syllabus for entrance examination - NTU.edu
... The elements magnesium, calcium, strontium and barium. A group of reactive metals which are essentially similar to each other with only gradual changes as their atomic numbers increase. (a) ...
... The elements magnesium, calcium, strontium and barium. A group of reactive metals which are essentially similar to each other with only gradual changes as their atomic numbers increase. (a) ...
Question 2
... Predict if a precipitation reaction will occur in each of the following cases. If it does, write the full, balanced equation AND the net ionic equation (including state symbols) to show the formation of the precipitate. If there is no reaction, say so, and indicate why. (9) a) CuSO4(aq) + Na2CO3(aq) ...
... Predict if a precipitation reaction will occur in each of the following cases. If it does, write the full, balanced equation AND the net ionic equation (including state symbols) to show the formation of the precipitate. If there is no reaction, say so, and indicate why. (9) a) CuSO4(aq) + Na2CO3(aq) ...
Types of Chemical Reactions
... Divide the smallest number of moles of an element into the moles of each element present. Convert the fractional ratios for each element into whole numbers by multiplying all the ratios by the same number. The resulting numbers are the subscripts for the each element in the empirical formula. ...
... Divide the smallest number of moles of an element into the moles of each element present. Convert the fractional ratios for each element into whole numbers by multiplying all the ratios by the same number. The resulting numbers are the subscripts for the each element in the empirical formula. ...
Chapter 3 Atomic Mass
... C3H8 (g)+ 5O2 (g) 3CO2 (g) + H2O (g) Usually CO2 (carbon dioxide) / CO (carbon monoxide) and water are produced. ...
... C3H8 (g)+ 5O2 (g) 3CO2 (g) + H2O (g) Usually CO2 (carbon dioxide) / CO (carbon monoxide) and water are produced. ...
Answers - U of L Class Index
... aluminium products should rust easily; however, they do not. Why don’t aluminium products rust? Pure aluminium reacts quickly with oxygen to form a thin layer of aluminium oxide on its surface. This layer of aluminium oxide does not react further with oxygen. As such, aluminium products do not rust. ...
... aluminium products should rust easily; however, they do not. Why don’t aluminium products rust? Pure aluminium reacts quickly with oxygen to form a thin layer of aluminium oxide on its surface. This layer of aluminium oxide does not react further with oxygen. As such, aluminium products do not rust. ...
Know (main topic)
... divide, add, and subtract, very large and very small numbers. -describe the difference bet. the four states of matter. ...
... divide, add, and subtract, very large and very small numbers. -describe the difference bet. the four states of matter. ...
Chemical changes
... 2. There are three rules for naming compounds: the “ide” rule, the “ate” rule and the “same” rule. ...
... 2. There are three rules for naming compounds: the “ide” rule, the “ate” rule and the “same” rule. ...
Chemical Reactions
... – atoms are neither created nor destroyed (they only change bonding partners) – same atoms are present in reactants as in products ...
... – atoms are neither created nor destroyed (they only change bonding partners) – same atoms are present in reactants as in products ...
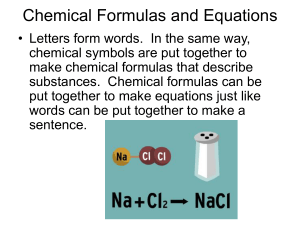
Chemical Formulas and Equations
... • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
... • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
Objective 4
... Group 17 (7A) - elements have 7 electrons in their valance shell, (they want 1 more electron to make 8) *very reactive Group 18 - NOBLE GASES are INERT GASES (not reactive) ...
... Group 17 (7A) - elements have 7 electrons in their valance shell, (they want 1 more electron to make 8) *very reactive Group 18 - NOBLE GASES are INERT GASES (not reactive) ...
Ionic bonding - Nidderdale High School
... What is the mass number of this chlorine atom? What is the atomic number of this chlorine atom? How many protons neutrons and electrons does this chlorine atom have? 4. What is the electron configuration of a chlorine atom? 5. What is the relative mass of an electron? 6. What is the charge on a neut ...
... What is the mass number of this chlorine atom? What is the atomic number of this chlorine atom? How many protons neutrons and electrons does this chlorine atom have? 4. What is the electron configuration of a chlorine atom? 5. What is the relative mass of an electron? 6. What is the charge on a neut ...
Chapter 1-3 Exam Review
... --------------------------------------------------------------------------------------------------------------------Elements found as diatomic molecules in nature include: hydrogen, oxygen, fluorine, bromine, iodine, nitrogen, chlorine H2 O2 F2 Br2 I2 N2 Cl2 ...
... --------------------------------------------------------------------------------------------------------------------Elements found as diatomic molecules in nature include: hydrogen, oxygen, fluorine, bromine, iodine, nitrogen, chlorine H2 O2 F2 Br2 I2 N2 Cl2 ...
AP Chemistry Summer Assignment
... 2. a molecular species > For general chemistry this is water. 3. a precipitate (ppt) must form > You need to use the solubility rules below or if given a solubility chart. The ppt must be identified as a product with a symbol in the reaction. ...
... 2. a molecular species > For general chemistry this is water. 3. a precipitate (ppt) must form > You need to use the solubility rules below or if given a solubility chart. The ppt must be identified as a product with a symbol in the reaction. ...
CHEM 115 EXAM #1 - chem.wilkes.edu
... temperature is listed as 320 K. Express this temperature in both ºC and ºF. °C = K – 273.15 = 46.85 (rounds to 47°C) °F = 1.8(°C) +32 = 116°F (note, I used 46.85 in this second calculation and then rounded) ...
... temperature is listed as 320 K. Express this temperature in both ºC and ºF. °C = K – 273.15 = 46.85 (rounds to 47°C) °F = 1.8(°C) +32 = 116°F (note, I used 46.85 in this second calculation and then rounded) ...
Materials Science for Chemical Engineers
... - When 2 atoms or more atoms are more stable as an aggregate, a chemical bond is formed. What is the criteria for an unstable atom?- an incompletely filled quantum shell How does the atom fill this cell?- by forming a chemical bond Types of Bonding (1) strong, primary or chemical bonds: Covalent Bon ...
... - When 2 atoms or more atoms are more stable as an aggregate, a chemical bond is formed. What is the criteria for an unstable atom?- an incompletely filled quantum shell How does the atom fill this cell?- by forming a chemical bond Types of Bonding (1) strong, primary or chemical bonds: Covalent Bon ...
Ch. 07 Notes ch7notes
... • Chemical formulas give a ratio of elemental components. • Ionic formulas show the simplest ratio of cations and anions. • Covalent formulas (including polyatomic ions) show both elements and the number of each element. Formulas can be used to calculate Molar Masses • From formulas we can tell what ...
... • Chemical formulas give a ratio of elemental components. • Ionic formulas show the simplest ratio of cations and anions. • Covalent formulas (including polyatomic ions) show both elements and the number of each element. Formulas can be used to calculate Molar Masses • From formulas we can tell what ...
Transition metals and coordination chemistry
... molecule of the chemical formula [Pt(NH3)2Cl2] were connected. The theories at the time predicted such molecules to be connected as linear chains: [Pt-NH3-NH3-Cl]Cl or Cl-NH3-Pt-NH3-Cl ...
... molecule of the chemical formula [Pt(NH3)2Cl2] were connected. The theories at the time predicted such molecules to be connected as linear chains: [Pt-NH3-NH3-Cl]Cl or Cl-NH3-Pt-NH3-Cl ...
04 Biochemistry
... • e- like to be in pairs, but fill single e- first before pairing them up. • Octet rule = an atom in 2nd energy level always likes to have 8 e- on the outermost energy level. • When bonds form between two atoms, only the unpaired valence e- from the two atoms pair up. ...
... • e- like to be in pairs, but fill single e- first before pairing them up. • Octet rule = an atom in 2nd energy level always likes to have 8 e- on the outermost energy level. • When bonds form between two atoms, only the unpaired valence e- from the two atoms pair up. ...
Chapter 8 Test Review
... Empirical Formula – formula of a compound that expresses lowest whole number ratio of atoms. Molecular Formula – actual formula of a compound showing the number of atoms present ...
... Empirical Formula – formula of a compound that expresses lowest whole number ratio of atoms. Molecular Formula – actual formula of a compound showing the number of atoms present ...
Chapter 2: Atoms, Molecules, and Ions
... D) A and C E) A, B, and C 18. Which one of the following statements about atomic structure is false? A) An atom is mostly empty space. B) Almost all of the mass of the atom is concentrated in the nucleus. C) The protons and neutrons in the nucleus are very tightly packed. D) The number of protons an ...
... D) A and C E) A, B, and C 18. Which one of the following statements about atomic structure is false? A) An atom is mostly empty space. B) Almost all of the mass of the atom is concentrated in the nucleus. C) The protons and neutrons in the nucleus are very tightly packed. D) The number of protons an ...
3.1 Atomic Mass - Pace University Webspace
... • In nature, most elements have more than one isotope, meaning that the same element with a different number of neutrons exists. • The average atomic mass that is seen on the periodic table is the average mass of the different isotopes of an element that occur naturally. • To figure out the average ...
... • In nature, most elements have more than one isotope, meaning that the same element with a different number of neutrons exists. • The average atomic mass that is seen on the periodic table is the average mass of the different isotopes of an element that occur naturally. • To figure out the average ...
Section 1-2 Matter and Its Properties
... States of Matter Matter in the solid state has definite volume and definite shape; example: rocks, glass Matter in the liquid state has a definite volume, but no definite shape; example: water Matter in the gas state has no definite volume or definite shape; example: helium, oxygen Plasma is ...
... States of Matter Matter in the solid state has definite volume and definite shape; example: rocks, glass Matter in the liquid state has a definite volume, but no definite shape; example: water Matter in the gas state has no definite volume or definite shape; example: helium, oxygen Plasma is ...