c2 atomic structure f pmh
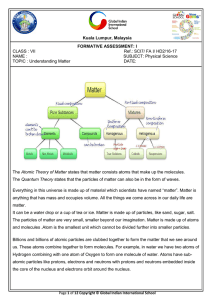
... Elements can be divided into two groups, metals and non-metals. The list below gives some properties of elements. ...
... Elements can be divided into two groups, metals and non-metals. The list below gives some properties of elements. ...
01 Intro Chemistry
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
6.1 Notes - Trimble County Schools
... Continuous spectrum = where all colors merge or blend into one another to form a continuous band ...
... Continuous spectrum = where all colors merge or blend into one another to form a continuous band ...
02Ch02chemistry2005
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
Chapter 2 Chemistry
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
... Pair of electrons not shared equally by 2 atoms Water = O + H oxygen has stronger “attraction” for the shared electrons than hydrogen oxygen has higher ...
worksheer format 11-12
... Plasma is a state of matter in which all matter is ionized and it occurs in the form of ions and electrons. Since, we know that for the ionization of the element or matter to occur energy is required to pull the electron from the attraction of the nuclear charge, high energy is required. So for the ...
... Plasma is a state of matter in which all matter is ionized and it occurs in the form of ions and electrons. Since, we know that for the ionization of the element or matter to occur energy is required to pull the electron from the attraction of the nuclear charge, high energy is required. So for the ...
rp oc4
... 14. Suppose an unknown radioactive substance with a mass of 120 g has a half-life of 6 substance will remain after 2 years? 75g ...
... 14. Suppose an unknown radioactive substance with a mass of 120 g has a half-life of 6 substance will remain after 2 years? 75g ...
Lecture 3: Electronic Band Theory: A Many
... at the ions, vanishing away. This means the wave function lives on a lattice, not in the continuum. In other words, our wave function looks like ψ(nx , ny ) instead of ψ(x, y). The consequence of this is that momenta for extended electron wave functions live on a torus. ...
... at the ions, vanishing away. This means the wave function lives on a lattice, not in the continuum. In other words, our wave function looks like ψ(nx , ny ) instead of ψ(x, y). The consequence of this is that momenta for extended electron wave functions live on a torus. ...
DART Activities for Atomic Structure
... existence of three orbitals accounts for spectral observations in the presence of an applied magnetic field (the Zeeman effect). In the presence of an applied magnetic field all three orbitals have slightly different _____(7)_______depending on their orientation with respect to the magnetic field; i ...
... existence of three orbitals accounts for spectral observations in the presence of an applied magnetic field (the Zeeman effect). In the presence of an applied magnetic field all three orbitals have slightly different _____(7)_______depending on their orientation with respect to the magnetic field; i ...
Case Study 6
... inside atoms?’ In the picture favoured by Thomson, the positive charge was distributed throughout the atom and, within this sphere, the negatively-charged electrons were placed on carefully chosen orbits – the rather subtle ‘plum-pudding’ model of the atom (see later). ...
... inside atoms?’ In the picture favoured by Thomson, the positive charge was distributed throughout the atom and, within this sphere, the negatively-charged electrons were placed on carefully chosen orbits – the rather subtle ‘plum-pudding’ model of the atom (see later). ...
Chemistry Post-Enrolment Worksheet C
... Section 3 – Balancing Chemical Equations To represent a chemical reaction we could write a word or symbol equation. At A level, you will be expected to interpret, construct and balance symbol equations. ...
... Section 3 – Balancing Chemical Equations To represent a chemical reaction we could write a word or symbol equation. At A level, you will be expected to interpret, construct and balance symbol equations. ...
Chap 2
... Analyzing the possible fragments gives you insight into the overall structure of the molecule. It’s like putting together a puzzle. ...
... Analyzing the possible fragments gives you insight into the overall structure of the molecule. It’s like putting together a puzzle. ...
6.1 Organizing the Periodic Table
... • Arranged the elements into rows in order of increasing mass so that elements with similar properties were in the same column • Left empty spaces where undiscovered elements would fit ...
... • Arranged the elements into rows in order of increasing mass so that elements with similar properties were in the same column • Left empty spaces where undiscovered elements would fit ...
Quantization of Energy
... Einstein explanation of Photoelectric effect Einstein (1905) successfully resolved this paradox by employing Planck’s idea of quantization of energy and proposed that the incident light consisted of individual quanta, called photons, that interacted with the electrons in the metal like discrete par ...
... Einstein explanation of Photoelectric effect Einstein (1905) successfully resolved this paradox by employing Planck’s idea of quantization of energy and proposed that the incident light consisted of individual quanta, called photons, that interacted with the electrons in the metal like discrete par ...
H2, N2, O2, F2, Cl2, Br2, I2
... 3. • Only change the coefficient ( the number in front of the formula ) when balancing. This tells us how many of each molecule or atom we have in the balanced equation. If there is no number in front, a " 1 " is there but we usually leave out the 1's. • Do not change subscripts to balance. They are ...
... 3. • Only change the coefficient ( the number in front of the formula ) when balancing. This tells us how many of each molecule or atom we have in the balanced equation. If there is no number in front, a " 1 " is there but we usually leave out the 1's. • Do not change subscripts to balance. They are ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.