Naming Ionic Compounds
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
... and classical calculations for He 3-He 4 and the experimental results is presented in Fig. 6. The He 3 nucleus has a spin of Y2, which in a suitable spin resonance experiment may be partially oriented. If the time to diffuse from one region to the other is short compared with the natural flip-over t ...
Lecture. Photoelectric Effect
... J.J. Thomson (Nobel 1906) and P. Lenard (Nobel 1905) determined the ration e/m for the particles emitted by the body under illumination – the same as for electrons. The effect remained unexplained until 1905 when Albert Einstein postulated the existence of quanta of light -- photons -- which, when a ...
... J.J. Thomson (Nobel 1906) and P. Lenard (Nobel 1905) determined the ration e/m for the particles emitted by the body under illumination – the same as for electrons. The effect remained unexplained until 1905 when Albert Einstein postulated the existence of quanta of light -- photons -- which, when a ...
The Wizard Test Maker
... 3. Two isotopes of uranium are U-237 and U-238. Both would be expected to have the same (A) mass (D) number of neutrons (B) number of protons (E) half-life (C) decay mode 4. Whose gold foil experiment concluded that the positive charge of an atom is concentrated in a small region? (A) Thompson (D) C ...
... 3. Two isotopes of uranium are U-237 and U-238. Both would be expected to have the same (A) mass (D) number of neutrons (B) number of protons (E) half-life (C) decay mode 4. Whose gold foil experiment concluded that the positive charge of an atom is concentrated in a small region? (A) Thompson (D) C ...
PYP001-122-Final Exam Solution [Choice A is the correct
... Q23. Which of the following statements is FALSE? A) Magnets made of materials that are easy to magnetize, but lose their magnetism easily, are called electromagnets. B) Magnetic induction occurs when one magnet makes another material magnetic. C) Magnets always have two poles. D) Moving electric cha ...
... Q23. Which of the following statements is FALSE? A) Magnets made of materials that are easy to magnetize, but lose their magnetism easily, are called electromagnets. B) Magnetic induction occurs when one magnet makes another material magnetic. C) Magnets always have two poles. D) Moving electric cha ...
The Periodic table
... set of orbitals of equal energy the order of filling the orbitals is such that each orbital will be occupied by one electron before any orbital receives a second electron. This minimizes the repulsion between the electrons. ...
... set of orbitals of equal energy the order of filling the orbitals is such that each orbital will be occupied by one electron before any orbital receives a second electron. This minimizes the repulsion between the electrons. ...
Lectures 10-11: Multi-electron atoms System of non
... There are two electrons => S = s1+ s2 = 0 or 1. S = 0 states are called singlets because they only have one ms value. S = 1 states are called triplets as ms = +1, 0, -1. ...
... There are two electrons => S = s1+ s2 = 0 or 1. S = 0 states are called singlets because they only have one ms value. S = 1 states are called triplets as ms = +1, 0, -1. ...
File - Flipped Out Science with Mrs. Thomas!
... does not always indicate a chemical change. Color change can also be a physical change - for example, mixing kool-aid in water. It is an expected color change. It is a chemical change only when it is unexpected- for example mixing two clear liquids and having the substance turn blue. ...
... does not always indicate a chemical change. Color change can also be a physical change - for example, mixing kool-aid in water. It is an expected color change. It is a chemical change only when it is unexpected- for example mixing two clear liquids and having the substance turn blue. ...
Electromagnetic Radiation
... 3.) When energy is absorbed by the atom, the electron moves into a higher energy orbit. This energy is released when the electron falls back to a lower energy orbit. A photon of light is emitted. ...
... 3.) When energy is absorbed by the atom, the electron moves into a higher energy orbit. This energy is released when the electron falls back to a lower energy orbit. A photon of light is emitted. ...
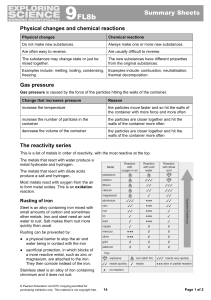
9F Reactivity - Parrs Wood High School
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
CHAPTER 3: The Experimental Basis of Quantum Theory
... In the 1890s scientists and engineers were familiar with “cathode rays”. These rays were generated from one of the metal plates in an evacuated tube across which a large electric potential had been established. It was surmised that cathode rays had something to do with atoms. It was known that catho ...
... In the 1890s scientists and engineers were familiar with “cathode rays”. These rays were generated from one of the metal plates in an evacuated tube across which a large electric potential had been established. It was surmised that cathode rays had something to do with atoms. It was known that catho ...
Atomic Physics - CAFE SYSTEM CANARIAS
... interaction of radiation with atoms marks the transition between the earlier chapters on structure and the second half of the book which covers laser spectroscopy, laser cooling, Bose–Einstein condensation of dilute atomic vapours, matter-wave interferometry and ion trapping. The exciting new develo ...
... interaction of radiation with atoms marks the transition between the earlier chapters on structure and the second half of the book which covers laser spectroscopy, laser cooling, Bose–Einstein condensation of dilute atomic vapours, matter-wave interferometry and ion trapping. The exciting new develo ...
Lecture: Resonance and Atomic
... where τ0 is the characteristic decay time of the oscillator. In other words, if the electric field is turned off, the oscillator will stop moving after time τ0 . Since the decay term is proportional to the velocity of the oscillating electron, it can be incorporated into the equation of motion by ẍ ...
... where τ0 is the characteristic decay time of the oscillator. In other words, if the electric field is turned off, the oscillator will stop moving after time τ0 . Since the decay term is proportional to the velocity of the oscillating electron, it can be incorporated into the equation of motion by ẍ ...
Chapter 19: Fermi
... • Each atom in the crystal lattice of the metal is assumed to part with some number of its outer valence electrons, which can then move freely about in the metal. ...
... • Each atom in the crystal lattice of the metal is assumed to part with some number of its outer valence electrons, which can then move freely about in the metal. ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.