Chapter 2 - San Joaquin Memorial High School
... Where does one start in learning chemistry? Clearly we must consider some essential vocabulary and something about the origins of the science before we can proceed very far. Thus, while Chapter 1 provided background on the fundamental ideas and procedures of science in general, Chapter 2 covers the ...
... Where does one start in learning chemistry? Clearly we must consider some essential vocabulary and something about the origins of the science before we can proceed very far. Thus, while Chapter 1 provided background on the fundamental ideas and procedures of science in general, Chapter 2 covers the ...
Chapter #3
... There are N of the above equations, one for each element (atom type) in the reaction. Generally there are M coefficients to find using the N equations. Unfortunately, in most chemical equations, M > N. Usually, we have the case that M = N+1. Thus, we need to find one additional equation. One simple ...
... There are N of the above equations, one for each element (atom type) in the reaction. Generally there are M coefficients to find using the N equations. Unfortunately, in most chemical equations, M > N. Usually, we have the case that M = N+1. Thus, we need to find one additional equation. One simple ...
Introductory Chemistry Test Review
... 9. For the following chemical compounds, predict whether each will be soluble or insoluble in aqueous solution. a. Al(OH)3 b. Hg2Cl2 c. (NH4)2CO3 10. For the following aqueous chemical reactions, predict the possible products and identify any products that will be insoluble. a. CaCl2 + K2S b. MgCl2 ...
... 9. For the following chemical compounds, predict whether each will be soluble or insoluble in aqueous solution. a. Al(OH)3 b. Hg2Cl2 c. (NH4)2CO3 10. For the following aqueous chemical reactions, predict the possible products and identify any products that will be insoluble. a. CaCl2 + K2S b. MgCl2 ...
PDF(343KB)
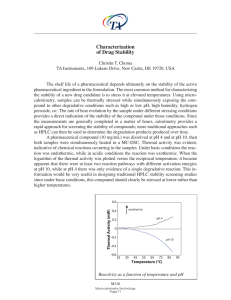
... TA Instruments, 109 Lukens Drive, New Castle, DE 19720, USA The shelf life of a pharmaceutical depends ultimately on the stability of the active pharmaceutical ingredient in the formulation. The most common method for characterizing the stability of a new drug candidate is to stress it at elevated t ...
... TA Instruments, 109 Lukens Drive, New Castle, DE 19720, USA The shelf life of a pharmaceutical depends ultimately on the stability of the active pharmaceutical ingredient in the formulation. The most common method for characterizing the stability of a new drug candidate is to stress it at elevated t ...
Chapter 8
... 1.) Based on the activity series of metals and halogens, which element is more likely to replace the other element in a compound? a. K or Na b. Al or Ni c. Bi or Cr d. Cl or F e. Au or Ag f. Cl or I g. Fe or Sr h. I or F ...
... 1.) Based on the activity series of metals and halogens, which element is more likely to replace the other element in a compound? a. K or Na b. Al or Ni c. Bi or Cr d. Cl or F e. Au or Ag f. Cl or I g. Fe or Sr h. I or F ...
File
... Write the word equation and then the balanced equation, including state symbols for the following reactions: 1. Hydrogen and oxygen combining to form water. 2. Calcium carbonate decomposing into calcium oxide and carbon dioxide. 3. Hydrochloric acid reacting with magnesium to produce hydrogen gas ...
... Write the word equation and then the balanced equation, including state symbols for the following reactions: 1. Hydrogen and oxygen combining to form water. 2. Calcium carbonate decomposing into calcium oxide and carbon dioxide. 3. Hydrochloric acid reacting with magnesium to produce hydrogen gas ...
Quantitative Chemistry
... a.) Calculate % of Magnesium (Mg) in Magnesium oxide (MgO). Ar of Mg = 24; Ar of O = 16; Mr of MgO = 24 + 16 = 40 % Mg = 24/40 x 100 = 60% b.) Calculate % of Aluminium (Al) in Aluminium bromide (AlBr3) Ar of Al = 27 Ar of Br = 80 ...
... a.) Calculate % of Magnesium (Mg) in Magnesium oxide (MgO). Ar of Mg = 24; Ar of O = 16; Mr of MgO = 24 + 16 = 40 % Mg = 24/40 x 100 = 60% b.) Calculate % of Aluminium (Al) in Aluminium bromide (AlBr3) Ar of Al = 27 Ar of Br = 80 ...
Organic Chemistry 2014 finalzzz
... same type, a locating number is given to each branch and a prefix indicating the number of that type of branch is attached to the name. ...
... same type, a locating number is given to each branch and a prefix indicating the number of that type of branch is attached to the name. ...
Chapter Five
... to increase. Note that you may never change the subscripts already in place in a chemical formula! { Why? ...
... to increase. Note that you may never change the subscripts already in place in a chemical formula! { Why? ...
The Language of Chemistry
... • In solids these particles are packed closely together, usually in a regular array. The particles vibrate back and forth about their average positions, but seldom does a particle in a solid squeeze past its immediate neighbors to come into contact with a new set of particles. • The atoms or molecul ...
... • In solids these particles are packed closely together, usually in a regular array. The particles vibrate back and forth about their average positions, but seldom does a particle in a solid squeeze past its immediate neighbors to come into contact with a new set of particles. • The atoms or molecul ...
Physical Science Semester 2 Final Exam 2013 –STUDY GUIDE
... 26. a chemical formula, the number of each type of atom in the compound is shown by numbers called ____. 27. The elements that make up a compound and the exact number of atoms of each element in a unit of the compound can be shown in a ____. 28. A chemical bond that occurs when atoms share electrons ...
... 26. a chemical formula, the number of each type of atom in the compound is shown by numbers called ____. 27. The elements that make up a compound and the exact number of atoms of each element in a unit of the compound can be shown in a ____. 28. A chemical bond that occurs when atoms share electrons ...
Lecture 1: RDCH 710 Introduction
... Np organic solution complexes • Most data with Np(V) • Evaluated with spectroscopy Monocarboxylic ligands 1:3 Np:L ratio Complexation constants increase with increasing pKa of ligand Aromatic polycarboxylates Strength based on number of carboxylic acids ...
... Np organic solution complexes • Most data with Np(V) • Evaluated with spectroscopy Monocarboxylic ligands 1:3 Np:L ratio Complexation constants increase with increasing pKa of ligand Aromatic polycarboxylates Strength based on number of carboxylic acids ...
Chemical Building Blocks Chapter One
... Matter: anything that has mass and occupies space (pg. 14) Characteristic Property: a quality of a substance that never changes and can be used to identify the substances (pg. 15) Boiling Point: the temperature at which a substance changes from a liquid to a gas (pg. 16) Physical Change: a change in ...
... Matter: anything that has mass and occupies space (pg. 14) Characteristic Property: a quality of a substance that never changes and can be used to identify the substances (pg. 15) Boiling Point: the temperature at which a substance changes from a liquid to a gas (pg. 16) Physical Change: a change in ...
Chapter 1: Quiz Review - Wetaskiwin Composite High School
... 1. What is true about the composition of one formula unit of an ionic compound, such as zinc oxide? A. The total positive charge on the cations equals the total negative charge on the anions. B. There are more positive charge C. There is more negative charge D. The charge depends upon the compound 2 ...
... 1. What is true about the composition of one formula unit of an ionic compound, such as zinc oxide? A. The total positive charge on the cations equals the total negative charge on the anions. B. There are more positive charge C. There is more negative charge D. The charge depends upon the compound 2 ...
Chapter 3 - Higher Education | Kendall Hunt Publishing
... exactly like every other part. The salt can be separated from the water by physical means. Seawater and air are also examples of homogeneous mixtures. We know that there are two types of homogeneous matter: pure substances and homogeneous mixtures. According to this classification scheme, matter can ...
... exactly like every other part. The salt can be separated from the water by physical means. Seawater and air are also examples of homogeneous mixtures. We know that there are two types of homogeneous matter: pure substances and homogeneous mixtures. According to this classification scheme, matter can ...
chapt 1 - Cantt Academy, Tahli Mohri Chowk, Rawalpindi
... Pseudo – Science or Alchemy:In the beginning of chemistry during 600 – 1600 AD some scientist tried to convert cheap metals in to gold. They performed many experiment but could not succeed and wasted their time and money. These scientists are called alchemists and this branch of chemistry is called ...
... Pseudo – Science or Alchemy:In the beginning of chemistry during 600 – 1600 AD some scientist tried to convert cheap metals in to gold. They performed many experiment but could not succeed and wasted their time and money. These scientists are called alchemists and this branch of chemistry is called ...
Chapter 2 - OrgSites.com
... Carbon atoms can form diverse molecules by bonding to four other atoms 3. How many covalent bonds can the following elements form? a. C: ___ c. O: ___ b. H: ___ d. N: ___ 4. Draw a structural formula for C6H12O6. Be sure that it conforms to the rules from the previous question. ...
... Carbon atoms can form diverse molecules by bonding to four other atoms 3. How many covalent bonds can the following elements form? a. C: ___ c. O: ___ b. H: ___ d. N: ___ 4. Draw a structural formula for C6H12O6. Be sure that it conforms to the rules from the previous question. ...
Resource for Final Exam Prep
... Which one of the following is not one of the postulates of Dalton’s atomic theory? 1) Atoms are composed of protons, neutrons, and electrons. 2) All atoms of a given element are identical; the atoms of different elements are different and have different properties. 3) Atoms of an element are not cha ...
... Which one of the following is not one of the postulates of Dalton’s atomic theory? 1) Atoms are composed of protons, neutrons, and electrons. 2) All atoms of a given element are identical; the atoms of different elements are different and have different properties. 3) Atoms of an element are not cha ...
Chapter 7 - Chemical Quantities
... quantities. For example, we buy eggs by the dozen (12), paper by the ream (500), and pop by the case (24). In chemistry, we count molecules, atoms, and ions by the mole. A mole contains 6.02 X 1023 items. This very large number is called Avogadro’s number, after the Italian ...
... quantities. For example, we buy eggs by the dozen (12), paper by the ream (500), and pop by the case (24). In chemistry, we count molecules, atoms, and ions by the mole. A mole contains 6.02 X 1023 items. This very large number is called Avogadro’s number, after the Italian ...
chemistry of living things
... • An atom is the smallest piece of an element • Atoms are made of subatomic particles – Protons have a positive charge – Neutrons have no charge – Electrons have a negative charge © 2004 Delmar Learning, a Division of Thomson Learning, Inc. ...
... • An atom is the smallest piece of an element • Atoms are made of subatomic particles – Protons have a positive charge – Neutrons have no charge – Electrons have a negative charge © 2004 Delmar Learning, a Division of Thomson Learning, Inc. ...
Chapter 3 Molecules Molecules, Compounds, and Chemical
... relative numbers of reactant and product molecules that are required can be used to determine weights of reactants used and products that can be made ...
... relative numbers of reactant and product molecules that are required can be used to determine weights of reactants used and products that can be made ...
R E V I E W -- P R A C T I C E E X A
... 78. As the number of electrons in the outer shells of atoms within a period decreases, the atoms (in general) have: a. increasing atomic radii, decreasing ionization energies and electronegativity values b. decreasing atomic radii, ionization energies and electronegativity values c. decreasing atomi ...
... 78. As the number of electrons in the outer shells of atoms within a period decreases, the atoms (in general) have: a. increasing atomic radii, decreasing ionization energies and electronegativity values b. decreasing atomic radii, ionization energies and electronegativity values c. decreasing atomi ...