* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Haloalkanes-haloarenes
Physical organic chemistry wikipedia , lookup
Aromaticity wikipedia , lookup
Asymmetric induction wikipedia , lookup
Aromatization wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Stille reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Ene reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
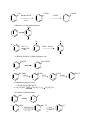
HALOALKANES & HALOARENES – Gr 12 Give reasons: 1. For the preparation of an alkyl chloride from alcohol, thionyl chloride( SOCl2 )is preferred over other reagents. A. Because the by-products, SO2 & HCl are escapable gases which leave pure alkyl chloride as product. 2. In preparation of R–I from R–OH, NaI or KI with H3PO4 is used but not H2SO4. A. Because H2SO4 converts KI or NaI to HI & then oxidise HI to I2. 3. The order of reactivity of alcohols to give haloalkanes with HX is 3o>2o>1o. A. Due to e releasing inductive effect of alkyl group(+I effect), the polarity of C–O bond is in the order 3o>2o>1o. CH3 CH3 C-OH > CH3 CH3 CH CH > CH3 CH2 CH2 OH OH 4. Aryl halides cannot be prepared from phenol, like the preparation alkyl halide from alcohol. A. Due to resonance C–O in phenol has partial double bond character and is more difficult to break than C–O single bond in alcohols. 5. Preparation of iodobenzene from benzene by electrophilic substitution requires the presence of an oxidizing agent – HNO3 or HIO3. A. The reaction is reversible. Backward reaction is prevented by oxidation of HI formed during iodination by HNO3 or HIO3. 6. Haloalkanes are only slightly soluble in water. A. A substance will dissolve readily in a solvent when the solute-solvent intermolecular forces are stronger than the solute-solute as well as solvent-solvent forces. In case of haloalkanes, the attractive force with water is weaker than the hydrogen bonds between water molecules. Therefore, solubility is low. 7. Boiling points of haloalkanes are considerably higher than those of hydrocarbons of comparable molecular mass. A. Due to polarity of C–X bond, the intermolecular forces of attraction are stronger, i.e., dipole-dipole forces, whereas hydrocarbons are non-polar and the intermolecular forces are only weaker dispersion forces. 8. For the same alkyl group, the boiling point decreases in the order – RI > RBr > RCl > RF. 1 A. With increase in size and mass of halogen atom, the magnitude of intermolecular forces increases. 9. The order of boiling point for isomeric haloalkanes is in the order 1o > 2o > 3o, CH e.g.: CH3CH2CH2CH2Br > CH3CH2–CH–CH3 > CH3–C–CH3 Br Br A. Boiling point decreases with increase in branching, as size of molecule decreases. 10.Melting point of p-dibromobenzene is higher than its o- & m- isomers. A. Due to the symmetry of its structure, p-isomer fits better in the crystal lattice. 11. Free radical halogenation of alkanes is not preferred for the preparation of haloalkanes. A. This method gives a complex mixture of isomeric mono and polyhaloalkanes which is difficult to separate. Consequently the yield of any one compound is low. 12. Haloalkanes react with KCN to form alkyl cyanides RCN as main product while AgCN forms isocyanides as main product. A. Answer on page 293. 13. The order of reactivity of alkyl halides by SN2 mechanism is 1o > 2o > 3o. A. Bulky alkyl groups on or near the C with halogen atom cause steric hindrance to the approaching nucleophile. Of the pr. alkyl halides, methyl halide is the most reactive. 14. The order of reactivity of alkyl halides by SN1 mechanism is 3o > 2o > 1o. A. Rate of reaction depends on the slowest step, which involves the formation of carbocation. Greater the stability of the carbocation, greater will be its ease of formation from alkyl halide and faster the reaction. A tertiary carbocation is the most stable of the three, then comes 2o and least stable is 1o. So the order is 3o > 2o > 1o. 15. Allylic and benzylic halides show high reactivity towards SN1 reaction. A. The carbocations formed here are stabilised by resonance. H2C=CH–CH2+ H2C+ CH=CH2 (allyl carbocation) + + CH2 CH2 CH2 CH2 + + 16. For a given alkyl group, the reactivity of R–X in both the mechanisms is RI > RBr > RCl > RF. A. As size of halogen atom increases the R–X bond weakens and can be broken more easily. 17. A racemic mixture is optically inactive. 2 A. Racemic mixture is a 50:50 mixture of enantiomers and the optical rotation of one enatiomer is opposite to the other. 18. An alkyl halide with aq. KOH undergoes nucleophilic substitution, while with alc. KOH undergoes elimination. A. Answer on page 301. 19. Aryl halides X are extremely less reactive towards nucleophilic substitution reactions. A. i) C–X bond in haloarenes is less polar than in haloalkanes because the C in haloarenes is sp2 hybridised which is sp3 hybridised and less electronegative. ii) Due to resonance C–X bond in haloarenes acquires partial double bond character and is more difficult to break. iii) Phenyl carbocation is not resonance stabilized so SN1 mechanism is ruled out. iv) Because of possible repulsion e rich nucleophiles are less likely to approach e rich arenes. 20. The dipole moment of is lower than that of cyclohexyl chloride . Cl Cl A. C–Cl bond in haloarenes is less polar due to sp2 hybridised C which is more electronegative than sp3 hybridised C in Cl 21. Reactivity towards nucleophilic substitution is in order Cl Cl O2N N2O Cl Cl NO2 NO2 NO2 NO2 A. Nucleophilic substitution in haloarenes is difficult (refer Q. 19). The presence of e withdrawing groups like –NO2 at ortho- and para- positions increases the reactivity. –NO2 groups withdraw the e density from the benzene ring and then facilitate the attack of the nucleophile. The carbanion thus formed is stabilised by resonance. More the no. of –NO2 groups at o- and p- positions, greater the stability of the carbanion. 22. There is no difference in the reactivity of haloarenes if –NO2 is in m- position. A. In none of the resonating structures of the carbanion, negative charge is on the C bearing –NO2 group. Therefore –NO2 at m- position does not stabilise the carbanion. 23. Electrophilic substitution in haloarenes X occurs at o- and p- positions. A. Due to resonance e density is high in o- and p- positions. 3 24. Although Cl is an electron withdrawing group, yet it is o- & p- directing in electrophilic substitution reactions. A. Answer on page 306. 25. Grignard reagent should be prepared under anhydrous conditions. A. Grignard reagent is highly reactive and reacts with any source of proton to give hydrocarbon. They react with water, alcohol, amines. 26. Chloroform is stored in closed dark coloured bottles filled to the brim. A. CHCl3 is slowly oxidised by air in the presence of light to form extremely poisonous gas, phosgene COCl2. 2CHCl2 + O2 light 2COCl2 + 2HCl ******************************** Important conversions: Q. 10.19 (textbook) – i) Propene to propan–1–ol CH3–CH=CH2 to CH3–CH2–CH2–OH A. CH3–CH2=CH2 + HBr org. peroxide CH3–CH2–CH2Br ii) Ethanol to butyne–1 CH3–CH2OH A. CH3CH2OH HBr CH3–CH2 –CCH CH3CH2BR CHC–Na NaOH (aq) CH3–CH2–CH2–OH CH3–CH2–CCH iii) 1–Bromopropane to 2–bromopropane CH3CH2CH2Br CH3CH–CH3 A. CH3CH2CH2Br alc. KOH iv) Toluene to benzyl alcohol CH3 Br CH3–CH=CH2 HBr CH2OH A. 4 CH3–CH–CH3 Br KMnO4 / KOH LiAlH4 + CH3 H3O COOH KMnO4/KOH CH2OH LiAlH4 H3O+ v) Benzene to 4–Bromonitrobenzene NO2 Br Br Br A. Br2 / Fe dark HNO3 / H2SO4 NO2 vi) Benzyl alcohol to 2–phenyl ethanoic acid CH2OH A . CH2COOH CH2COOH SOCl2 CH2OH KCN CH2Cl H2O/H+ vii) Ethanol to propane nitrile CH3CH2OH to CH3CH2CN A. CH3CH2OH CH3CH2Cl viii) Aniline to chlorobenzene NH2 A. KCN CH3CH2CN Cl N2+ Cl- NH2 NaNO2/HCl 0 5o C Cl Cu2Cl2 HCl 5 CH2CN 0 – 5oC HCl ix) 2–Chlorobutane to 3,4–Dimethyl hexane CH3–CH–CH2CH3 to CH3–CH2–CH–CH–CH2–CH3 Cl CH3 CH3 Na A. 2 CH3CH2–CH–Cl CH3–CH2–CH–CH–CH2–CH3 dry ether CH3 CH3 CH3 x) 2–methyl–1–propene to 2–chloro–2–methyl propane Cl CH3–C=CH2 CH3–C–CH3 CH3 CH3 Cl A. CH3–C=CH2 + HCl CH3–C–CH3 CH3 CH3 xi) Ethyl chloride to propanoic acid CH3CH2Cl CH3CH2COOH A. CH3CH2Cl KCN CH3CH2CN CH3CH2COOH xii) But–1–ene to n–butyl iodide CH3–CH2–CH=CH2 CH3CH2CH2CH2–I HBr A. CH3–CH2–CH=CH2 CH3–CH2–CH2–CH2Br xiii) 2–chloropropane to 1–propanol CH3–CH–CH3 CH3CH2CH2OH Cl A. CH3–CH–CH3 alc. KOH CH3–CH=CH2 HBr Cl xiv) Isopropyl alcohol to iodoform CH3–CH–OH CHI3 CH3 A. CH3–CH–CH3 OH CHI3 + CH3COONa 6 NaI CH3CH2CH2CH2I CH3–CH2–CH2Br NaOH CH3CH2CH2OH xv) Chlorobenzene to p-nitrophenol A. Cl Cl OH HNO3/H2SO4 NaOH/443K H+ NO2 NO2 xvi) 2–Bromopropane to 1–bromopropane A. CH3–CH–CH3 alc. KOH CH3–CH=CH2 Br xvii) Chloroethane to butane A. CH3–CH2Cl Na CH3–CH2–CH2–CH3 xvii) Benzene to diphenyl A. HBr CH3–CH2–CH2Br Cl Cl2/Fe dark Na ether (Fittig Rn.) xix) tert-butylbromide to isobutylbromide A. CH3 CH3–C–CH3 alc.KOH CH3–C=CH2 HBr CH3 – CH – CH2Br Br CH3 org. peroxide CH3 xx) Aniline to phenyl isocyanide A. NH2 NC CHCl3 & + KCl + H2O alc. KOH phenyl isocyanide **************************** Q 10.11 (textbook-Pg 311) i) Ethanol to but–1–yne A. CH3CH2OH CH3CH2Cl + CHC–Na CH3CH2–CCH ii) Propene to 1–nitropropane A. CH3–CH=CH2 HBr CH3CH2–CH2–NO2 CH3CH2CH2Br iii) Toluene to benzyl alcohol A. CH3 Cl2 hυ CH2Cl aq. KOH or aq. NaOH 7 CH2OH iv) Propene to propyne A. CH3–CH=CH2 + Br2(aq.) CH3–CCH CH3–CH–CH2Br alc. KOH CH3–CH=CHBr v) Ethanol to ethyl fluoride A. CH3CH2OH CH3CH2Cl AgF CH3CH2F vi) Bromomethane to propanone A. CH3Mg KCN CH3–C=NMgBr CH3 CH3CN ether CH3–C–CH3 O vii) But–1–ene to but–2–ene alc. KOH A. CH3–CH2–CH=CH2 HBr CH3–CH2–CH–CH3 CH3–CH=CH–CH3 │ Br viii) 1–chlorobutane to n–octane A. CH3CH2CH2–CH2Cl Na CH3CH2CH2–CH2–CH2–CH2–CH2–CH3 ix) Benzene to biphenyl A. Br2 FeBr3 Br Na dry ether x) Bromobenzene to benzene A. Br + Mg dry ether MgBr H2 O ************************************** Practice the following conversions: 1) 2–bromo–2–methyl propane to 2–methyl propene 2) Benzene to 2–nitrophenol 3) Sodium ethoxide to dimethyl ether 4) Bromobenzene to benzene 5) Benzene to toluene 6) Bromoethane to ethane 7) Propene to 1–nitropropane 8) Propene to propyne 9) Ethanol to ethyl fluoride 10) But–1–ene to but–2–ene 11) 1–chlorobutane to n–octane ******************************************* 8 Br + Mg OH