* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Herpes Simplex Virus Lec. 7
Epigenetics of neurodegenerative diseases wikipedia , lookup
DNA supercoil wikipedia , lookup
Ridge (biology) wikipedia , lookup
Viral phylodynamics wikipedia , lookup
Epigenomics wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Biology and consumer behaviour wikipedia , lookup
Genome evolution wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Gene expression profiling wikipedia , lookup
Point mutation wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Microevolution wikipedia , lookup
Genomic library wikipedia , lookup
Designer baby wikipedia , lookup
Non-coding DNA wikipedia , lookup
Minimal genome wikipedia , lookup
DNA vaccination wikipedia , lookup
Epigenetics of human development wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Helitron (biology) wikipedia , lookup
History of genetic engineering wikipedia , lookup
Extrachromosomal DNA wikipedia , lookup
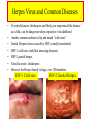
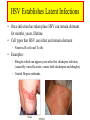
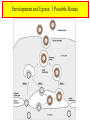
HERPES SIMPLEX VIRUS Characteristics of HSV • • • • • • DNA double stranded virus, linear 125-250 Kb long, relatively big Enveloped Virion size 200 nm, relatively big 9 HSVs, Ex. Varicella, EBV, CMV Diseases: Chickenbox, Mononucleosis, Hepatitis, Encephalitis • Recurrent eye, mouth and genital lesions Chickenpox, Varicella Zoster Herpes Virus and Common Diseases • Everybody knows chickenpox and likely you experienced the disease as a child, can be dangerous when exposed to it in adulthood • Another common ailment is lip and mouth “cold sores” • Genital Herpes lesions caused by HSV, sexually transmitted • HSV-1 cold sores (mild but annoying diseases) • HSV-2 genital herpes • Varicella zoster: chickenpox • However the Herpes family is huge, over 100 members HSV-1 Cold sore HSV-2 Genital Herpes HSV Establishes Latent Infections • Once infection has taken place HSV can remain dormant for months, years, lifetime • Cell types that HSV can infect and remain dormant – Neurons, B-cells and T-cells • Examples: – Shingles which can appear years after first chickepox infection (caused by varicella zoster, causes both chickenpox and shingles) – Genital Herpes outbreaks Herpes (1-2) Simplex Virus Genome HSV Capsid • • • • Enclosed in an envelope Capsid has icosahedral structure Capsid is bilayered Constructed from 6 proteins – VP5 is the main one • Envelope contains at least 10 different glycoproteins gB-gM • Envelope also contains non-glycosylated proteins HSV Entry Into Host Occurs Via Heparan Sulfate Proteoglycans • • • • • • • • gB and gC bind to host glycoproteins with heparan sulfate moieties (repeating dissacharide: glucoronic and n-acetyl glucosamine) Following gB and gC is gD which binds to nectin1D OR HVEM (herpes virus entry mediator) Fusion occurs between viral envelope and host membrane Nucleocapsid is released into cytosol OR in acidified endosomes Transport to nuclear envelope occurs via T and capsid interaction DNA is released into nucleus Capsid disintegrates http://www.dipartimentobiologia.it/doctoraltraining/campadelli.htm Genome Expression in Nucleus • Viral DNA is circularized once inside nucleus • Viral DNA is localized in regions referred to as ND10 (nuclear domain 10) • Viral genes transcribed by cellular RNA Poly II • Gene expression divided into 4 groups • Group occurs within hours of viral infection (these genes also referred to as “immediate early genes”) • genes (early genes) transcription occurs 4-8 hrs past infection – genes involved with viral DNA replication • 1 and 2 (late genes) are the bulk of viral genes Tegument Proteins • -TIF (a-trans-inducing factor) interacts w/Oct-1 and HCF-1 (both cellular proteins) • Significantly increases transcription of viral genes • Vhs (virion host shutoff) protein – This protein interacts with cellular proteins – Mediates degradation of both cellular and viral mRNAs – Degradation rate of viral is much lower compared to cellular, therefore they dominate Genes Set Stage For Viral DNA Replication • • • • HSV makes its own DNA polymerase 3 Replication Origins (2 oriS, oriL) Viral DNA is circularized UL9 binds ori S and unwind dsDNA, ICP8 helps in stabilizing ss DNA • UL5, UL8 and UL52 (referred to as DNA helicase-primase complex) bind ss DNA and synthesize RNA primers • UL30 (DNA polymerase) replicates DNA • UL42 significantly enhances processivity Viral Genes Block Immune Response • Out of 84 genes only 37 involved in replication • Some of the remainder involved in blocking immune response against virus • Vhs and ICP27 block interferon effects by degrading cellular mRNAs • ICP47 binds transporter proteins that aid antigen presentation – Self and viral peptides are constantly being presented thru MHC I and provoke immune responses when appropriate – ICP47 prevents transport of viral peptides on surface of cell – no viral antigen presentation which means no immune response Viral Genes Block Immune Response HSV Latency • Latency is typical in HSVs • In case of infected neurons retrograde transport occurs and virus gains access to nucleus and can stay dormant for years • Latency is attributed to – Limited amount of VP16 (viral tegument protein) enters nucleus – No VP16 no gene expression • Neurons contain Luman and Zhangfei transcription factors – These transcription factors bind HCF-1 and inhibit formation of transcription complex Oct-1/HCF-1/VP16 • Only viral transcription that takes place is LAT’s (Latency associated transcripts) Envelopment and Egress: 3 Possible Routes Envelopment and Egress: 3 Possible Routes • HSV nucleocapsids are assembled in the nucleus • It is thought that nuclear membrane is the source of the envelope • Budding occurs from inner nuclear membrane to nuclear lumen • Three theories are currently used to describe the transport from nucleus to outside the cell • One theory predicts that virions exit nucleus without envelope thru nuclear pores (they enlarge to accommodate exit) They gain envelops in the cytosol by mixing with fragmented golgi fragments