AP Chem II Instructor: Mr. Malasky Name Period ______ Due Date
... AP Chem II Instructor: Mr. Malasky Name _______________________________________ Period ________ Due Date __________ Ch. 7A.2 Atomic Structure. Multiple Choice. Select the letter for the correct response. ____ 1. The Heisenberg uncertainty principle states that a. electrons have no momentum b. the po ...
... AP Chem II Instructor: Mr. Malasky Name _______________________________________ Period ________ Due Date __________ Ch. 7A.2 Atomic Structure. Multiple Choice. Select the letter for the correct response. ____ 1. The Heisenberg uncertainty principle states that a. electrons have no momentum b. the po ...
Light/Electrons
... Louis de Broglie suggested that matter in motion has properties that are normally associated with waves. The wave properties are especially applicable to very small particles, such as electrons. Each particle’s wavelength is related to its mass, its velocity and Planck’s constant. Smaller the mass, ...
... Louis de Broglie suggested that matter in motion has properties that are normally associated with waves. The wave properties are especially applicable to very small particles, such as electrons. Each particle’s wavelength is related to its mass, its velocity and Planck’s constant. Smaller the mass, ...
Topic 4: Classifying Elements What did the early chemists use to
... They are called periods, and there are 7 of them. What is the VERTICAL COLUMN in a periodic table called (it can have two names)? How many of these are there in the periodic table? The ...
... They are called periods, and there are 7 of them. What is the VERTICAL COLUMN in a periodic table called (it can have two names)? How many of these are there in the periodic table? The ...
A More Efficient Way to Describe Interacting Quantum Particles in 1D
... adopting a hydrodynamics approach. Here, one takes a “zoomed-out” view of the particles, such that they no longer appear as individual particles but, instead, as one continuous fluid (Fig. 1). The approach is used in many areas of physics because it replaces the complex problem of describing large ...
... adopting a hydrodynamics approach. Here, one takes a “zoomed-out” view of the particles, such that they no longer appear as individual particles but, instead, as one continuous fluid (Fig. 1). The approach is used in many areas of physics because it replaces the complex problem of describing large ...
Energy levels, photons and spectral lines
... Niels Bohr developed a model of the atom where the electrons had certain stable states that had quantized radii and energy ...
... Niels Bohr developed a model of the atom where the electrons had certain stable states that had quantized radii and energy ...
Using mass to calculate molecular formula
... Empirical formula and Molecular formula Benzene consists of 7.69% H and 92.31%C. Converting this to a formula gives CH. This is the simplest integer ratio. In fact a molecule of benzene has the formula C6H6. Empirical formula CH – simplest whole number ratio. Molecular formula C6H6 – actual number o ...
... Empirical formula and Molecular formula Benzene consists of 7.69% H and 92.31%C. Converting this to a formula gives CH. This is the simplest integer ratio. In fact a molecule of benzene has the formula C6H6. Empirical formula CH – simplest whole number ratio. Molecular formula C6H6 – actual number o ...
Review II
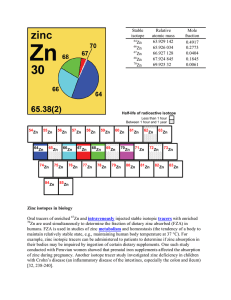
... A. Isotopes have the same chemical properties, but different masses B. Some isotopes are more prevalent than others C. Chemical symbol with mass number indicates which isotope D. Some elements have many isotopes, some very few Elements: Defined by Their Number of Protons A. Identity of atom comes fr ...
... A. Isotopes have the same chemical properties, but different masses B. Some isotopes are more prevalent than others C. Chemical symbol with mass number indicates which isotope D. Some elements have many isotopes, some very few Elements: Defined by Their Number of Protons A. Identity of atom comes fr ...
lecture 3
... something that cannot be broken down into any other substances. For example, gold, silver, and iron are elements. – A compound also is defined by its unique physical and chemical properties, but it can be broken down into simpler constituents (elements). For example, water can be broken down into hy ...
... something that cannot be broken down into any other substances. For example, gold, silver, and iron are elements. – A compound also is defined by its unique physical and chemical properties, but it can be broken down into simpler constituents (elements). For example, water can be broken down into hy ...
Assignment 1
... 1. N independent particles exist in one of the 3 non-degenerate energy levels of energies −E, 0, +E. The system is in contact with a thermal reservoir at temperature T. What is the partition function of the system? Use canonical ensemble to show that (i) The maximum possible entropy in the limit T 1 ...
... 1. N independent particles exist in one of the 3 non-degenerate energy levels of energies −E, 0, +E. The system is in contact with a thermal reservoir at temperature T. What is the partition function of the system? Use canonical ensemble to show that (i) The maximum possible entropy in the limit T 1 ...
Study Sheet for Chemistry and Physics Chemistry Atomic Structure
... (rounded to nearest whole number). The atomic mass (measured in amu – atomic mass unit) can be found by adding protons and neutrons together. Matter Atom – smallest unit of an element – can’t be broken down any further Element – cannot be broken down by any chemical means Molecule – combination of 2 ...
... (rounded to nearest whole number). The atomic mass (measured in amu – atomic mass unit) can be found by adding protons and neutrons together. Matter Atom – smallest unit of an element – can’t be broken down any further Element – cannot be broken down by any chemical means Molecule – combination of 2 ...
Packet
... 25. What is the density of a wood block with a volume of 16.5 mL and a mass of 6.39g? a. 0.3872 g/mL b. 0.387 g/mL c. 0.388 g/mL d. 0.39 g/mL 26. Calculate the density of matter that has a volume of 12.4 ml and a mass of 4.20 ...
... 25. What is the density of a wood block with a volume of 16.5 mL and a mass of 6.39g? a. 0.3872 g/mL b. 0.387 g/mL c. 0.388 g/mL d. 0.39 g/mL 26. Calculate the density of matter that has a volume of 12.4 ml and a mass of 4.20 ...
1 - WordPress.com
... example of each. Heterogeneous Mixture : A mixture that does not blend smoothly throughout – ex. sand and water Homogeneous Mixture: A mixture that has constant composition throughout – ex. salt and water 11. Explain the difference between a physical change and a chemical change. Give an example of ...
... example of each. Heterogeneous Mixture : A mixture that does not blend smoothly throughout – ex. sand and water Homogeneous Mixture: A mixture that has constant composition throughout – ex. salt and water 11. Explain the difference between a physical change and a chemical change. Give an example of ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.