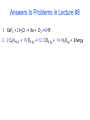* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter #3
Double layer forces wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Al-Shifa pharmaceutical factory wikipedia , lookup
Rate equation wikipedia , lookup
Safety data sheet wikipedia , lookup
California Green Chemistry Initiative wikipedia , lookup
Chemical weapon proliferation wikipedia , lookup
Drug discovery wikipedia , lookup
History of molecular theory wikipedia , lookup
Chemical plant wikipedia , lookup
Chemical weapon wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Chemical Corps wikipedia , lookup
Computational chemistry wikipedia , lookup
History of chemistry wikipedia , lookup
Chemical industry wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Spinodal decomposition wikipedia , lookup
Chemical potential wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Transition state theory wikipedia , lookup
Atomic theory wikipedia , lookup
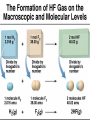
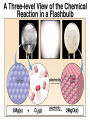
Lecture #8 Balancing Chemical Equations: The Atom Conservation Approach Chemistry 142 B James B. Callis, Instructor Autumn Quarter, 2004 Chemical Equations Qualitative Information: Reactants Products States of Matter: (s) solid (l) liquid (g) gaseous (aq) aqueous 2 H2 (g) + O2 (g) But also Quantitative Information! 2 H2O (g) Information Contained in a Balanced Equation Viewed in terms of: Reactants Products 2 C2H6 (g) + 7 O2 (g) = 4 CO2 (g) + 6 H2O(g) + Energy Molecules Amount (mol) Mass (amu) Mass (g) Total Mass (g) Balancing Chemical Equations by Explicit Consideration of the Atom Conservation Equations We start by considering the general algebraic expression for a chemical equation: xC yC ... uC vC ... x y u v Where Cx, Cy, …. represent the chemical formulas of the reactants and Cu, Cv, … represent the chemical formula of the products. The coefficients x, y, …. represent the (sought for) coefficients of the reactants and the coefficients u, v, …. represent the (sought for) coefficients of the reactants We assume that the molecular formula for each of the reactants and products is known and is represented in the format: C An x Bn x ... x A B Where Cx is the reactant whose chemical balance coefficient is x. The integers nxA are the number of atoms of type A in the compound Cx. For each elemental specie in the reaction, there must be the same number of atoms on both sides of the chemical equation. We express this as a series of atom conservation equations, one for each element. For the element A: xn yn ... un vn .... x A y A u A v A Similar equations are written for each one of the atom types. All of these equations must be simultaneously obeyed. There are N of the above equations, one for each element (atom type) in the reaction. Generally there are M coefficients to find using the N equations. Unfortunately, in most chemical equations, M > N. Usually, we have the case that M = N+1. Thus, we need to find one additional equation. One simple way to solve the problem is to arbitrarily set one of the coefficients to one and then solve the system of equations. We find this approach works best when the coefficient of the most complicated chemical species (the one with the largest number of elements) is set to 1. After all of the coefficients have been solved, we then multiply the equation by the smallest integer that will eliminate any fractions. Thus our balanced equation contains the set of minimum integers. Problem 8-1: Balance the following chemical equation using the atom conservation method. x XeF4 + y H2O -> u Xe + v O2 + w HF Atom Conservation Equations: Solution: Xe: F: H: O: Final Result: Balancing Chemical Equations- Example #2 Problem 8-2: The hydrocarbon hexane is a component of Gasoline that burns in an automobile engine to produce carbon dioxide and water as well as energy. Write the balanced chemical equation for the combustion of hexane (C6H14). Plan: Write the skeleton equation from the words into chemical compounds with blanks before each compound. Assign unknowns x, y, … etc. to each blank Skeleton equation: Assign unknowns: Problem 8-2: Balance the following: x C6H14 (l) + y O2 (g) Atom Conservation Equations: -> u CO2 (g) + v H2O(g) + Energy Solution: C: H: O: Final Result: C6H14 (l) + O2 (g) -> CO2 (g) + H2O(g) + Energy Answers to Problems in Lecture #8 1. XeF4 + 2 H2O -> Xe + O2 +4 HF 2. 2 C6H14 (l) + 19 O2 (g) -> 12 CO2 (g) + 14 H2O(g) + Energy