Microbial fermentation (Enzymology,metabolic pathways and
... Enzymes. Enzymes equilibrium state. Factors effects enzymes catalytic activity. Natural mechanisms for regulating enzyme activity. Enzymes activators. Classification of enzymes. ...
... Enzymes. Enzymes equilibrium state. Factors effects enzymes catalytic activity. Natural mechanisms for regulating enzyme activity. Enzymes activators. Classification of enzymes. ...
Identification of the Human Erythrocyte Glucose Transporter (GLUT1
... composed primarily of lipids ...
... composed primarily of lipids ...
Biochemical Screening of Pyrimidine
... source may be the most sensitive component of the system. Agents which act upon one or more steps in the oxidative cycle could appear to be inhibitors of reactions in which they actually have no effect. However, in this study oxidative phosphorylation was deliberately chosen as an energy source beca ...
... source may be the most sensitive component of the system. Agents which act upon one or more steps in the oxidative cycle could appear to be inhibitors of reactions in which they actually have no effect. However, in this study oxidative phosphorylation was deliberately chosen as an energy source beca ...
Energy Substrate Metabolism in - Journal of Clinical Investigation
... in 24 hr and 7.5% in 48 hr. By comparison with the much greater per cent decreases in the rates of oleic acid-1-'C oxidation after storage of the platelets, the small changes in specific radioactivity of the fatty acid pool were considered negligible. Changes in the specific activity of the fatty ac ...
... in 24 hr and 7.5% in 48 hr. By comparison with the much greater per cent decreases in the rates of oleic acid-1-'C oxidation after storage of the platelets, the small changes in specific radioactivity of the fatty acid pool were considered negligible. Changes in the specific activity of the fatty ac ...
Expression of phosphofructokinase in Neisseria meningitidis
... step in the vaccine production process. For this reason, an N. meningitidis serogroup B genome-scale metabolic model has been built and used to study primary metabolism (Baart et al., 2007a, 2008). Earlier studies on glucose utilization in N. meningitidis confirm the presence of enzymes related to t ...
... step in the vaccine production process. For this reason, an N. meningitidis serogroup B genome-scale metabolic model has been built and used to study primary metabolism (Baart et al., 2007a, 2008). Earlier studies on glucose utilization in N. meningitidis confirm the presence of enzymes related to t ...
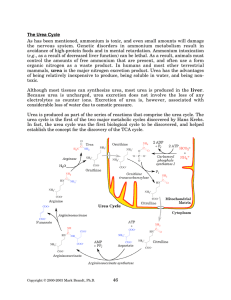
The Urea Cycle - Rose
... N-acetylglutamate (the product of the first step in ornithine biosynthesis) for activity. This regulation means that carbamoyl phosphate synthetase I is the rate-limiting enzyme of the urea cycle. The other four enzymes are part of the actual cycle. The cycle begins with the addition of carbamoyl ph ...
... N-acetylglutamate (the product of the first step in ornithine biosynthesis) for activity. This regulation means that carbamoyl phosphate synthetase I is the rate-limiting enzyme of the urea cycle. The other four enzymes are part of the actual cycle. The cycle begins with the addition of carbamoyl ph ...
Analysis and simulation of metabolic networks: Application to HEPG2
... Figure 1. Example of a reconstructed metabolic network. In the second case, i.e., the dynamic analysis, the objective is to obtain the time dependent changes of concentration of metabolites, proteins or other cellular components and reaction fluxes. An important consideration is the different time c ...
... Figure 1. Example of a reconstructed metabolic network. In the second case, i.e., the dynamic analysis, the objective is to obtain the time dependent changes of concentration of metabolites, proteins or other cellular components and reaction fluxes. An important consideration is the different time c ...
enzyme structure
... common being the ribosome; these are referred to as either RNA-enzymes or ribozymes. The activities of enzymes are determined by their three-dimensional structure. However, although structure does determine function, predicting a novel enzyme's activity just from its structure is a very difficult pr ...
... common being the ribosome; these are referred to as either RNA-enzymes or ribozymes. The activities of enzymes are determined by their three-dimensional structure. However, although structure does determine function, predicting a novel enzyme's activity just from its structure is a very difficult pr ...
Metabolomic and flux‐balance analysis of age‐related decline of
... controversial (Opie, 1998a), but is most likely to involve both necrotic and apoptotic mechanisms, stemming from metabolic stresses introduced during reperfusion as well as the ischaemic event itself. Reduced O2 causes reduction in oxidative metabolism and increased dependence on glycolysis. Under n ...
... controversial (Opie, 1998a), but is most likely to involve both necrotic and apoptotic mechanisms, stemming from metabolic stresses introduced during reperfusion as well as the ischaemic event itself. Reduced O2 causes reduction in oxidative metabolism and increased dependence on glycolysis. Under n ...
Gluconeogenesis, Glycogen Metabolism, and the Pentose
... cytoplasm by a transport protein, and once in the cytoplasm it is converted back to oxaloacetate by the action of the cytoplasmic isoenzyme of Malate Dehydrogenase. The net result of these three reactions is the conversion of pyruvate in the cytoplasm to oxaloacetate in the cytoplasm. Oxaloacetate i ...
... cytoplasm by a transport protein, and once in the cytoplasm it is converted back to oxaloacetate by the action of the cytoplasmic isoenzyme of Malate Dehydrogenase. The net result of these three reactions is the conversion of pyruvate in the cytoplasm to oxaloacetate in the cytoplasm. Oxaloacetate i ...
Biochemistry 304 2014 Student Edition Glycolysis Lectures
... Berg, Tymoczko & Stryer, 2002 Fig. 16.X ...
... Berg, Tymoczko & Stryer, 2002 Fig. 16.X ...
Amino Acid Oxidation, the Production of Urea, and Amino Acid
... Digestion of Proteins Protein digestion begins in the stomach. The stomach secretes HCl which lowers the pH of the stomach contents to between 1 and 2. The low pH serves to denature the dietary proteins. Denatured proteins are more easily attacked by the digestive proteases (protein hydrolases). Sto ...
... Digestion of Proteins Protein digestion begins in the stomach. The stomach secretes HCl which lowers the pH of the stomach contents to between 1 and 2. The low pH serves to denature the dietary proteins. Denatured proteins are more easily attacked by the digestive proteases (protein hydrolases). Sto ...
Photo Album
... Figure 3.9 Anaplerotic reactions are required for net synthesis of glutamate, glutamine, and aspartate and for normal synaptic transmission. (A) Net synthesis of TCA cycle-derived amino acids requires the ATP-dependent CO2 fixation reaction catalyzed by pyruvate carboxylase to generate oxaloacetate ...
... Figure 3.9 Anaplerotic reactions are required for net synthesis of glutamate, glutamine, and aspartate and for normal synaptic transmission. (A) Net synthesis of TCA cycle-derived amino acids requires the ATP-dependent CO2 fixation reaction catalyzed by pyruvate carboxylase to generate oxaloacetate ...
Electron transport chain…
... explain oxidative phosphorylation – electron transport chain organized so protons move outward from the mitochondrial matrix as electrons are transported down the chain – proton expulsion during electron transport results in the formation of a concentration gradient of protons and a charge gradient ...
... explain oxidative phosphorylation – electron transport chain organized so protons move outward from the mitochondrial matrix as electrons are transported down the chain – proton expulsion during electron transport results in the formation of a concentration gradient of protons and a charge gradient ...
Chapter 29 The Organic Chemistry of Metabolic Pathways
... Breakdown of Smaller Molecules Sugars and fat components are broken down in steps ...
... Breakdown of Smaller Molecules Sugars and fat components are broken down in steps ...
18. Metabolism of lipids 1
... Insulin stimulates fatty acid synthesis causing dephosphorylation of carboxylase. Glucagon and epinephrine have the reverse effect (keep the carboxylase in the inactive phosphorylated state). Protein kinase is activated by AMP and inhibited by ATP. Carboxylase is inactivated when the energy charge i ...
... Insulin stimulates fatty acid synthesis causing dephosphorylation of carboxylase. Glucagon and epinephrine have the reverse effect (keep the carboxylase in the inactive phosphorylated state). Protein kinase is activated by AMP and inhibited by ATP. Carboxylase is inactivated when the energy charge i ...
Chapter 29 The Organic Chemistry of Metabolic Pathways
... Breakdown of Smaller Molecules Sugars and fat components are broken down in steps ...
... Breakdown of Smaller Molecules Sugars and fat components are broken down in steps ...
Document
... Bloom's Taxonomy: Comprehension Chapter Section: 3.5 9) Free-energy calculations are dependent on the rates of the reactions. Answer: FALSE Bloom's Taxonomy: Knowledge Chapter Section: 3.4 10) With respect to nitrogen utilization, relatively few bacteria can use NH3 whereas many more can make use of ...
... Bloom's Taxonomy: Comprehension Chapter Section: 3.5 9) Free-energy calculations are dependent on the rates of the reactions. Answer: FALSE Bloom's Taxonomy: Knowledge Chapter Section: 3.4 10) With respect to nitrogen utilization, relatively few bacteria can use NH3 whereas many more can make use of ...
1 - Free
... 12. list the allosteric regulators of citrate synthase. Indicate whether the given substance is an activator or an inhibitor. Citrate, ATP, (-) ADP(+) 13. what is the first freely diffusible intermediate which takes over hydrogens deriving from succinate? FAD+ 14. name the compound which can carry t ...
... 12. list the allosteric regulators of citrate synthase. Indicate whether the given substance is an activator or an inhibitor. Citrate, ATP, (-) ADP(+) 13. what is the first freely diffusible intermediate which takes over hydrogens deriving from succinate? FAD+ 14. name the compound which can carry t ...
In Vitro Nail Penetration of Nitric Oxide
... were determined following exposure to each concentration of drug substance at 4 and 24 hrs. In Vitro Nitric Oxide Release Assay: The active drug phase was mixed in a 1:1 ratio with the appropriate hydrogel phase at room temperature in a custom reactor with a controlled gas flow of nitrogen to carry ...
... were determined following exposure to each concentration of drug substance at 4 and 24 hrs. In Vitro Nitric Oxide Release Assay: The active drug phase was mixed in a 1:1 ratio with the appropriate hydrogel phase at room temperature in a custom reactor with a controlled gas flow of nitrogen to carry ...
Adenosine triphosphate
Adenosine triphosphate (ATP) is a nucleoside triphosphate used in cells as a coenzyme often called the ""molecular unit of currency"" of intracellular energy transfer.ATP transports chemical energy within cells for metabolism. It is one of the end products of photophosphorylation, cellular respiration, and fermentation and used by enzymes and structural proteins in many cellular processes, including biosynthetic reactions, motility, and cell division. One molecule of ATP contains three phosphate groups, and it is produced by a wide variety of enzymes, including ATP synthase, from adenosine diphosphate (ADP) or adenosine monophosphate (AMP) and various phosphate group donors. Substrate-level phosphorylation, oxidative phosphorylation in cellular respiration, and photophosphorylation in photosynthesis are three major mechanisms of ATP biosynthesis.Metabolic processes that use ATP as an energy source convert it back into its precursors. ATP is therefore continuously recycled in organisms: the human body, which on average contains only 250 grams (8.8 oz) of ATP, turns over its own body weight equivalent in ATP each day.ATP is used as a substrate in signal transduction pathways by kinases that phosphorylate proteins and lipids. It is also used by adenylate cyclase, which uses ATP to produce the second messenger molecule cyclic AMP. The ratio between ATP and AMP is used as a way for a cell to sense how much energy is available and control the metabolic pathways that produce and consume ATP. Apart from its roles in signaling and energy metabolism, ATP is also incorporated into nucleic acids by polymerases in the process of transcription. ATP is the neurotransmitter believed to signal the sense of taste.The structure of this molecule consists of a purine base (adenine) attached by the 9' nitrogen atom to the 1' carbon atom of a pentose sugar (ribose). Three phosphate groups are attached at the 5' carbon atom of the pentose sugar. It is the addition and removal of these phosphate groups that inter-convert ATP, ADP and AMP. When ATP is used in DNA synthesis, the ribose sugar is first converted to deoxyribose by ribonucleotide reductase.ATP was discovered in 1929 by Karl Lohmann, and independently by Cyrus Fiske and Yellapragada Subbarow of Harvard Medical School, but its correct structure was not determined until some years later. It was proposed to be the intermediary molecule between energy-yielding and energy-requiring reactions in cells by Fritz Albert Lipmann in 1941. It was first artificially synthesized by Alexander Todd in 1948.