Electron Notes
... • In the 1900s, scientist studied interactions of light and matter. • One experiment involved the photoelectric effect, which refers to the emission of electrons from a metal when light shines on the metal. • This involved the frequency of the light. It was found that light was a form of energy that ...
... • In the 1900s, scientist studied interactions of light and matter. • One experiment involved the photoelectric effect, which refers to the emission of electrons from a metal when light shines on the metal. • This involved the frequency of the light. It was found that light was a form of energy that ...
Unit 10: Structure and Bonding
... Atoms are electrically neutral, and the positive charge of the protons is balanced by the negative charge of the electrons. It follows that in a neutral atom: no of electrons = no of protons So, if an oxygen atom (atomic number = 8) has 8 protons, it must also have 8 electrons; if a chlorine atom (a ...
... Atoms are electrically neutral, and the positive charge of the protons is balanced by the negative charge of the electrons. It follows that in a neutral atom: no of electrons = no of protons So, if an oxygen atom (atomic number = 8) has 8 protons, it must also have 8 electrons; if a chlorine atom (a ...
Mass Spectrum – Interpretation
... Bromine has two isotopes (79Br and 81Br – each 50% abudance), so the mass spectrum of Br (one atom) will show two roughly equal peaks – ...
... Bromine has two isotopes (79Br and 81Br – each 50% abudance), so the mass spectrum of Br (one atom) will show two roughly equal peaks – ...
Excitations
... energy E=ћ. The center of a wave packet moves with the group velocity vg . That determines how fast a signal pulse propagates. Solitons In a non-linear medium, the phase velocity depends on the amplitude. The spread of a wave packet due to dispersion can be compensated by an opposite spread due to ...
... energy E=ћ. The center of a wave packet moves with the group velocity vg . That determines how fast a signal pulse propagates. Solitons In a non-linear medium, the phase velocity depends on the amplitude. The spread of a wave packet due to dispersion can be compensated by an opposite spread due to ...
Electricity notes part
... An electric field exerts a force on any charged object placed in the field. is the study of the behavior of electric charges, including how charge is transferred between objects. states that the total charge in an isolated system is constant. Charge can be transferred by friction, by contact, and by ...
... An electric field exerts a force on any charged object placed in the field. is the study of the behavior of electric charges, including how charge is transferred between objects. states that the total charge in an isolated system is constant. Charge can be transferred by friction, by contact, and by ...
Document
... The system under examination A gas can be ionized under non equilibrium conditions (too low temperature for equilibrium ionization) with constant energy dissipation, like in electric discharges, photoionized media, preshock regions, and so on. ...
... The system under examination A gas can be ionized under non equilibrium conditions (too low temperature for equilibrium ionization) with constant energy dissipation, like in electric discharges, photoionized media, preshock regions, and so on. ...
Physics SAE - broward.k12.fl.us
... Determined chemical properties of elements are largely due to the number of electrons in the outer orbits Originated the idea that photons are emitted when electrons drop from a higher energy orbit to a lower one Received the Nobel Prize for Physics for his work on quantum mechanics ...
... Determined chemical properties of elements are largely due to the number of electrons in the outer orbits Originated the idea that photons are emitted when electrons drop from a higher energy orbit to a lower one Received the Nobel Prize for Physics for his work on quantum mechanics ...
1s 2s 2p - Solon City Schools
... This slide contains classified material and cannot be shown to high school students. Please continue as if everything is normal. ...
... This slide contains classified material and cannot be shown to high school students. Please continue as if everything is normal. ...
Quantum Atom PPT - River Dell Regional School District
... Orbits are not circular. They are not uniquely defined. There is no definite path for the motion of the electron. The model predicts the probability of finding an electron a certain distance from the nucleus. The space is defined by the solution of Schrödinger equation. Orbitals are found in e ...
... Orbits are not circular. They are not uniquely defined. There is no definite path for the motion of the electron. The model predicts the probability of finding an electron a certain distance from the nucleus. The space is defined by the solution of Schrödinger equation. Orbitals are found in e ...
Homework No. 01 (Spring 2016) PHYS 530A: Quantum Mechanics II
... (a) Find the Lagrangian for this system that implies the equation of motion of Eq. (1) using Hamilton’s principle of stationary action. (b) Determine the canonical momentum for this system (c) Determine the Hamilton H(p, x) for this system. 2. (10 points.) The Hamiltonian is defined by the relation ...
... (a) Find the Lagrangian for this system that implies the equation of motion of Eq. (1) using Hamilton’s principle of stationary action. (b) Determine the canonical momentum for this system (c) Determine the Hamilton H(p, x) for this system. 2. (10 points.) The Hamiltonian is defined by the relation ...
AQA C2 revision book
... 2) The volumes of acid and alkali used are noted, and the experiment is repeated using the same volumes, but no indicator. 3) The solution is evaporated to leave the salt Method 2 Where an acid is reacted with an insoluble substance (base, metal or carbonate). 1) Some acid is measured into a beaker ...
... 2) The volumes of acid and alkali used are noted, and the experiment is repeated using the same volumes, but no indicator. 3) The solution is evaporated to leave the salt Method 2 Where an acid is reacted with an insoluble substance (base, metal or carbonate). 1) Some acid is measured into a beaker ...
Study Guide Answers
... 1. A change of state is a _c__ a. Process by which two states of matter co-exist b. Chemical change c. Physical change that converts a substance from one physical form to another 2. Particles in a ___solid_______ move slower than particles in a ____liquid or gas______. 3. Particles in a _a__ vibrate ...
... 1. A change of state is a _c__ a. Process by which two states of matter co-exist b. Chemical change c. Physical change that converts a substance from one physical form to another 2. Particles in a ___solid_______ move slower than particles in a ____liquid or gas______. 3. Particles in a _a__ vibrate ...
Chapter 10 Handouts_1
... 10-2. Two Classes of Matter In a compound, the elements are present in a specific ratio by mass according to the law of definite proportions. In a mixture, the components are not present in a specific ratio by mass. ...
... 10-2. Two Classes of Matter In a compound, the elements are present in a specific ratio by mass according to the law of definite proportions. In a mixture, the components are not present in a specific ratio by mass. ...
Chapter 10_Handouts_6
... 10-2. Two Classes of Matter In a compound, the elements are present in a specific ratio by mass according to the law of definite proportions. In a mixture, the components are not present in a specific ratio by mass. ...
... 10-2. Two Classes of Matter In a compound, the elements are present in a specific ratio by mass according to the law of definite proportions. In a mixture, the components are not present in a specific ratio by mass. ...
Solution
... 25°C. In which direction will the reaction shift in order to regain equilibrium. Show all work and explain your reasoning. We have K calculated above and here we can calculate Q. Q = 0.02, which is much less than K, so we would predict that the reaction will shift towards products to regain equilibr ...
... 25°C. In which direction will the reaction shift in order to regain equilibrium. Show all work and explain your reasoning. We have K calculated above and here we can calculate Q. Q = 0.02, which is much less than K, so we would predict that the reaction will shift towards products to regain equilibr ...
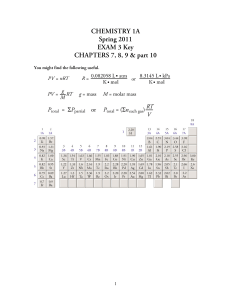
Exam 3 Key
... a. What is the hybridization for the left oxygen atom? sp3 b. What is the hybridization for the right oxygen atom? sp2 c. What is the hybridization for the top oxygen atom? sp2 d. What is the hybridization for the nitrogen atom? sp2 e. Write a description of the bonding, stating whether each bond is ...
... a. What is the hybridization for the left oxygen atom? sp3 b. What is the hybridization for the right oxygen atom? sp2 c. What is the hybridization for the top oxygen atom? sp2 d. What is the hybridization for the nitrogen atom? sp2 e. Write a description of the bonding, stating whether each bond is ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.