Reflection of light from semi-infinite absorbing turbid media. Part 1
... represents the dependence r(s) for all phase functions studied (Fig. 1). This means that reflectance measurements can be used to derive s [e.g., see Eq. (7)] and, therefore, to reduce the multiple scattering inverse problem to the single scattering one. In particular, the spectrum s() is easily simu ...
... represents the dependence r(s) for all phase functions studied (Fig. 1). This means that reflectance measurements can be used to derive s [e.g., see Eq. (7)] and, therefore, to reduce the multiple scattering inverse problem to the single scattering one. In particular, the spectrum s() is easily simu ...
Sample pages 2 PDF
... occurring 5 carat (1g) diamond costs in the region of £100,000. 3D Graphite Graphite has also been known to humans for thousands of years with the Ancient Greeks having used it in pencils just like we do today. Graphite is built up from many stacked layers of graphene planes each slightly offset fro ...
... occurring 5 carat (1g) diamond costs in the region of £100,000. 3D Graphite Graphite has also been known to humans for thousands of years with the Ancient Greeks having used it in pencils just like we do today. Graphite is built up from many stacked layers of graphene planes each slightly offset fro ...
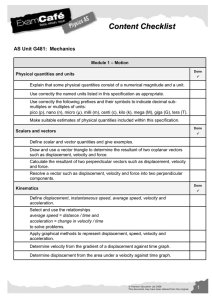
Significant Figures
... 2. All atoms of a given element are identical - all carbon atoms have the same chemical and physical properties 3. Atoms of a given element are different from those of another element. - carbon atoms have different chemical and physical properties than sulfur atoms. 4. Atoms of one element can combi ...
... 2. All atoms of a given element are identical - all carbon atoms have the same chemical and physical properties 3. Atoms of a given element are different from those of another element. - carbon atoms have different chemical and physical properties than sulfur atoms. 4. Atoms of one element can combi ...
F = 0 x = 0 F =
... PHYSICS 151 – Notes for Online Lecture 4.1 Periodicity Periodic means that something repeats itself. For example, every twenty-four hours, the Earth makes a complete rotation. Heartbeats are an example of periodic behavior. If you look at heartbeats on an electrocardiogram, they make a regular patte ...
... PHYSICS 151 – Notes for Online Lecture 4.1 Periodicity Periodic means that something repeats itself. For example, every twenty-four hours, the Earth makes a complete rotation. Heartbeats are an example of periodic behavior. If you look at heartbeats on an electrocardiogram, they make a regular patte ...
Quantum Correlated Interstitials and the Hall Resistivity of the
... that a two-dimensional electron gas should form a crystalline lattice, in the absence of disorder. A growing body of experimental evidence has accumulated in the last five years that this so-called “Wigner crystal” [1] (WC) may have been realized in high-quality heterojunction systems. Evidence of t ...
... that a two-dimensional electron gas should form a crystalline lattice, in the absence of disorder. A growing body of experimental evidence has accumulated in the last five years that this so-called “Wigner crystal” [1] (WC) may have been realized in high-quality heterojunction systems. Evidence of t ...
... contains a localised “sigma” (σ) bond which forms a strong chemical bond. In addition, every double bond also contains a less strongly localised “pi” (π) bond which is weaker. However, conjugation is not enough to make the polymer material conductive. In addition – and this is what the dopant does ...
Zprime_150411
... – Z removal: reject events containing a pair of the fakeable objects of base selection with invariant mass between 70 and 110 GeV, if one of the object also passed isEM loose cut (0.4% event rejected) ...
... – Z removal: reject events containing a pair of the fakeable objects of base selection with invariant mass between 70 and 110 GeV, if one of the object also passed isEM loose cut (0.4% event rejected) ...
OXIDATION NUMBERS
... 2 If different numbers of the relevant species are on both sides, balance them 3 Work out the oxidation number of the element before and after the change 4 Add electrons to one side of the equation so the oxidation numbers balance 5 If the charges on all the species (ions and electrons) on either si ...
... 2 If different numbers of the relevant species are on both sides, balance them 3 Work out the oxidation number of the element before and after the change 4 Add electrons to one side of the equation so the oxidation numbers balance 5 If the charges on all the species (ions and electrons) on either si ...
Lecture Notes: Condensed Matter Theory I (TKM1)
... Condensed matter physics is concerned with the behavior of large aggregates of atoms or molecules in liquid or solid form. It is one of the largest branches of physics, with a wide variety of di¤erent systems, approaches, challenges and concepts. Often, it is subdivided in soft condensed matter phys ...
... Condensed matter physics is concerned with the behavior of large aggregates of atoms or molecules in liquid or solid form. It is one of the largest branches of physics, with a wide variety of di¤erent systems, approaches, challenges and concepts. Often, it is subdivided in soft condensed matter phys ...
We have provided a template for your use in submitting Multiple
... Describe constructive interference and destructive interference in terms of path difference and phase difference. Use the relationship intensity ∝ amplitude2. Describe the Young double-slit experiment and explain how it is a classical confirmation of the wave nature of light. Select and use the equa ...
... Describe constructive interference and destructive interference in terms of path difference and phase difference. Use the relationship intensity ∝ amplitude2. Describe the Young double-slit experiment and explain how it is a classical confirmation of the wave nature of light. Select and use the equa ...
Excitation of Rydberg states in rubidium with near infrared diode lasers
... With the application of a small electric field, a dipole-dipole energy exchange between pairs of atoms can be tuned into resonance [1, 2]. This interaction can also lead to a blockade effect, which limits the number of excited Rydberg atoms [3, 4] and can even limit the excitation in a small sample ...
... With the application of a small electric field, a dipole-dipole energy exchange between pairs of atoms can be tuned into resonance [1, 2]. This interaction can also lead to a blockade effect, which limits the number of excited Rydberg atoms [3, 4] and can even limit the excitation in a small sample ...
Document
... * Complex ions of the same central atom but different ligands have different colours. Colours of various complex ions formed by copper(II) ion are listed below : ...
... * Complex ions of the same central atom but different ligands have different colours. Colours of various complex ions formed by copper(II) ion are listed below : ...
4. Photometric Concepts and Magnitudes
... Depending on the method of observation, we can define various magnitude systems. Different magnitudes have different zero points, i. e. they have different flux densities F0 corresponding to the magnitude 0. The zero points are usually defined by a few selected standard stars. In daylight the human ...
... Depending on the method of observation, we can define various magnitude systems. Different magnitudes have different zero points, i. e. they have different flux densities F0 corresponding to the magnitude 0. The zero points are usually defined by a few selected standard stars. In daylight the human ...
Nature of chemical reaction - Environmental-Chemistry
... Energy and chemical reactions: • Chemical reactions are breaking of old bonds from reactant-molecules and formation of new bonds in product-molecules. • Chemical reactions involve changes in energy. Photosynthesis is an endothermic reaction. • Energy is released (exothermic) during formation of bon ...
... Energy and chemical reactions: • Chemical reactions are breaking of old bonds from reactant-molecules and formation of new bonds in product-molecules. • Chemical reactions involve changes in energy. Photosynthesis is an endothermic reaction. • Energy is released (exothermic) during formation of bon ...
Chemistry 101L
... will be making. Remember to include room for multiple trials and average values, if appropriate. If appropriate, have room for classmates’ data. Now organize your list into things that are similar or data that should be compared. Tables columns/rows do not have to be listed in the same order that th ...
... will be making. Remember to include room for multiple trials and average values, if appropriate. If appropriate, have room for classmates’ data. Now organize your list into things that are similar or data that should be compared. Tables columns/rows do not have to be listed in the same order that th ...
ready
... neutron to the nuclei. The nuclear burning rates1 of the fusion between different nuclei depends sensitively on temperature and pressure. The large dependence on temperature comes from the thermal motions needed to overcome the repulsion between two nuclei. The repulsion between two nuclei is the co ...
... neutron to the nuclei. The nuclear burning rates1 of the fusion between different nuclei depends sensitively on temperature and pressure. The large dependence on temperature comes from the thermal motions needed to overcome the repulsion between two nuclei. The repulsion between two nuclei is the co ...
2016 - Specimen Paper 4 - Cambridge International Examinations
... Chromium is a transition element. (a) (i) State two differences in the physical properties of chromium and sodium. ...
... Chromium is a transition element. (a) (i) State two differences in the physical properties of chromium and sodium. ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.