges elements by atomic number into rows and columns according to similarities in their properties ...
Chemistry Unit Study Guide Key
... or more elements that are bonded together; An element is a pure, single substance made of one type of atom. 2) examples of compounds – CO2, C6H12O6, NaCl, N2, O2, Fe2O3, H2O 3) where metals and nonmetals are found on the periodic table – Metals are to the left of the zig-zag line; Non-metals are to ...
... or more elements that are bonded together; An element is a pure, single substance made of one type of atom. 2) examples of compounds – CO2, C6H12O6, NaCl, N2, O2, Fe2O3, H2O 3) where metals and nonmetals are found on the periodic table – Metals are to the left of the zig-zag line; Non-metals are to ...
Chemical Formulas
... If part of the formula is enclosed in a parenthesis, with an outside subscript number to the right of the parenthesis pair, the subscript number should be multiplied by the subscript numbers for each symbol within. For example in CO(NH 2 ) 2 there are a total of 8 atoms: one carbon (C), one oxygen ( ...
... If part of the formula is enclosed in a parenthesis, with an outside subscript number to the right of the parenthesis pair, the subscript number should be multiplied by the subscript numbers for each symbol within. For example in CO(NH 2 ) 2 there are a total of 8 atoms: one carbon (C), one oxygen ( ...
TEK 8.5D: Chemical Formulas
... If part of the formula is enclosed in a parenthesis, with an outside subscript number to the right of the parenthesis pair, the subscript number should be multiplied by the subscript numbers for each symbol within. For example in CO(NH 2 ) 2 there are a total of 8 atoms: one carbon (C), one oxygen ( ...
... If part of the formula is enclosed in a parenthesis, with an outside subscript number to the right of the parenthesis pair, the subscript number should be multiplied by the subscript numbers for each symbol within. For example in CO(NH 2 ) 2 there are a total of 8 atoms: one carbon (C), one oxygen ( ...
Notes
... Chemical Compound - A substance formed by the chemical combination of two or more elements in a fixed proportion. ...
... Chemical Compound - A substance formed by the chemical combination of two or more elements in a fixed proportion. ...
Learning Standards vocab chemical basis and molecules of life 09
... Given the number of protons, identify the element using a Periodic Table. Explain the arrangement of the elements on the Periodic Table, including the significant relationships among elements in a given column or row. Explain how ions and ionic bonds are formed (e.g., sodium atoms lose an elec ...
... Given the number of protons, identify the element using a Periodic Table. Explain the arrangement of the elements on the Periodic Table, including the significant relationships among elements in a given column or row. Explain how ions and ionic bonds are formed (e.g., sodium atoms lose an elec ...
Chapter 4 4.1 Defining the Atom • Early Models of the Atom atom
... 1) All elements are composed of tiny indivisible particles called atoms 2) Atoms of the same element are identical. Atoms of any one element are different from those of any other element 3) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratio ...
... 1) All elements are composed of tiny indivisible particles called atoms 2) Atoms of the same element are identical. Atoms of any one element are different from those of any other element 3) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratio ...