
Midterm Review 4
... Chemistry: First Semester Exam Prep #4 (Choose the BEST answer.) 52. The most stable atoms are those of the a. metals b. metalloids c. noble gases d. nonmetals 53. The ion with a charge of +1 and the same electron configuration as argon is a. potassium b. sodium c. neon d. magnesium 54. The tendenc ...
... Chemistry: First Semester Exam Prep #4 (Choose the BEST answer.) 52. The most stable atoms are those of the a. metals b. metalloids c. noble gases d. nonmetals 53. The ion with a charge of +1 and the same electron configuration as argon is a. potassium b. sodium c. neon d. magnesium 54. The tendenc ...
Unit 1 Powerpoint
... High energy bonds are found in organic molecules such as lipids and ATP. They are covalent bonds that, when broken, release high amounts of energy. So, the energy needed for your cells to function is stored in high energy bonds. When your cell needs energy, it breaks those bonds by breaking down mol ...
... High energy bonds are found in organic molecules such as lipids and ATP. They are covalent bonds that, when broken, release high amounts of energy. So, the energy needed for your cells to function is stored in high energy bonds. When your cell needs energy, it breaks those bonds by breaking down mol ...
Matter - GEOCITIES.ws
... Relative molecular mass: The molecular mass of a substance is the relative mass of the substance as compared to 1/12 of the mass of the C-12 atom. For eg. Relative molecular mass = mass of one molecule of the substance 1/12 of the mass of the C-12 atom Gram Atomic mass: atomic mass expressed in gram ...
... Relative molecular mass: The molecular mass of a substance is the relative mass of the substance as compared to 1/12 of the mass of the C-12 atom. For eg. Relative molecular mass = mass of one molecule of the substance 1/12 of the mass of the C-12 atom Gram Atomic mass: atomic mass expressed in gram ...
Chapter 7 – Chemical Formulas and Chemical
... 2 on pg 226 gives common polyatomics and their names. Ex: Mg+2 + ClO2- Mg(ClO2)2 magnesium chlorite Ag+2 + NO3- Ag(NO3)2 silver (II) nitrate Naming Binary Molecular Compounds The old system of naming molecular compounds is based on using prefixes. These prefixes are on pg 228. The prefix mono- m ...
... 2 on pg 226 gives common polyatomics and their names. Ex: Mg+2 + ClO2- Mg(ClO2)2 magnesium chlorite Ag+2 + NO3- Ag(NO3)2 silver (II) nitrate Naming Binary Molecular Compounds The old system of naming molecular compounds is based on using prefixes. These prefixes are on pg 228. The prefix mono- m ...
chem final review
... 35) Which of the following formulas represents an ionic compound? A) Kr B) BaI2 C) PCl3 D) N2O4 E) CS2 36) In which of the following groups of ions are the charges all shown correctly? A) Ca2+, Al3+, BrB) Be2+, Cl2-, Sr2+ C) Li-, O2-, S2+ D) Na+, I-, RbE) K2-, F-, Mg2+ 37) In which of the following ...
... 35) Which of the following formulas represents an ionic compound? A) Kr B) BaI2 C) PCl3 D) N2O4 E) CS2 36) In which of the following groups of ions are the charges all shown correctly? A) Ca2+, Al3+, BrB) Be2+, Cl2-, Sr2+ C) Li-, O2-, S2+ D) Na+, I-, RbE) K2-, F-, Mg2+ 37) In which of the following ...
Section 8.3 Names and Formulas of Ionic Compounds Formula Unit
... • Many ionic compounds contain polyatomic ions, which are ions made up of more than one atom. • The charge given to a polyatomic ion applies to the entire group of atoms. ...
... • Many ionic compounds contain polyatomic ions, which are ions made up of more than one atom. • The charge given to a polyatomic ion applies to the entire group of atoms. ...
Chemistry - Halifax County Public Schools
... Dr. S. Stuart Flanagan, Professor Emeritus College of William and Mary Copyright 2005, S. Stuart Flanagan and David E. W. Mott Do not reproduce without permission. ...
... Dr. S. Stuart Flanagan, Professor Emeritus College of William and Mary Copyright 2005, S. Stuart Flanagan and David E. W. Mott Do not reproduce without permission. ...
3UE-Exam Review-June2010 - Savita Pall and Chemistry
... 39. The increase in boiling points observed for F2 , Cl2 , Br2 , I2 is best attributed to... a) an increase in Van der Waals’ forces with increasing atomic number. b) a decrease in the electronegativity with increasing atomic number. c) an increase in the X - X bond energy with increasing atomic nu ...
... 39. The increase in boiling points observed for F2 , Cl2 , Br2 , I2 is best attributed to... a) an increase in Van der Waals’ forces with increasing atomic number. b) a decrease in the electronegativity with increasing atomic number. c) an increase in the X - X bond energy with increasing atomic nu ...
Chapter 3 Molecules, Compounds, and Chemical Equations
... element furthest left and down on the Periodic Table Use the full name of the element, Write the name of the second element in the formula with an -ide suffix. ...
... element furthest left and down on the Periodic Table Use the full name of the element, Write the name of the second element in the formula with an -ide suffix. ...
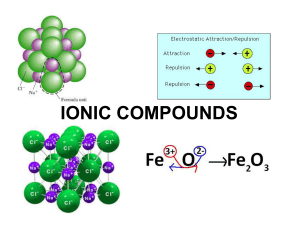
ChemChapter_7sec1_and_section2[1]FORMULA
... Rules for Oxidation Numbers 1. The oxidation number of an element in its elemental form is zero. Examples of this are N2 (g), O2 (g), Na (s), Cl2 (g), etc. 2. The oxidation number of a monatomic ion is exactly the same as its charge. So, Group IA ions will all have an oxidation number of +1, since ...
... Rules for Oxidation Numbers 1. The oxidation number of an element in its elemental form is zero. Examples of this are N2 (g), O2 (g), Na (s), Cl2 (g), etc. 2. The oxidation number of a monatomic ion is exactly the same as its charge. So, Group IA ions will all have an oxidation number of +1, since ...
Chapter 4
... Atomic Structures of Ions (cont.) • Nonmetals form anions. • For each negative charge the ion has 1 more electron than the neutral atom. – F = 9 e-, F- = 10 e– P = 15 e-, P3- = 18 e- ...
... Atomic Structures of Ions (cont.) • Nonmetals form anions. • For each negative charge the ion has 1 more electron than the neutral atom. – F = 9 e-, F- = 10 e– P = 15 e-, P3- = 18 e- ...
Chemical Reactions and Equations
... Starting with elements that only occur in one substance on each side of the equation, make sure that each side of the equation has an equal # of that element. Proceed with all elements. Remember that changing the # of one element may alter elements that have already been ...
... Starting with elements that only occur in one substance on each side of the equation, make sure that each side of the equation has an equal # of that element. Proceed with all elements. Remember that changing the # of one element may alter elements that have already been ...
- Aboriginal Access to Engineering
... iron compounds with carbon and limestone under temperatures of more than 500oC. Depending on what the steel will be used for, other metallic compounds may be added to the molten mixture in order to get specific properties in the final product like strength, chemical-resistance or hardness. For examp ...
... iron compounds with carbon and limestone under temperatures of more than 500oC. Depending on what the steel will be used for, other metallic compounds may be added to the molten mixture in order to get specific properties in the final product like strength, chemical-resistance or hardness. For examp ...
Worksheet
... Don’t forget, once you switch cations, you must again write the charges as superscripts and then cross them. Synthesis Reactions In a synthesis reaction you start out with two separate elements on the reactants side and then combine them to form a compound on the products side. The general pattern o ...
... Don’t forget, once you switch cations, you must again write the charges as superscripts and then cross them. Synthesis Reactions In a synthesis reaction you start out with two separate elements on the reactants side and then combine them to form a compound on the products side. The general pattern o ...
ASFG High School Summer Assignment Summer 2016
... f. Krypton g. Fluorine h. Scandium I. Arsenic J. Potassium K. Sodium l. chloride m. Iron n. Zinc ...
... f. Krypton g. Fluorine h. Scandium I. Arsenic J. Potassium K. Sodium l. chloride m. Iron n. Zinc ...
Stoichiometry …like a beautiful sunset on a serene lake – NOT!
... • In a chemical reaction, atoms have been rearranged but have NOT been created or destroyed. This is why we have to balance every chemical reaction/equation. Java-Balancing! • Balance CH4 + O2 → CO2 + H2O • Balance HCl + NaHCO3 → CO2 + H2O + NaCl • When balancing equations, the formulas must NEVER b ...
... • In a chemical reaction, atoms have been rearranged but have NOT been created or destroyed. This is why we have to balance every chemical reaction/equation. Java-Balancing! • Balance CH4 + O2 → CO2 + H2O • Balance HCl + NaHCO3 → CO2 + H2O + NaCl • When balancing equations, the formulas must NEVER b ...
Writing and Balancing Chemical Equations
... Reactants must be an element and a compound. Products will be a different element and a different compound. Na + KCl K + NaCl (Cations switched) (Anions switched) F2 + LiCl LiF + Cl2 ...
... Reactants must be an element and a compound. Products will be a different element and a different compound. Na + KCl K + NaCl (Cations switched) (Anions switched) F2 + LiCl LiF + Cl2 ...
CP - Fundamentals
... Recall that Dalton suggested atoms were very small. In contrast, we are very big. So, if we want to work with reasonable quantities of materials (like amounts we can hold in our hand and see), we have to deal with incredibly large numbers of atoms. It is out of this need to hold an amount we can see ...
... Recall that Dalton suggested atoms were very small. In contrast, we are very big. So, if we want to work with reasonable quantities of materials (like amounts we can hold in our hand and see), we have to deal with incredibly large numbers of atoms. It is out of this need to hold an amount we can see ...
Directed Reading
... a. It is ignored. b. It is figured at 1 over 1,840. c. It is figured at 1 for every proton. d. It is figured at 1,840 for every proton. ______ 41. Although all atoms of the same element contain the same number of protons, the number of its a. neutrons may differ. b. neutrons is always smaller. c. po ...
... a. It is ignored. b. It is figured at 1 over 1,840. c. It is figured at 1 for every proton. d. It is figured at 1,840 for every proton. ______ 41. Although all atoms of the same element contain the same number of protons, the number of its a. neutrons may differ. b. neutrons is always smaller. c. po ...
Chapter 2
... • All mater is made of atoms and molecules that act like tiny particles. • These tiny particles are always moving..the higher the temp. the faster they move. • At the same temp. more massive particles move slower than less massive particles. ...
... • All mater is made of atoms and molecules that act like tiny particles. • These tiny particles are always moving..the higher the temp. the faster they move. • At the same temp. more massive particles move slower than less massive particles. ...
Chapter 11 Chemical Reactions
... replacement reaction will happen: –Because some chemicals are more “active” than others –More active replaces less active There is a list called the Activity Series of Metals Higher on the list replaces those lower. ...
... replacement reaction will happen: –Because some chemicals are more “active” than others –More active replaces less active There is a list called the Activity Series of Metals Higher on the list replaces those lower. ...
CHEMICAL EQUATIONS NAME PERIOD_______ DATE________
... reaction. In a chemical equation, the substances on the left side of the arrow are the starting substances. These substances are called ______________. The substances on the right side of the arrow are the substances that result from the reaction. These substances are called ____________________. Th ...
... reaction. In a chemical equation, the substances on the left side of the arrow are the starting substances. These substances are called ______________. The substances on the right side of the arrow are the substances that result from the reaction. These substances are called ____________________. Th ...









![ChemChapter_7sec1_and_section2[1]FORMULA](http://s1.studyres.com/store/data/000546743_1-278f96ccbbfd49e292510ec017e27124-300x300.png)












