How Cells Harvest Energy: Cellular Respiration
... The human body uses energy from ATP for all its activities ...
... The human body uses energy from ATP for all its activities ...
CELLULAR RESPIRATION
... Cellular Respiration - Cells transfer energy from food molecules to ATP, the byproduct is water and carbon dioxide ...
... Cellular Respiration - Cells transfer energy from food molecules to ATP, the byproduct is water and carbon dioxide ...
ADP, ATP and Cellular Respiration Powerpoint
... • An Oxidation-Reduction Process or REDOX Reaction • Oxidation of GLUCOSE --> CO2 + H2O (e- removed from C6H12O6) • Reduction O2 to H2O (epassed (added) to O2) ...
... • An Oxidation-Reduction Process or REDOX Reaction • Oxidation of GLUCOSE --> CO2 + H2O (e- removed from C6H12O6) • Reduction O2 to H2O (epassed (added) to O2) ...
micro notes chpt. 8
... a. Chemiosmosis involves the creation of a proton gradient across a membrane by the electron transport system and use of the gradient and proton diffusion (proton motive force or PMF) to power a membrane-embedded ATP synthase, which converts ADP and inorganic phosphate to ATP by oxidative phosphoryl ...
... a. Chemiosmosis involves the creation of a proton gradient across a membrane by the electron transport system and use of the gradient and proton diffusion (proton motive force or PMF) to power a membrane-embedded ATP synthase, which converts ADP and inorganic phosphate to ATP by oxidative phosphoryl ...
Module 3 Notes
... o Yields ____________ than aerobic respiration because only part of the Krebs cycle operates under ___________________ conditions _____________ sources can be used o Eg, can oxidize lipids, _____________ Polymers broken down by enzymes Smaller subunits enter catabolism at various points of gly ...
... o Yields ____________ than aerobic respiration because only part of the Krebs cycle operates under ___________________ conditions _____________ sources can be used o Eg, can oxidize lipids, _____________ Polymers broken down by enzymes Smaller subunits enter catabolism at various points of gly ...
lec33_F2015
... to water. Note that the oxygen only serves as a final acceptor of electrons in this process. In many organisms other compounds besides oxygen can serve as electron sinks, allowing organisms to perform 'oxidative' phosphorylation in the absence of O2. The actual synthesis of ATP is from a proton grad ...
... to water. Note that the oxygen only serves as a final acceptor of electrons in this process. In many organisms other compounds besides oxygen can serve as electron sinks, allowing organisms to perform 'oxidative' phosphorylation in the absence of O2. The actual synthesis of ATP is from a proton grad ...
MICR 201 Microbiology for Health Related Sciences
... ATP contains energy that can be easily released (highenergy or unstable energy bond) Required for anabolic reactions Produced by ...
... ATP contains energy that can be easily released (highenergy or unstable energy bond) Required for anabolic reactions Produced by ...
ELECTRON TRANSPORT CHAIN (student)
... • NADH + FADH2 eventually transfer the electrons they carry to a series of proteins that are located in the inner membrane • The components of the ETC are arranged in order of increasing electronegativity • Thus, allowing the electrons to flow, or BE TRANSPORTED, between the compounds • Every step i ...
... • NADH + FADH2 eventually transfer the electrons they carry to a series of proteins that are located in the inner membrane • The components of the ETC are arranged in order of increasing electronegativity • Thus, allowing the electrons to flow, or BE TRANSPORTED, between the compounds • Every step i ...
Practice exam #1 review
... 3. Delta G is negative when the products have less free energy that the reactants T F 4. In the synthesis of ATP the products have less free energy that the reactants T F 5. When a reaction is spontaneous Delta G is negative T F 6. Kinetic energy is called Delta G T F 7. Oxidized NAD+ is highly ener ...
... 3. Delta G is negative when the products have less free energy that the reactants T F 4. In the synthesis of ATP the products have less free energy that the reactants T F 5. When a reaction is spontaneous Delta G is negative T F 6. Kinetic energy is called Delta G T F 7. Oxidized NAD+ is highly ener ...
Note 4.1 - Cellular Respiration
... energy from organic components. Obligate aerobe – is an organism that cannot live without oxygen. Most eukaryotes, plants and animals, and some prokaryotes are obligate aerobes, requiring oxygen to live. They oxidize glucose to obtain free energy, but they can also use lipids and proteins to obtain ...
... energy from organic components. Obligate aerobe – is an organism that cannot live without oxygen. Most eukaryotes, plants and animals, and some prokaryotes are obligate aerobes, requiring oxygen to live. They oxidize glucose to obtain free energy, but they can also use lipids and proteins to obtain ...
UNIT 7 Metabolism and generation of ATP
... 4.5 Reduced electron carriers donate their electrons to acceptor molecules and become reoxidized in the process. The acceptor molecules are reduced because the oxidation of one species (e.g., the reduced electron carrier) cannot occur without the simultaneous reduction of another species (e.g., the ...
... 4.5 Reduced electron carriers donate their electrons to acceptor molecules and become reoxidized in the process. The acceptor molecules are reduced because the oxidation of one species (e.g., the reduced electron carrier) cannot occur without the simultaneous reduction of another species (e.g., the ...
Chapter 7 Active Reading Guide
... 16. The starting product of glycolysis is the six-carbon sugar __________, and the ending products are two __________-carbon molecules of ___________________. 17. The ten individual steps of glycolysis can be divided into two stages: energy investment and energy payoff. These steps are shown in Figu ...
... 16. The starting product of glycolysis is the six-carbon sugar __________, and the ending products are two __________-carbon molecules of ___________________. 17. The ten individual steps of glycolysis can be divided into two stages: energy investment and energy payoff. These steps are shown in Figu ...
Anaerobic metabolism is the production of ATP with oxygen
... 2. True or False: An enzyme is not changed by the reaction it causes. 3. True or False: An enzyme does not need to fit precisely with the reactant to catalyze the reaction. 4. True or False: The electron transport system is where most of the ATP is produced during aerobic metabolism. 5. True or Fals ...
... 2. True or False: An enzyme is not changed by the reaction it causes. 3. True or False: An enzyme does not need to fit precisely with the reactant to catalyze the reaction. 4. True or False: The electron transport system is where most of the ATP is produced during aerobic metabolism. 5. True or Fals ...
Assignment 6 Cell Respiration
... passing continues through the rest of the transport molecules. The FADH2’s have to drop off their electrons farther down the chain, but the transport mechanism is the same. As the electrons are passed from cytochrome to cytochrome they lose energy and the energy loss is used to pump the hydrogen ion ...
... passing continues through the rest of the transport molecules. The FADH2’s have to drop off their electrons farther down the chain, but the transport mechanism is the same. As the electrons are passed from cytochrome to cytochrome they lose energy and the energy loss is used to pump the hydrogen ion ...
lecture 02b
... • In biological redox reactions, electrons are often accompanied by protons (e.g. dehydrogenations) • In understanding metabolism, we are not only concerned with electrons but also protons. – Also called hydrogen ions or H+ – H+ (hydrogen ions) and electrons are opposites!! Don’t get them confused! ...
... • In biological redox reactions, electrons are often accompanied by protons (e.g. dehydrogenations) • In understanding metabolism, we are not only concerned with electrons but also protons. – Also called hydrogen ions or H+ – H+ (hydrogen ions) and electrons are opposites!! Don’t get them confused! ...
Metabolism Objective Project
... by blocking substrates from entering active sites Non competitive inhibitors bind away from the active site, but change the shape of the enzyme so the active ...
... by blocking substrates from entering active sites Non competitive inhibitors bind away from the active site, but change the shape of the enzyme so the active ...
OXIDATIVE PHOSPHORYLATION
... generated in succinate dehydrogenase or (succinate-Q reductase) in the citric acid cycle, to Q-cytochrome c oxidoreductase Cytochrome c, a small, soluble protein, shuttles electrons from Q-cytochrome c oxidoreductase to cytochrome c oxidase (complex IV), which catalyzes the reduction of O2. Succinat ...
... generated in succinate dehydrogenase or (succinate-Q reductase) in the citric acid cycle, to Q-cytochrome c oxidoreductase Cytochrome c, a small, soluble protein, shuttles electrons from Q-cytochrome c oxidoreductase to cytochrome c oxidase (complex IV), which catalyzes the reduction of O2. Succinat ...
Photosynthesis and Cellular Respiration
... CO2 mixes with water helping maintaining the blood’s pH (around 7.5). Too much carbon dioxide causes to pH to lower, so CO2 has to leave on a continuous process. ...
... CO2 mixes with water helping maintaining the blood’s pH (around 7.5). Too much carbon dioxide causes to pH to lower, so CO2 has to leave on a continuous process. ...
Fig. 5-1
... and the inner membrane of mitochondria in eukaryotic cells. Electrons are transferred along the electron transport chain, protons are pumped outside the membrane, so the ions’ concentration is higher outside. This produces a force that drives the protons back into the cell or mitochondrial matrix. I ...
... and the inner membrane of mitochondria in eukaryotic cells. Electrons are transferred along the electron transport chain, protons are pumped outside the membrane, so the ions’ concentration is higher outside. This produces a force that drives the protons back into the cell or mitochondrial matrix. I ...
Ch 7 outline
... into three stages. 2. In the first stage, acetyl-CoA joins the cycle, binding to a four-carbon molecule to produce a six-carbon molecule. 3. Second, two carbons are removed as CO2, their electrons donated to NAD+, and a fourcarbon molecule is left. 4. Third, more electrons are extracted and taken aw ...
... into three stages. 2. In the first stage, acetyl-CoA joins the cycle, binding to a four-carbon molecule to produce a six-carbon molecule. 3. Second, two carbons are removed as CO2, their electrons donated to NAD+, and a fourcarbon molecule is left. 4. Third, more electrons are extracted and taken aw ...
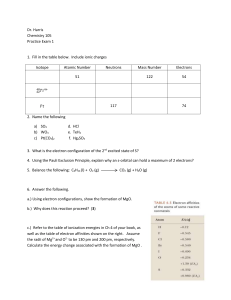
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 3. What is the electron configuration of the 2nd excited state of S? 4. Using the Pauli Exclusion Principle, explain why an s-orbital can hold a maximum of 2 electrons? 5. Balance the following: C6H14 (l) + O2 (g) ...
... 3. What is the electron configuration of the 2nd excited state of S? 4. Using the Pauli Exclusion Principle, explain why an s-orbital can hold a maximum of 2 electrons? 5. Balance the following: C6H14 (l) + O2 (g) ...
chapter 23
... Oxidative phosphorylation: the production of ATP from ADP and Pi using the energy released during the electron transport chain Chemiosmotic model links this energy to a proton gradient. As electrons are passed along the chain, H+ is passed into the intermembrane space. A proton gradient is c ...
... Oxidative phosphorylation: the production of ATP from ADP and Pi using the energy released during the electron transport chain Chemiosmotic model links this energy to a proton gradient. As electrons are passed along the chain, H+ is passed into the intermembrane space. A proton gradient is c ...
Quiz #3 - San Diego Mesa College
... A) the combustion of wood B) the combustion of gasoline in a car engine C) the metabolism of glucose in a living cell D) the build-up of glucose from carbon dioxide and water E) the breakdown of brown fat in babies to generate heat Q. 4: An ATP molecule is made of following molecular components A) t ...
... A) the combustion of wood B) the combustion of gasoline in a car engine C) the metabolism of glucose in a living cell D) the build-up of glucose from carbon dioxide and water E) the breakdown of brown fat in babies to generate heat Q. 4: An ATP molecule is made of following molecular components A) t ...
Cellular Respiration PPT 12-13-Cooke
... – Pyruvate is converted to Acetly-CoA, releasing CO2 as a waste – Acetyl-CoA is added to a 4-carbon molecule to make citric acid – Citric acid goes through a series of reactions producing ATP, and NADH & FADH2 that carry high energy electrons to the Electron Transport Chain ...
... – Pyruvate is converted to Acetly-CoA, releasing CO2 as a waste – Acetyl-CoA is added to a 4-carbon molecule to make citric acid – Citric acid goes through a series of reactions producing ATP, and NADH & FADH2 that carry high energy electrons to the Electron Transport Chain ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.