Chapter 3 Atoms: the Building Blocks of Matter
... • If the numbers are both whole numbers, these will be the subscripts of the elements in the formula • If the whole numbers are identical, substitute the number 1 Example: C2H2 and C8H8 have an empirical formula of CH • If either or both numbers are not whole numbers, numbers in the ratio must be mu ...
... • If the numbers are both whole numbers, these will be the subscripts of the elements in the formula • If the whole numbers are identical, substitute the number 1 Example: C2H2 and C8H8 have an empirical formula of CH • If either or both numbers are not whole numbers, numbers in the ratio must be mu ...
NYS Regents Chemistry June 21, 2002
... b Using the graph, predict the electronegativity of nitrogen. _________________ c For these elements, state the trend in electronegativity in terms of atomic number. Base your answers to questions 64 through 69 on the following redox reaction, which occurs spontaneously. ...
... b Using the graph, predict the electronegativity of nitrogen. _________________ c For these elements, state the trend in electronegativity in terms of atomic number. Base your answers to questions 64 through 69 on the following redox reaction, which occurs spontaneously. ...
Sample Exercise 2.1 Illustrating the Size of an Atom
... (b) Two AL3+ ions are required to balance the charge of three O2– ions (that is, the total positive charge is 6+, and the total negative charge is 6–). Thus, the formula is Al2O3. (c) Two NO3– ions are needed to balance the charge of one MG2+. Thus, the formula is Mg(NO3)2. In this case the formula ...
... (b) Two AL3+ ions are required to balance the charge of three O2– ions (that is, the total positive charge is 6+, and the total negative charge is 6–). Thus, the formula is Al2O3. (c) Two NO3– ions are needed to balance the charge of one MG2+. Thus, the formula is Mg(NO3)2. In this case the formula ...
the ap chemistry summer assignment
... general chemistry class, but AP Chemistry is very different. Rather than memorizing how to do particular types of problems, you must really understand the chemistry and be able to apply it to different kinds of problems. AP Chemistry is a difficult course. To succeed you must keep up with the assign ...
... general chemistry class, but AP Chemistry is very different. Rather than memorizing how to do particular types of problems, you must really understand the chemistry and be able to apply it to different kinds of problems. AP Chemistry is a difficult course. To succeed you must keep up with the assign ...
Ch02-sample-and-practice-set-2
... (b) Two AL3+ ions are required to balance the charge of three O2– ions (that is, the total positive charge is 6+, and the total negative charge is 6–). Thus, the formula is Al2O3. (c) Two NO3– ions are needed to balance the charge of one MG2+. Thus, the formula is Mg(NO3)2. In this case the formula ...
... (b) Two AL3+ ions are required to balance the charge of three O2– ions (that is, the total positive charge is 6+, and the total negative charge is 6–). Thus, the formula is Al2O3. (c) Two NO3– ions are needed to balance the charge of one MG2+. Thus, the formula is Mg(NO3)2. In this case the formula ...
IT IS ELEMENTARY - the OLLI at UCI Blog
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
CLASS NOTES- Balancing Chemical Equations.pptx
... 4. Check your answer to see if: • The numbers of atoms on both sides of the equation are now balanced • The coefficients are in the lowest possible whole number ratios. (reduced) ...
... 4. Check your answer to see if: • The numbers of atoms on both sides of the equation are now balanced • The coefficients are in the lowest possible whole number ratios. (reduced) ...
Chemistry Review - pams-hoey
... • Reactant: substance that enters into the reaction (original substance) • Product: substance that is produced by a chemical reaction (new substance) • The Law of Conservation of Mass states that mass remains constant in a chemical reaction ...
... • Reactant: substance that enters into the reaction (original substance) • Product: substance that is produced by a chemical reaction (new substance) • The Law of Conservation of Mass states that mass remains constant in a chemical reaction ...
Worksheet 20.2
... 1- Atoms can achieve a noble gas structure by gaining, losing or sharing electrons with other atoms. 2- The rule states that, except for hydrogen , an atom combines with other atoms to form bonds in order to have 8 electrons in its valence energy level ( like noble gases). Lewis dot symbols are repr ...
... 1- Atoms can achieve a noble gas structure by gaining, losing or sharing electrons with other atoms. 2- The rule states that, except for hydrogen , an atom combines with other atoms to form bonds in order to have 8 electrons in its valence energy level ( like noble gases). Lewis dot symbols are repr ...
Exam 2
... 6. In the reduction of 2-butanone to (2)-butanol using the (S)-CBS reagent (2-methyloxazaborolidine + BH3), what is transferred in the critical step in the reaction mechanism? a) a hydride ion, H- b) a hydrogen radical, H c) a proton, H+ d) both hydrogens simultaneously as molecular hydrogen, H2 e) ...
... 6. In the reduction of 2-butanone to (2)-butanol using the (S)-CBS reagent (2-methyloxazaborolidine + BH3), what is transferred in the critical step in the reaction mechanism? a) a hydride ion, H- b) a hydrogen radical, H c) a proton, H+ d) both hydrogens simultaneously as molecular hydrogen, H2 e) ...
Metals, Nonmetals, and Metalloids (Vocabulary)
... are arranged by properties and are represented by one or two letter chemical symbols. ...
... are arranged by properties and are represented by one or two letter chemical symbols. ...
Document
... Check for Understanding Aqueous potassium nitrate and a precipitate of barium chromate are formed when aqueous solutions of barium nitrate and potassium chromate are mixed. Ba(NO3)2 (aq) + K2CrO4 (aq) ...
... Check for Understanding Aqueous potassium nitrate and a precipitate of barium chromate are formed when aqueous solutions of barium nitrate and potassium chromate are mixed. Ba(NO3)2 (aq) + K2CrO4 (aq) ...
Metals, Nonmetals, and Metalloids (Vocabulary)
... Metals, Nonmetals, and Metalloids (Vocabulary) ...
... Metals, Nonmetals, and Metalloids (Vocabulary) ...
Chemistry I Final Exam Review Problems 2016
... d. 0.40 L ____ 70. At a certain temperature and pressure, 0.20 mol of carbon dioxide has a volume of 3.1 L. A 3.1-L sample of hydrogen at the same temperature and pressure ____. a. has the same mass b. contains the same number of atoms c. has a higher density d. contains the same number of molecules ...
... d. 0.40 L ____ 70. At a certain temperature and pressure, 0.20 mol of carbon dioxide has a volume of 3.1 L. A 3.1-L sample of hydrogen at the same temperature and pressure ____. a. has the same mass b. contains the same number of atoms c. has a higher density d. contains the same number of molecules ...
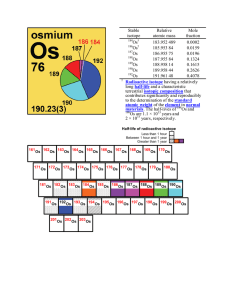
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... of amount ratios of isotopes of the same element and mole ratios of isotopes of different elements (one of which is radioactive and decays to an isotope of the other element). The time formed can indicate time since metamorphism, crystallization, shock events, differentiation of precursor melts, etc ...
... of amount ratios of isotopes of the same element and mole ratios of isotopes of different elements (one of which is radioactive and decays to an isotope of the other element). The time formed can indicate time since metamorphism, crystallization, shock events, differentiation of precursor melts, etc ...
CHEM 11 Practice Exam 2
... 8) Which of the following is a general trend from left to right in the periodic table of elements? A) atomic radius increases; ionization energy increases B) atomic radius increases; ionization energy decreases C) atomic radius decreases; ionization energy increases D) atomic radius decreases; ioni ...
... 8) Which of the following is a general trend from left to right in the periodic table of elements? A) atomic radius increases; ionization energy increases B) atomic radius increases; ionization energy decreases C) atomic radius decreases; ionization energy increases D) atomic radius decreases; ioni ...
Mole Equation Homework Hint: Start equations with the numbers
... Hint: Start equations with the numbers given, and pay close attention to what the question is asking you to find. Usually, the first step in most stoichiometry problems (calculation of quantities in chemical equations) is to convert the given numbers to moles. SHOW YOUR WORK!!!!!!!!!!!!!!!!!!!!!!!!! ...
... Hint: Start equations with the numbers given, and pay close attention to what the question is asking you to find. Usually, the first step in most stoichiometry problems (calculation of quantities in chemical equations) is to convert the given numbers to moles. SHOW YOUR WORK!!!!!!!!!!!!!!!!!!!!!!!!! ...
CHEM 120 WEEK 11 LECTURES (INORGANIC WEEK 2) Dr. MD
... Contains only metals, apart from boron. Boron is also the only element which does not form a stable trication (B3+) again will have too high a charge density to be stable. Why do the other elements form tri-cations (M3+ )? Soln. √ Because they have the valence electronic configuration ns2np1 and ...
... Contains only metals, apart from boron. Boron is also the only element which does not form a stable trication (B3+) again will have too high a charge density to be stable. Why do the other elements form tri-cations (M3+ )? Soln. √ Because they have the valence electronic configuration ns2np1 and ...
NYS Regents Chemistry
... i. Group 1 “Alkali Metals”; Group 2 “Alkaline Earth Metals”; Groups 3-11 “Transition Metals”; Group 17 “The Halogens”; Group 18 “Noble Gases” ee. Valence shell: the outermost level containing electrons i. Lewis Dot Structure: Is the atomic symbol with its valence electrons ...
... i. Group 1 “Alkali Metals”; Group 2 “Alkaline Earth Metals”; Groups 3-11 “Transition Metals”; Group 17 “The Halogens”; Group 18 “Noble Gases” ee. Valence shell: the outermost level containing electrons i. Lewis Dot Structure: Is the atomic symbol with its valence electrons ...
Year End Chemistry Review
... If 5.0 moles of water are decomposed, how many moles of oxygen are formed? If 5.0 liters of water are decomposed, how many liters of oxygen are formed? (Notice this is the same problem and can be done the same way.) ...
... If 5.0 moles of water are decomposed, how many moles of oxygen are formed? If 5.0 liters of water are decomposed, how many liters of oxygen are formed? (Notice this is the same problem and can be done the same way.) ...
Midterm 2 from Summer 2012
... Hydrazine is an extremely combustible compound composed of Nitrogen and Hydrogen that is used as a fuel for ICBM's. This compound has a composition of 87.42% Nitrogen and 12.58% Hydrogen. What is the Empirical Formula of hydrazine? a) b) c) d) ...
... Hydrazine is an extremely combustible compound composed of Nitrogen and Hydrogen that is used as a fuel for ICBM's. This compound has a composition of 87.42% Nitrogen and 12.58% Hydrogen. What is the Empirical Formula of hydrazine? a) b) c) d) ...
File
... a. carbon dioxide and water b. oxygen and water c. carbon dioxide and oxygen d. none of these 2. The solvent in an aqueous solution is always a. Oxygen b. an acid c. a base d. water 3. A solid produced during a chemical reaction is called a (an) a. solute b. precipitate c. spectator ion d. solvent 4 ...
... a. carbon dioxide and water b. oxygen and water c. carbon dioxide and oxygen d. none of these 2. The solvent in an aqueous solution is always a. Oxygen b. an acid c. a base d. water 3. A solid produced during a chemical reaction is called a (an) a. solute b. precipitate c. spectator ion d. solvent 4 ...
AP CHEMISTRY SUMMER 2016
... a. A compound with the molecular formula C6H6 has the same simplest formula. b. The mass percent of copper in CuO is less than in Cu2O. c. The limiting reactant is the one present in the smallest number of grams. d. Since C3H6O3 and C6H12O6 reduce to the same formula, they represent the same comp ...
... a. A compound with the molecular formula C6H6 has the same simplest formula. b. The mass percent of copper in CuO is less than in Cu2O. c. The limiting reactant is the one present in the smallest number of grams. d. Since C3H6O3 and C6H12O6 reduce to the same formula, they represent the same comp ...