1. Review (MC problems, due Monday) 2. - mvhs
... 3. A solution of barium hydroxide is titrated with 0.1-M sulfuric acid and the electrical conductivity of the solution is measured as the titration proceeds. a) For the reaction that occurs during the titration described above, write a balanced net ionic equation. (b) Explain why the conductivity de ...
... 3. A solution of barium hydroxide is titrated with 0.1-M sulfuric acid and the electrical conductivity of the solution is measured as the titration proceeds. a) For the reaction that occurs during the titration described above, write a balanced net ionic equation. (b) Explain why the conductivity de ...
Document
... classical mechanics, which predict that electrons in orbit would fall towards and collide with the nucleus. Stable Bohr atoms are not possible. • Modern quantum mechanics, with orbitals rather than orbits, provides the only reasonable explanation for the observed properties of the atoms ...
... classical mechanics, which predict that electrons in orbit would fall towards and collide with the nucleus. Stable Bohr atoms are not possible. • Modern quantum mechanics, with orbitals rather than orbits, provides the only reasonable explanation for the observed properties of the atoms ...
chapter 2
... Ethanol because it is polar so the water molecules would be attracted to the polar end of the ethanol molecule. Ethane would not mix because it is non-polar so the water molecules are not attracted to it and they would just stay together. 2. Why are some solvents polar and some non-polar? Which woul ...
... Ethanol because it is polar so the water molecules would be attracted to the polar end of the ethanol molecule. Ethane would not mix because it is non-polar so the water molecules are not attracted to it and they would just stay together. 2. Why are some solvents polar and some non-polar? Which woul ...
The Hydrogen Atom 22.1 Radial Wavefunction
... As with the infinite square well, it makes sense to let κ = −2~m E (negative inside the square root, now – bound states will have E < 0 and we want to make κ real). We want to define a new “coordinate” ρ ≡ κ r. The advantage is to render the coordinate variable itself unitless. Whenever we want to c ...
... As with the infinite square well, it makes sense to let κ = −2~m E (negative inside the square root, now – bound states will have E < 0 and we want to make κ real). We want to define a new “coordinate” ρ ≡ κ r. The advantage is to render the coordinate variable itself unitless. Whenever we want to c ...
Model of molecular bonding based on the Bohr
... infinite dimensional reduction of the Schrödinger equation [3]. The resulting electron configurations are reminiscent of the Lewis electron-dot structure introduced in 1916 [4]. The surprising feature of our work is that all molecular binding energy curves studied below can be accounted for by most ...
... infinite dimensional reduction of the Schrödinger equation [3]. The resulting electron configurations are reminiscent of the Lewis electron-dot structure introduced in 1916 [4]. The surprising feature of our work is that all molecular binding energy curves studied below can be accounted for by most ...
Pre Test - broward.k12.fl.us
... c) Are the conclusions supported by other research on the topic? d) Is the researcher well respected among leaders in the field? 3) Which of the following best describes how degrees of complexity of a system are influenced by the scale at which the system is investigated? a) The principle that energ ...
... c) Are the conclusions supported by other research on the topic? d) Is the researcher well respected among leaders in the field? 3) Which of the following best describes how degrees of complexity of a system are influenced by the scale at which the system is investigated? a) The principle that energ ...
Section1 Final Key
... 5. (10 pts) Postulates and Principles: True/False. T / F : According to the variational principle, the quantum mechanical energy is always lower than the classical energy. T / F: A spherical harmonic function Ylm (θ, φ) is an eigenfunction of the L̂2 operator with eigenvalue h̄2 l(l + 1). T / F : A ...
... 5. (10 pts) Postulates and Principles: True/False. T / F : According to the variational principle, the quantum mechanical energy is always lower than the classical energy. T / F: A spherical harmonic function Ylm (θ, φ) is an eigenfunction of the L̂2 operator with eigenvalue h̄2 l(l + 1). T / F : A ...
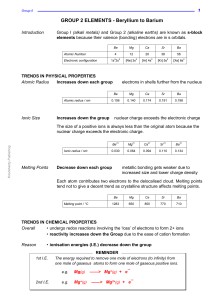
GROUP 2 ELEMENTS - Beryllium to Barium
... basic strength also increases down group this is because the solubility increases the metal ions get larger so charge density decreases there is a lower attraction between the OH¯ ions and larger dipositive ions the ions will split away from each other more easily there will be a greater concentrati ...
... basic strength also increases down group this is because the solubility increases the metal ions get larger so charge density decreases there is a lower attraction between the OH¯ ions and larger dipositive ions the ions will split away from each other more easily there will be a greater concentrati ...
CHEM 115 EXAM #1
... A basketball has a diameter of 9.4 inches. The Earth has a surface area of 196,935,000 sq miles. How many basketballs are required to cover the Earth with a single layer of basketballs? (note: if your calculator does not have a key for π, try using 22/7) ...
... A basketball has a diameter of 9.4 inches. The Earth has a surface area of 196,935,000 sq miles. How many basketballs are required to cover the Earth with a single layer of basketballs? (note: if your calculator does not have a key for π, try using 22/7) ...
10.4: Helium Atom - PhysWiki
... Hence, we conclude that in excited states of helium the spin singlet state has a higher energy than the spin triplet state. Incidentally, helium in the spin singlet state is known as para-helium, whereas helium in the triplet state is called ortho-helium. As we have seen, for the ground state, only ...
... Hence, we conclude that in excited states of helium the spin singlet state has a higher energy than the spin triplet state. Incidentally, helium in the spin singlet state is known as para-helium, whereas helium in the triplet state is called ortho-helium. As we have seen, for the ground state, only ...
Answers
... a) What is the empirical formula for the substance? b) What is the molecular formula for the substance? 8) How many liters of ammonia gas would be produced by 3.00 grams of magnesium nitride, according to the reaction Mg3N2 + 6 H2O --> 3 Mg(OH)2 + 2 NH3? 9) Solid iron (II) sulfide reacts with aqueou ...
... a) What is the empirical formula for the substance? b) What is the molecular formula for the substance? 8) How many liters of ammonia gas would be produced by 3.00 grams of magnesium nitride, according to the reaction Mg3N2 + 6 H2O --> 3 Mg(OH)2 + 2 NH3? 9) Solid iron (II) sulfide reacts with aqueou ...
QM1
... We have no right to assume that any physical laws exist, or if they have existed up until now, or that they will continue to exist in a similar manner in the future. ...
... We have no right to assume that any physical laws exist, or if they have existed up until now, or that they will continue to exist in a similar manner in the future. ...
Definitions are in Book
... the same. We can think of ∆Hof as the most direct path to forming a molecule or molecules, since this is merely the energy of the bonds in that molecule or molecules. Hess’s law says we can add the change in enthalpy of several reactions to find the change in enthalpy of a single, final reaction. Th ...
... the same. We can think of ∆Hof as the most direct path to forming a molecule or molecules, since this is merely the energy of the bonds in that molecule or molecules. Hess’s law says we can add the change in enthalpy of several reactions to find the change in enthalpy of a single, final reaction. Th ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.